Abstract
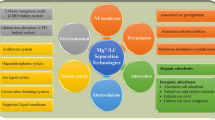
Thorium-230 has many research applications, but there is not a commercial source of this isotope. However, since 230Th is part of the 238U decay chain, it can be separated from naturally occurring uranium. In this work, a novel procedure was developed to separate thorium from uranium ore, consisting of leaching, liquid–liquid extraction, precipitations and ion exchange chromatography. The final product was 91.32 ± 0.77 mg of thorium with a purity of 99.5 ± 1.2 wt%. Of that, 7.65 ± 0.10 mg was 230Th and the remainder 232Th. The total yield of 230Th was 71.1 ± 5.4%. Ways to improve the yield by further processing the back-extraction solution are suggested.
















Similar content being viewed by others
References
Zhao J, Yu K, Feng Y (2009) High-precision 238U–234U–230Th disequilibrium dating of the recent past: a review. Quat Geochronol 4:423–433
Cheng H, Edwards L, Shen C, Polyak V, Asmerom Y, Woodhead J, Hellstrom J, Wang Y, Kong X, Spötl C, Wang X, Alexander E (2013) Improvements in 230Th dating, 230Th and 234U half-life values, and U–Th isotopic measurements by multi-collector inductively coupled plasma mass spectrometry. Earth Planet Sci Lett 371–372:82–91
Goldblum B, Stroberg S, Allmond J, Angell C, Bernstein L, Bleuel D, Gibelin J, Phair L, Scielzo N, Swanberg E, Wiedeking M, Norman E (2009) Indirect determination of the 230Th(n, f) and 231Th(n, f) cross sections for thorium-based nuclear energy systems. Phys Rev C 80:044610
Singh JT (2019) A new concept for searching for time-reversal symmetry violation using Pa-229 ions trapped in optical crystals. Hyperfine Interact 240:29
Elgqvist J, Frost S, Pouget J, Albertsson P (2013) The potential and hurdles of targeted alpha therapy—clinical trials and beyond. Front Oncol 3:324
Stracener D (2011) R&D on accelerator-based production of 229Th. 2011 Nuclear Physics (NP) Review, Oak Ridge
Robertson A, Ramogida CF, Schaffer P, Radchenko V (2018) Development of 225Ac radiopharmaceuticals: TRIUMF perspectives and experiences. Curr Radiopharm 11:156–172
Zhuikov B, Kalmykov S, Ermolaev V, Aliev R, Kokhanyuk V, Matushko L, Tananaev I, Myasoedov B (2011) Production of 225Ac and 223Ra by irradiation of Th with accelerated protons. Radiochemistry 53(1):73–80
Peppard D, Asanovich G, Atteberry R, DuTemple O, Gergel M, Gnaedinger A, Mason G, Meschke V, Nachtman E, Winsch I (1953) Studies of the solvent extraction behavior of the transition elements. II. Isolation of gram quantities of Th230 (ionium) from a pitchblende residue. J Am Chem Soc 75(18):4576–4578
Kluge E, Lieser K, Loc I, Quandt S (1997) Separation of 230Th (ionium) from uranium ores in sulfuric acid and in nitric acid solutions. Radiochim Acta 24:21–26
Nirdosh I (2002) Removal of 230Th by solvent extraction from high grade uranium ore liquid tailings to arrest 226Ra production in the tailings. Can J Chem Eng 80:525–529
Carswell D, Fletcher J, Clelland D (1957) The recovery of thorium-230 from uranium wastes. J Inorg Nucl Chem 5(2):147–152
Loveland W, Morrissey D, Seaborg G (2006) Modern nuclear chemistry. Wiley, Hoboken
National Research Council (2012) Uranium mining in virginia: scientific, technical, environmental, human health and safety, and regulatory aspects of uranium mining and processing in virginia. The National Academies Press, Washington, DC
National Nuclear Data Center “NNDC” (2013) Brookhaven National Laboratory. http://www.nndc.bnl.gov/. Accessed 20 June 2019
Sato T (1965) Extraction of uranium(VI) and thorium from nitric acid solutions by tri-n-butyl phosphate. J Appl Chem 15:489–495
Orabi A (2013) Determination of uranium after separation using solvent extraction from slightly nitric acid solution and spectrophotometric detection. J Radiat Res Appl Sci 6(2):1–10
Ishimori T, Nakamura E (1963) Data of inorganic solvent extraction Part I. Japan Atomic Energy Research Inst, Tokyo
Healy T, McKay H (1965) Complexes between tributyl phosphate and inorganic nitrates. Recueil 75:730–736
Peppard D, Mason G, Gergel M (1957) The mutual separation of thorium, protoactinium, and uranium by tributyl phosphate extraction from hydrochloric acid. J Inorg Nucl Chem 3:370–378
Steinnes E (1975) Precise and rapid determination of trace amounts of uranium in inorganic samples by neutron activation and TBP extraction. Anal Chim Acta 76:461–463
Altas Y, Tel H, Eral M (1999) Anion-exchange separation and determination of thorium and uranium in Eskisehir-Beylikahir ore in Turkey. J Radioanal Nucl Chem 241(3):637–641
Verma P, Mohapatra P, Bhattacharyya A, Yadav A, Jha S, Bhattacharyyab D (2018) Structural investigations on uranium(VI) and thorium(IV) complexation with TBP and DHOA: a spectroscopic study. New J Chem 42(7):5243–5255
Brown D, Whittaker B (1978) A new simple procedure for the recovery and purification of protactinium-231. J Less Common Metals 61:161–170
Hyde E (1960) The radiochemistry of thorium. Subcommittee on Radiochemistry National Academy of Sciences—National Research Council, Oak Ridge
Faris JP, Buchanan R (1964) Anion exchange characteristics of the elements in nitric acid medium. Anal Chem 36(6):1157–1158
Saliba-Silvaa A, Linardi M, Dos Santos Rocha L, Durazzoa M (2015) Thorium-234 electrodeposition using ionic and aqueous uranyl solutions. ECS Trans 66(21):11–20
Kirby H (1959) The radiochemistry of protactinium. Subcommittee on Radiochemistry National Academy of Sciences—National Research Council, Miamisburg
Gindler J (1962) The radiochemistry of uranium. Subcommittee on Radiochemistry National Academy of Sciences- National Research Council, Argonne
Acknowledgements
This study was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under Contract DE-AC52-07NA27344. This material is based upon work supported by the Department of Energy National Nuclear Security Administration through the Nuclear Science and Security Consortium under Award Number DE-NA0003180. The authors would like to thank Rachel Lindvall for making all mass spectrometry measurements and for providing the uranium ore, as well as Keenan J Thomas and the Nuclear Counting Facility at LLNL for their expertise with the counting systems.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kmak, K.N., Shaughnessy, D.A. & Vujic, J. Separation of thorium from uranium ore. J Radioanal Nucl Chem 323, 931–945 (2020). https://doi.org/10.1007/s10967-019-06980-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06980-1