Abstract
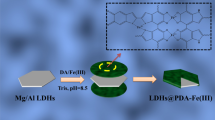
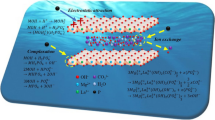
Mg-Al layered double hydroxides (LDH) (F-MgAl-LDHs and O-MgAl-LDHs) and Mg-Al layered double oxides (LDO) (F-MgAl-LDO and O-MgAl-LDO) nanosheets were prepared using a modified co-precipitation and oven/freeze dry route for adsorption of uranium. The freeze dry could evidently promote the adsorption ability. It’s resulted from larger specific surface (F-MgAl-LDO > F-MgAl-LDHs > O-MgAl-LDO > O-MgAl-LDHs) and pore size, as well as sufficient expose of vacant sites in the inner struture of F-MgAl-LDHs and F-MgAl-LDO. The pH, shaking time, initial uranium concentation and temperature influenced the adsorption capacity of F-MgAl-LDHs, O-MgAl-LDHs, F-MgAl-LDO and O-MgAl-LDO, while ionic strength exerted slightly little influence. Na2CO3 highlighted the best desorption effectivity, with desorption efficiency of 97.84% for F-MgAl-LDHs and 98.52% for F-MgAl-LDO, respectively. It is noteworthy that maximum adsorption capacity of F-MgAl-LDO reached 1099.93 mg/g, locating the top rank in the uranium-specific adsorbents. The adsorption conformed to the pseudo-second-order model, indicating chemical adsorption in nature. The thermodynamic was also investigated. The adsorption mechanism was determined that M-O and C-O bonds participated the complex process in the uranium adsorption. The study proposed the freeze dry as an efficient method to promote adsorbent performance.








Similar content being viewed by others
References
Vasudevamurthy G, Nelson AT (2022) Uranium carbide properties for advanced fuel modeling–A review. J Nucl Mater 558:153145
Şenol ZM, Keskin ZS, Özer A, Şimşek S (2022) Application of kaolinite-based composite as an adsorbent for removal of uranyl ions from aqueous solution: kinetics and equilibrium study. J Radioanal Nucl Chem 331:403–414
Sar SK, Diwan V, Biswas S, Singh S, Sahu M, Jindal MK, Arora A (2018) Study of uranium level in groundwater of Balod district of Chhattisgarh state, India and assessment of health risk. Hum Ecol Risk Assess 24:691–698
Şenol ZM (2021) A chitosan-based composite for adsorption of uranyl ions; mechanism, isothems, kinetics and thermodynamics. Int J Biol Macromol 183:1640–1648
Liao J, Liu P, Xie Y, Zhang Y (2021) Metal oxide aerogels: Preparation and application for the uranium removal from aqueous solution. Sci Total Environ 768:144212
Ma F, Gui Y, Liu P, Xue Y, Song W (2020) Functional fibrous materials-based adsorbents for uranium adsorption and environmental remediation. Chem Eng J 390:124597
Sen K, Mishra D, Debnath P, Mondal A, Mondal NK (2021) Adsorption of uranium (VI) from groundwater by silicon containing biochar supported iron oxide nanoparticle. Bioresour Technol Rep 14:100659
Yang L, Chen M, Lu Z, Huang Y, Wang J, Lu L, Cheng X (2020) Synthesis of CaFeAl layered double hydroxides 2D nanosheets and the adsorption behaviour of chloride in simulated marine concrete. Cem Concr Compos 114:103817
Wang H, Zhao W, Chen Y, Li Y (2020) Nickel aluminum layered double oxides modified magnetic biochar from waste corncob for efficient removal of acridine orange. Bioresour Technol 315:123834
Zubair M, Manzar MS, Mu’azu ND, Anil I, Blaisi NI, Al-Harthi MA (2020) Functionalized MgAl-layered hydroxide intercalated date-palm biochar for Enhanced Uptake of Cationic dye: Kinetics, isotherm and thermodynamic studies. Appl Clay Sci 190:105587
Jung KW, Lee SY, Choi JW, Hwang MJ, Shim WG (2021) Synthesis of Mg-Al layered double hydroxides-functionalized hydrochar composite via an in situ one-pot hydrothermal method for arsenate and phosphate removal: structural characterization and adsorption performance. Chem Eng J 420:129775
Shi X, Kang L, Hong J, Wang C, Wei R, Naik N, Guo Z (2021) Strong selectivity and high capacity in the adsorption of As (V) from wastewater by glycine-modified Fe/Cu-layered double hydroxides. J Alloys Compd 865:158956
Claros M, Kuta J, El-Dahshan O, Michalička J, Jimenez YP, Vallejos S (2021) Hydrothermally synthesized MnO2 nanowires and their application in Lead (II) and Copper (II) batch adsorption. J Mol Liq 325:115203
Sachan D, Das G (2022) Fabrication of Biochar-Impregnated MnO2 Nanocomposite: Characterization and Potential Application in Copper (II) and Zinc (II) Adsorption. J Hazard Toxic Radioact Waste 26:04021049
Ying D, Hong P, Jiali F, Qinqin T, Yuhui L, Youqun W, Zhibin Z, Xiaohong C, Yunhai L (2020) Removal of uranium using MnO2/orange peel biochar composite prepared by activation and in-situ deposit in a single step. Biomass Bioenergy 142:105772
Hong Y, Peng J, Zhao X, Yan Y, Lai B, Yao G (2019) Efficient degradation of atrazine by CoMgAl layered double oxides catalyzed peroxymonosulfate: optimization, degradation pathways and mechanism. Chem Eng J 370:354–363
Hong Y, Zhou H, Xiong Z, Liu Y, Yao G, Lai B (2020) Heterogeneous activation of peroxymonosulfate by CoMgFe-LDO for degradation of carbamazepine: Efficiency, mechanism and degradation pathways. Chem Eng J 391:123604
Deng H, Li A, Ye C, Sheng L, Li Z, Jiang Y (2020) Green Removal of Various Pollutants by Microsphere Adsorption: Material Characterization and Adsorption Behavior. Energy Fuels 34:16330–16340
Hou T, Yan L, Li J, Yang Y, Shan L, Meng X, Li X, Zhao Y (2020) Adsorption performance and mechanistic study of heavy metals by facile synthesized magnetic layered double oxide/carbon composite from spent adsorbent. Chem Eng J 384:123331
Wu H, Liu X, Wen J, Liu Y, Zheng X (2021) Rare-earth oxides modified Mg-Al layered double oxides for the enhanced adsorption-photocatalytic activity. Colloids Surf A 610:125933
Xu S-d, Dong L, Guo X-y, Wen Y, Jie G (2019) Selenium (VI) removal from caustic solution by synthetic Ca-Al-Cl layered double hydroxides. Trans Nonferrous Met Soc China 29:1763–1775
Karami Z, Jouyandeh M, Ali JA, Ganjali MR, Aghazadeh M, Maadani M, Rahn M, Luzi F, Torre L, Puglia D, Saeb MR (2019) Development of Mg-Zn-Al-CO3 ternary LDH and its curability in epoxy/amine system. Prog Org Coat 136:105264
Noli F, Kapashi E, Kapnisti M (2019) Biosorption of uranium and cadmium using sorbents based on Aloe vera wastes. J Environ Chem Eng 7:102985
Aniagor CO, Elshkankery M, Fletcher A, Morsy OM, Abdel-Halim E, Hashem A (2021) Equilibrium and kinetic modelling of aqueous cadmium ion and activated carbon adsorption system. Water Conserv Sci Eng 6:95–104
Al-Ghouti MA, Da’ana DA (2020) Guidelines for the use and interpretation of adsorption isotherm models: A review. J Hazard Mater 393:122383
Xie J, Lv R, Peng H, Fan J, Tao Q, Dai Y, Zhang Z, Cao X, Liu Y (2020) Phosphate functionalized poly (vinyl alcohol)/poly (acrylic acid)(PVA/PAA): an electrospinning nanofiber for uranium separation. J Radioanal Nucl Chem 326:475–486
Xie J, Dai Y, Wang Y, Liu Y, Zhang Z, Wang Y, Tao Q, Liu Y (2021) Facile immobilization of NiFeAl-LDHs into electrospun poly (vinyl alcohol)/poly (acrylic acid) nanofibers for uranium adsorption. J Radioanal Nucl Chem 329:1103–1117
Duan J, Ji H, Xu T, Pan F, Liu X, Liu W, Zhao D (2021) Simultaneous adsorption of uranium (VI) and 2-chlorophenol by activated carbon fiber supported/modified titanate nanotubes (TNTs/ACF): Effectiveness and synergistic effects. Chem Eng J 406:126752
Chaudhary M, Singh L, Rekha P, Srivastava VC, Mohanty P (2019) Adsorption of uranium from aqueous solution as well as seawater conditions by nitrogen-enriched nanoporous polytriazine. Chem Eng J 378:122236
Yuvaraja G, Su M, Chen DY, Pang Y, Kong LJ, Subbaiah MV, Reddy GM (2020) Impregnation of magnetic-Momordica charantia leaf powder into chitosan for the removal of U (VI) from aqueous and polluted wastewater. Int J Biol Macromol 149:127–139
Zhao C, Liu J, Deng Y, Tian Y, Zhang G, Liao J, Sun Q (2019) Uranium (IV) adsorption from aqueous solutions by microorganism-graphene oxide composites via an immobilization approach. J Clean Prod 236:117624
El-Bohy MN, Abdel-Monem YK, Rabie KA, Farag NM, Mahfouz MG, Galhoum AA, Guibal E (2017) Grafting of arginine and glutamic acid onto cellulose for enhanced uranyl sorption. Cellulose 24:1427–1443
Abdi S, Nasiri M, Mesbahi A, Khani MH (2017) Investigation of uranium (VI) adsorption by polypyrrole. J Hazard Mater 332:132–139
Shao D, Li Y, Wang X, Hu S, Wen J, Xiong J, Marwani H (2017) M. Phosphate-functionalized polyethylene with high adsorption of uranium (VI). Acs Omega 2(7):3267–3275
Qin X, Yang W, Yang W, Ma Y, Li M, Chen C, Pan Q (2021) Covalent modification of ZIF-90 for uranium adsorption from seawater. Micropor Mesopor Mat 323:111231
Zhao S, Feng T, Feng L, Yan B, Sun W, Luo G, Wang N (2022) Rapid recovery of uranium with magnetic-single-molecular amidoxime adsorbent,Sep. Purif. Technol.120524
Li Y, Dai Y, Tao QQ, Gao Z, Xu L (2022) Ultrahigh efficient and selective adsorption of U(VI) with amino acids-modified magnetic chitosan biosorbents: Performance and mechanism. Int J Biol Macromol 214:54–66
Şenol ZM, Kaya S, Şimşek S, Katin KP, Özer A, Marzouki R (2022) Synthesis and characterization of chitosan-vermiculite-lignin ternary composite as an adsorbent for effective removal of uranyl ions from aqueous solution: Experimental and theoretical analyses. Int J Biol Macromol 209:1234–1247
Meng J, Lin X, Li H, Zhang Y, Zhou J, Chen Y, Shang R, Luo X (2019) Adsorption capacity of kelp-like electrospun nanofibers immobilized with bayberry tannin for uranium (VI) extraction from seawater. RSC Adv 9:8091–8103
Skwarek E, Gładysz-Płaska A, Choromańska J, Broda E (2019) Adsorption of uranium ions on nano-hydroxyapatite and modified by Ca and Ag ions. Adsorption 25:639–647
Wang X, Cai Y, Han T, Fang M, Chen K, Tan X (2020) Phosphate functionalized layered double hydroxides (phos-LDH) for ultrafast and efficient U (VI) uptake from polluted solutions. J Hazard Mater 399:123081
Lyu P, Wang G, Wang B, Yin Q, Li Y, Deng N (2021) Adsorption and interaction mechanism of uranium (VI) from aqueous solutions on phosphate-impregnation biochar cross-linked MgAl layered double-hydroxide composite. Appl Clay Sci 209:106146
Chen M, Li S, Li L, Jiang L, Ahmed Z, Dang Z, Wu P (2021) Memory effect induced the enhancement of uranium (VI) immobilization on low-cost MgAl-double oxide: Mechanism insight and resources recovery. J Hazard Mater 401:123447
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (22066001) and the Natural Science Foundation of Jiangxi Province of China (20212ACB213001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tao, q., Xie, J., Li, Y. et al. Effects of dry processing on adsorption of uranium on Mg-Al layered double hydroxides and calcined layered double oxides. J Radioanal Nucl Chem 331, 4587–4600 (2022). https://doi.org/10.1007/s10967-022-08529-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08529-1