Abstract
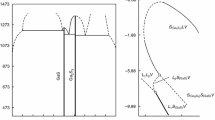
Experimental data for the system GaS–GaSe were subjected to a critical assessment using the differential thermal analysis (DTA), X-ray diffraction and thermodynamic approach. The physicochemical and thermodynamic parameters of the GaS and GaSe compounds were taken from the literature and the authors’ previous assessment, respectively. To reach a self-consistent thermodynamic description for the constituent phases in the system, the experimental on the melting point, the composition of the minimum point and the univariant curves of the GaS–GaSe system were reassessed. An ideal solution model for the liquid, which included non-molecular compounds, was employed to represent phase diagram and Gibbs free energy of mixing data. To make our investigation on invariant points more accurate, a new and complementary experimental DTA determination regarding the compounds was carried out. We clarify the temperature and melting enthalpy of the compounds that are needed to calculate thermodynamic parameters of the system. Our thermodynamic description, compatible with experimental data for the GaS–GaSe system, resulted in an agreement between the calculated and experimental data. The temperature–concentration dependences of the properties (thermodynamic functions, the width of the forbidden band, the heat capacity) of solid solutions of the GaSe–GaS system are established. Optical value was obtained on single-crystal samples. Solid solutions of the \({\text{GaSe}}_{{ 1- {\text{x}}}} {\text{S}}_{\text{x}}\) compounds were grown by the Bridgman method by directional crystallization.




Similar content being viewed by others
References
Madelung O. Semiconductors data handbook. Berlin: Springer; 2004.
Terhell ICI. Polytypism in the III–VI layer compounds. Prog Cryst Growth Charact Mater. 1983;7:55–110.
Mustafaeva SN, Asadov MM. Currents of isothermal relaxation in GaS <Yb> single crystals. Solid State Commun. 1983;45:491–4.
Mustafaeva SN, Asadov MM. Field kinetics of photocurrent in GaSe amorphous films. Mater Chem Phys. 1986;15:185–9.
Mamedov KK, Kerimov IG, Mekhtiev MI, Masimov EA, Izv Akad Nauk SSSR. Neorg Mater. 1972; 8:2096–98; Bull Acad Sci USSR Inorg Mater (English Transl). 1972; 8:1843–45.
Mamedov KK, Kerimov IG, Kostryukov VN, Mekhtiev MI. Heat capacity of gallium and thallium selenides. Fiz Tekh Poluprovodn (S Peterburg). 1967;1:441–2.
Tyurin AV, Gavrichev KS, Khoroshilov AV, Zlomanov VP. Heat capacity and thermodynamic functions of GaSe from 300 to 700 K. Inorg Mater. 2014;50:233–6.
Rustamov PG. Khal’kogenidy galliya. Baku: Akademiya nauk AzSSR; 1967. s. 130; Rustamov PG. Gallium chalcogenides. Baku: Academy of Sciences of the AzSSR; 1967. p. 130 (in Russian).
Asadov MM, Mustafaeva SN, Mamedov AN, Aljanov MA, Kerimova EM, Nadjafzade MD. Dielectric properties and heat capacity of (TlInSe2)1–x(TlGaTe2)x solid solutions. Inorg Mater. 2015;51:772–8.
Mustafaeva SN, Asadov MM, Kerimova EM, Gasanov NZ. Dielectric and optical properties of TlGa1–xErxS2 (x = 0, 0.001, 0.005, 0.01) single crystals. Inorg Mater. 2013;49:1175–9.
Mustafaeva SN, Asadov MM, Kyazimov SB, Gasanov NZ. T–x phase diagram of the TlGaS2–TlFeS2 system and band gap of TlGa1–xFexS2 (0 = x = 0.01) single crystals. Inorg Mater. 2012;48:984–6.
Asadov MM, Ahmedly KM. Procedure for calculating phase equilibrium in simple binary systems of ideal liquid and solid solution. Inorg Mater. 1996;32:133–4.
Asadov MM, Ahmedly KM. Calculation of some thermodynamic parameters for non-ideal solutions. Solid State Phenom. 2008;138:331–8.
Asadov SM, Mamedov AN, Kulieva SA. Composition- and temperature-dependent thermodynamic properties of the Cd, Ge||Se, Te system, containing solid solutions. Inorg Mater. 2016;52:876–85.
Zheng F, Shen JY, Liu YQ, Kim WK, Chu MY, Ider M, Bao XH, Anderson TJ. Thermodynamic optimization of the Ga–Se system. CALPHAD. 2008;32:432–8.
Abbasov AS. Termodinamicheskiye svoystva nekotorykh poluprovodnikovykh veshchestv (Thermodynamic properties of some semiconductor substances). Baku. Elm; 1981. (in Russian).
Vasilev VP. Correlations between the thermodynamic properties of II–VI and III–VI phases. Inorg Mater. 2007;43:115–24.
Ider M, Pankajavalli R, Zhuang W, Shen JY, Andersone TJ. Thermochemistry of the Ga–Se system. ECS J Solid State Sci Technol. 2015;4:51–60.
Mills KC. Thermodynamic data for inorganic sulphides, selenides and tellurides. London: Butterworth; 1974.
Voevodina OV, Morozov AN, Sarkisov SY, Bereznaya SA, Korotchenko ZV, Dikov DE. Properties of gallium selenide doped with sulfur from melt and from gas phase. KORUS 2005. Sci Technol. In: Proceedings of the 9th Russian-Korean international symposium, 2005; p. 551–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asadov, S.M., Mustafaeva, S.N. & Mammadov, A.N. Thermodynamic assessment of phase diagram and concentration–temperature dependences of properties of solid solutions of the GaS–GaSe system. J Therm Anal Calorim 133, 1135–1141 (2018). https://doi.org/10.1007/s10973-018-6967-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-6967-7