Abstract
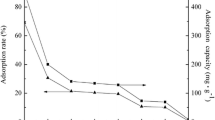
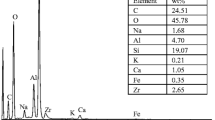
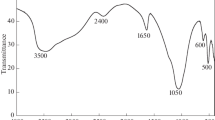
The ability of zirconium diphosphate (ZrP2O7) functionalized with oxalic acid to retain Europium (III) in solution has been studied. Zirconium diphosphate was synthetized and characterized by X-ray diffraction (XRD), Scanning Electron Microscopy (SEM-EDAX), Infrared Spectrometry (IR) and Atomic Force Microscopy (AFM) techniques, in order to determine its composition and purity. In order to understand the interaction between zirconium diphosphate and Eu (III), surface characteristic such as zero charge point (pHPCN) and surface site density (DS) were determined. Results from batch experiments showed that sorption capacity of zirconium diphosphate vis-a-vis Eu (III) was enhanced in the presence of oxalate ligand.





Similar content being viewed by others
Data availability
The authors hereby certify that this manuscript is an original research article, has not been published before and is not currently being considered for publication elsewhere.
References
IAEA (2006) Storage of radioactive waste. safety Guide No. WS-G-6.1, pp 55, Vienna. https://www-pub.iaea.org/mtcd/publications/pdf/pub1254_web.pdf
Rodney CE (1999) Nuclear waste forms for actinides. Proc. Natl. Acad. Sci. 96: 3432–3439
NEA (2005) NEA sorption project phase II. OECD Publishing, Paris
Lomenech C, Simoni E, Drot R, Ehrhardt JJ (2003) Sorption of uranium (VI) species on zircon: structural investigation of the solid/solution interface. J Colloid Interface Sci 261:221–232
Dacheux N, Clavier N, Robisson A-C, Terra O, Audubert F, Lartigue JÉ, Guy C (2004) Immobilisation of actinides in phosphate matrices. C R Chim 7(12):1141–1152
Ordoñez-Regil E, Drot R, Simoni E, Ehrhardt JJ (2002) Sorption of Uranium (VI) onto Lanthanum phosphate surfaces. Langmuir 18:7977–7984
Drot R, Simoni E (1999) Uranium(VI) and Europium(III) speciation at the phosphate compounds–solution interface. Langmuir 15(14):4820–4827
Finck N, Drot R, Lagarde G, Mercier-Bion F, Catalette H, Simoni E (2008) Temperature effects on the interaction mechanisms between U (VI) and Eu (III) and ZrP2O7: experiment and modeling. Radiochim Acta 96:11–21
García-González N, Ordoñez-Regil E, Almazán-Torres MG, Solis D, Simoni E (2012) Speciation of U (VI) sorbed onto ZrP2O7 in the presence of citric and oxalic acid. Radiochim Acta 100:305–309
Gschneidner KA, Eyring LR, Lander GH, Choppin GR (eds) (1994) Handbook on the physics and chemistry of rare earths, Lanthanides/Actinides: Chemistry, vol. 18. Elsevier
Drot R, Lindecker C, Fourest B, Simoni E (1998) Surface characterization of zirconium and thorium phosphate compounds. New J Chem 22:1105–1109
Alberti G, Constantino U, Millini R, Perego G, Vivan R (1994) Preparation, characterization, and structure of α-zirconium hydrogen phosphate hemihydrate. J Solid State Chem 113:289–295
Garcia Gonzalez N, Ordoñez Regil E, Simoni E, Barrera Díaz CE (2009) Effect of organic acids on sorption of uranyl ions in solution by ZrP2O7. J Radioanal Nucl Chem 283:409–415
Alamo J, Roy R (1984) Revision of crystalline phases in the system ZrO2-P2O5. Commun Am Ceram Soc 67:80–82
Noh JS, Schwarz JA (1989) Estimation of the point of zero charge of simple oxides by mass titration. J Colloid Interface Sci 130(1):157–164
Lomenech C, Drot R, Simoni E (2003) Speciation of uranium(VI) at the solid/solution interface: sorption modeling on zirconium silicate and zirconium oxide. Radiochim Acta 91(8):453–462
Herbelin AL, Westall JC (1996) FITEQL: a computer program for determination of chemical equilibrium constants from experimental. Report 96 – 01. Department of Chemistry, Oregon State University, Corvallis
Rocchicchioli C (1964) La spectrosgraphie d’absorption infrarouge, moyen d’étude de l’eau dans les substances minérales. Chim Anal 46:452–456
García-González N, Ordoñez-Regil E, Almazán-Torres MG, Simoni E (2018) Interaction of salicylic acid with zirconium diphosphate and its reactivity toward uranium (VI). Radiochim Acta 106(4):291–300
Andrew D, Wiesner LE, Katz C-C ChenThe (2006) Impact of ionic strength and background electrolyte on pH measurements in metal ion adsorption experiments. J Colloid Interface Sci 301(1):329–332
Hu XJ, Liu YG, Zeng GM, You SH, Wang H, Hu X, Guo YM, Tan XF, Guo FY (2014) Effects of background electrolytes and ionic strength on enrichment of cd(II) ions with magnetic graphene oxide–supported sulfanilic acid. J Colloid Interface Sci 435:138–144
Shirzadeh M, Sepehr E, Rasouli Sadaghiani MH, Ahmadi F (2020) Effect of pH, initial concentration, background Electrolyte, and ionic strength on Cadmium Adsorption by TiO2 and γ-Al2O3 nanoparticles. Pollution 6(2):223–235
Funding
The research data that support the findings of this study are available from corresponding author (guadalupe.almazan@inin.gob.mx) and can be shared in a public repository if required by publisher.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have approved the manuscript and agreed its submission to Journal of Radionalytical Nuclear Chemistry. The authors also certify have no conflicts of interest to declare that are relevant to the content of this article, likewise they have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
González, N.G., Regil, E.O., Torres, MG.A. et al. Europium (III) sorption on zirconium diphosphate functionalized with oxalic acid. J Radioanal Nucl Chem 332, 4573–4579 (2023). https://doi.org/10.1007/s10967-023-09168-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09168-w