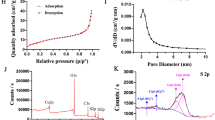
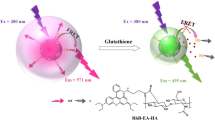
Abstract
The targeted delivery of anti-tumor drugs to diseased sites is significant in cancer therapy. Herein, a novel redox-responsive 5-fluorouracil (5-FU) prodrug delivery system based on mesoporous silica nanoparticles was proposed, in which polyethyleneimine (PEI, a bulky gatekeeper) and (3-aminopropyl) triethoxysilane-functionalized MCM41 were conjugated to an identical 3,3-diselanediyldipropionic acid linker via a condensation reaction between the amino group and carboxyl functional group. The chemotherapeutic reagent 5-FU was filled into the pores of Se-MCM41 nanocarriers as a model drug. The nanoparticles of PEI-Se-MCM41@5-FU exhibited a uniform and spherical morphology of approximately 175 nm. Under high concentrations of reactive oxygen species or glutathione (GSH) environments, such as the tumor microenvironment, 5-FU was rapidly released from the PEI-Se-MCM41 nanocarrier owing to its high sensitivity to both oxidizing and reducing agents. The release performance clearly verified the feasibility of PEI-Se-MCM41@5-FU nanoparticles for H2O2/GSH-triggered 5-FU release, particularly under GSH conditions. Additionally, the cytotoxic effects of PEI-Se-MCM41 nanoparticles findings indicating that there was no significant cytotoxicity against CT-26 colon cancer cells after 24 h incubation. However, PEI-Se-MCM41@5-FU nanoparticles exhibited a pronounced inhibitory effect on CT-26 cells at a concentration of 150 μg mL−1. Furthermore, the intracellular drug delivery performance of nanocarriers were evaluated using confocal laser scanning microscopy (CLSM), which disclosed that the PEI-Se-MCM41@RhB was successfully endocytosed into the CT-26 cells and released the loaded 5-FU. The conclusion was further proved by FACS analysis, which indicated that PEI-Se-MCM41@RhB was more easily uptaken into the CT-26 cells than free RhB. The findings of in vivo and in vitro studies suggest that PEI-Se-MCM41@5-FU holds great potential for effective tumor therapy.











Similar content being viewed by others
References
Z.Q. Wang, Y.Q. Guo, Y. Fan, J.W. Chen, H. Wang, M.W. Shen, X.Y. Shi, Adv. Mater. 34, 2107009 (2022). https://doi.org/10.1002/adma.202107009
B.Y. Chu, Y. Qu, X.L. He, Y. Hao, C.L. Yang, Y. Yang, D.R. Hu, F.F. Wang, Z.Y. Qian, Adv. Funct. Mater. 30, 2005918 (2020). https://doi.org/10.1002/adfm.202005918
Y. Fan, W.Z. Tu, M.W. Shen, X.M. Chen, Y.S. Ning, J.J. Li, T.F. Chen, H. Wang, F.F. Yin, Y. Liu, Adv. Funct. Mater. 30, 1909285 (2020). https://doi.org/10.1002/adfm.201909285
Y.Q. Guo, Y. Fan, G.M. Li, Z.Q. Wang, X.Y. Shi, M.W. Shen, A.C.S. Appl, Mater. Interfaces 13, 55815–55826 (2021). https://doi.org/10.1021/acsami.1c15282
B. Burtness, K. Harrington, R. Greil, D. Soulières, M. Tahara, G.D. De Castro Jr, A. Psyrri, N. Basté, P. Neupane, Å. Bratland, Lancet 394, 1915–1928 (2019). https://doi.org/10.1016/S0140-6736(19)32591-7
Y. Pommier, Nat. Rev. Cancer 6, 789–802 (2006). https://doi.org/10.1038/nrc1977
F. De Felice, D. Musio, V. Terenzi, V. Valentini, A. Cassoni, M. Tombolini, M. De Vincentiis, V. Tombolini, Radiat. Oncol. 9, 1–7 (2014). https://doi.org/10.1186/s13014-014-0263-x
R.Y.H. Tan, C.S. Lee, M.R. Pichika, S.F. Cheng, K. Y. Lam 14, 1672 (2022). https://doi.org/10.3390/polym14091672
R. Guillet-Nicolas, A. Popat, J.L. Bridot, G. Monteith, S.Z. Qiao, F. Kleitz, Angew. Chem. Int. Ed. 52, 2318–2322 (2013). https://doi.org/10.1002/anie.201208840
M. Vallet-Regí, F. Balas, D. Arcos, Angew. Chem. Int. Ed. 46, 7548–7558 (2007). https://doi.org/10.1002/anie.200604488
Q.L. Tang, Y. Xu, D. Wu, Y.H. Sun, Chem. Lett. 35, 474–475 (2006). https://doi.org/10.1246/cl.2006.474
Y.B. Jeon, S. Nagappan, X.H. Li, J.H. Lee, L.Y. Shi, S. Yuan, W.K. Lee, C.S. Ha, A.C.S. Appl, Mater. Interfaces 13, 6615–6630 (2021). https://doi.org/10.1021/acsami.0c20401
Z.C. Ma, W.T. Xu, Q.G. Wang, Q. Zhou, J. Zhou, Chem. Eng. J. 435, 135081 (2022). https://doi.org/10.1016/j.cej.2022.135081
Y.D. Wang, Z.L. Feng, X.F. Bai, Fuel 315, 123236 (2022). https://doi.org/10.1016/j.fuel.2022.123236
X. Li, Q.R.B. Xie, J.X. Zhang, W.L. Xia, H.C. Gu, Biomaterials 32, 9546–9556 (2011). https://doi.org/10.1016/j.biomaterials.2011.08.068
Q.J. He, J.L. Shi, Adv. Mater. 26, 391–411 (2014). https://doi.org/10.1002/adma.201303123
G. Singh, K. Ramadass, P. Sooriyakumar, O. Hettithanthri, M. Vithange, N. Bolan, E. Tavakkoli, L.V. Van Zwieten, A. Vinu, J Control Release 343, 187–206 (2022). https://doi.org/10.1016/j.jconrel.2022.01.036
H. Meng, M. Xue, T. Xia, Z.X. Ji, D.Y. Tarn, J.I. Zink, A.E. Nel, ACS Nano 5, 4131–4144 (2011). https://doi.org/10.1021/nn200809t
K.H. Bae, H.J. Chung, T.G. Park, Mol. Cells 31, 295–302 (2011). https://doi.org/10.1007/s10059-011-0051-5
X. Du, L. Xiong, S. Dai, S.Z. Qiao, Adv. Healthc. Mater. 4, 771–781 (2015). https://doi.org/10.1002/adhm.201400726
S. Park, M.H. Jung, Y.S. Lee, J.H. Bae, S.H. Kim, C.S. Ha, Mater. Des. 184, 108187 (2019). https://doi.org/10.1016/j.matdes.2019.108187
G. Lohiya, D.S. Katti, Carbohydr. Polym. 277, 118822 (2022). https://doi.org/10.1016/j.carbpol.2021.118822
W. Yin, W.D. Ke, W.J. Chen, L.C. Xi, Q.H. Zhou, J.F. Mukerabigwi, Z.S. Ge, Biomaterials 195, 63–74 (2019). https://doi.org/10.1016/j.biomaterials.2018.12.032
W.J. Zhang, Y.Z. Yan, S. Nagappan, S.S. He, C.S. Ha, Y.S. Jin, J. Macromol. Sci. A 58, 860–871 (2021). https://doi.org/10.1080/10601325.2021.1964368
C. Chen, Y.Y. Li, X.P. Yu, Q.L. Jiang, X.H. Xu, Q. Yang, Z.Y. Qian, Chin Chem Lett 29, 1609–1612 (2018). https://doi.org/10.1016/j.cclet.2018.02.010
S.J. Pan, T.Y. Li, Y.Z. Tan, H.P. Xu, Biomaterials 280, 121321 (2022). https://doi.org/10.1016/j.biomaterials.2021.121321
M. Karimi, A. Ghasemi, P.S. Zangabad, R. Rahighi, S.M. Basri, H. Mirshekari, M. Amiri, Z. Pishabad, A. Aslani, M. Bozorgomid, Chem. Soc. Rev. 45, 1457–1501 (2016). https://doi.org/10.1039/C5CS00798D
Q. Yin, J.N. Shen, Z.W. Zhang, H.J. Yu, Y.P. Li, Adv. Drug Deliv. Rev. 65, 1699–1715 (2013). https://doi.org/10.1016/j.addr.2013.04.011
L. Zhou, H. Wang, Y.P. Li, Theranostics 8, 1059 (2018). https://doi.org/10.7150/thno.22679
W. Lei, C.S. Sun, T.Y. Jiang, Y.K. Gao, Y. Yang, Q.F. Zhao, S.L. Wang, Mater. Sci. Eng. C 105, 110103 (2019). https://doi.org/10.1016/j.msec.2019.110103
Q.Q. Xu, Y.Q. Yang, J.Y. Lu, Y.Z. Lin, S.P. Feng, X.Y. Luo, Q.F. Zhao, Coord Chem Rev 469, 214687 (2022). https://doi.org/10.1016/j.ccr.2022.214687
K.L. Wang, J.Y. Lu, J.L. Li, Y.L. Gao, Y.L. Mao, Q.F. Zhao, S.L. Wang, J Control Release 339, 445–472 (2021). https://doi.org/10.1016/j.jconrel.2021.10.005
C.G. Qian, P.J. Feng, J.C. Yu, Y.L. Chen, Q.Y. Hu, W.J. Sun, X.Z. Xiao, X.L. Hu, A. Bellotti, Q.D. Shen, Angew. Chem. Int. Ed. 129, 2632–2637 (2017). https://doi.org/10.1002/ange.201611783
S.D. Zhai, X.L. Hu, Y.J. Hu, B.Y. Wu, D. Xing, Biomaterials 121, 41–54 (2017). https://doi.org/10.1016/j.biomaterials.2017.01.002
B.W. Chen, Y.J. Zhang, R.X. Ran, B. Wang, F.R. Qin, T. Zhang, G.Y. Wan, H.L. Chen, Y.S. Wang, Polym. Chem. 10, 4746–4757 (2019). https://doi.org/10.1039/C9PY00575G
P. Kuppusamy, H.Q. Li, G. Ilangovan, A.J. Cardounel, J.L. Zweier, K. Yamada, M. Krishna, J.B. Mitchell, Cancer Res. 62, 307–312 (2002)
T. Xia, M. Kovochich, M. Liong, H. Meng, S. Kabehie, S. George, J.I. Zink, A.E. Nel, ACS Nano 3, 3273–3286 (2009). https://doi.org/10.1021/nn900918w
H.W. Duan, S.M. Nie, J. Am. Chem. Soc. 129, 3333–3338 (2007). https://doi.org/10.1021/ja068158s
G.L. Zhang, J.L. Gao, J.C. Qian, L.L. Zhang, K. Zheng, K. Zhong, D.Q. Cai, X. Zhang, Z.Y. Wu, A.C.S. Appl, Mater. Interfaces 7, 14192–14200 (2015). https://doi.org/10.1021/acsami.5b04294
J. Liu, L. Zhao, L. Shi, Y. Yuan, D.A. Fu, Z.L. Ye, Q.L. Li, Y. Deng, X.X. Liu, Q.Y. Lv, Z. Wand, A.C.S. Appl, Mater. Interfaces 12, 54343–54355 (2020). https://doi.org/10.1021/acsami.0c13852
N.P. Gabrielson, D.W. Pack, Biomacromol 7, 2427–2435 (2006). https://doi.org/10.1021/bm060300u
Y.K. Buchman, E. Lellouche, S. Zigdon, M. Bechor, S. Michaeli, J.P. Lellouche, Bioconjug. Chem. 24, 2076–2087 (2013). https://doi.org/10.1021/bc4004316
C.H. Lee, L.W. Lo, C.Y. Mou, C.S. Yang, Adv. Funct. Mater. 18, 3283–3292 (2008). https://doi.org/10.1002/adfm.200800521
R. Nechikkattu, S.S. Park, C.S. Ha, Microporous Mesoporous Mater. 279, 117–127 (2019). https://doi.org/10.1016/j.micromeso.2018.12.022
D. Shao, M.Q. Li, Z. Wang, X. Zheng, Y.H. Lao, Z.M. Chang, F. Zhang, M.M. Lu, J. Yue, H.Z. Hu, Adv. Mater. 30, 1801198 (2018). https://doi.org/10.1002/adma.201801198
Q.S. Pan, T.T. Chen, C.P. Nie, J.T. Yi, C. Liu, Y.L. Hu, X. Chu, A.C.S. Appl, Mater. Interfaces 10, 33070–33077 (2018). https://doi.org/10.1021/acsami.8b13393
X.D. She, L.J. Chen, L. Velleman, C.P. Li, H.J. Zhu, C.Z. He, L.X. Kong, J. Colloid Interface Sci. 445, 151–160 (2015). https://doi.org/10.1016/j.jcis.2014.12.053
R. Starosta, A. Bykowska, A. Kyzioł, M. Płotek, M. Florek, J. Król, M. Jeżowska-Bojczuk, Chem Biol Drug Des 82, 579–586 (2013). https://doi.org/10.1111/cbdd.12187
J.S. Li, X.Y. Miao, Y.X. Hao, J.Y. Zhao, X.Y. Sun, L.J. Wang, J. Colloid Interface Sci. 318, 309–314 (2008). https://doi.org/10.1016/j.jcis.2007.09.093
J. Pokhrel, N. Bhoria, S. Anastasiou, T. Tsoufis, D. Gournis, G. Romanos, G.N. Karanikolos, Microporous Mesoporous Mater. 267, 53–67 (2018). https://doi.org/10.1016/j.micromeso.2018.03.012
J.H. Peng, F.M. Chen, Y.L. Liu, F. Zhang, L. Cao, Q.N. You, D. Yang, Z.M. Chang, M.F. Ge, L. Li, W.F. Dong, Theranostics 12, 1756 (2022). https://doi.org/10.7150/thno.68756
K. Zhang, E.H. Yuan, L.L. Xu, Q.S. Xue, C. Luo, B. Albela, L. Bonneviot, Eur. J. Inorg. Chem. 2012, 4183–4189 (2012). https://doi.org/10.1002/ejic.201200316
C. T. Kresge, M. E. Leonowicz, W. J. Roth, J. C. Vartuli, J. S. 1992 Beck, nature 359, 710–712. https://www.nature.com/articles/359710a0
S. Rohani, G. Mohammadi Ziarani, A. Badiei, A. Ziarati, M. Jafari, A. Shayesteh, Appl Organomet Chem (2018). https://doi.org/10.1002/aoc.4397
M. Abbasian, F. Mahmoodzadeh, J. Polym. Mater. 32, 527 (2015). https://www.researchgate.net/profile/Mojtaba-Abbasian/publication/293047060
A.D. Oliveira, C.A.G. Beatrice, F.R. Passador, L.A. Pessan, AIP Conf Proc 1914, 030020 (2017). https://doi.org/10.1063/1.5016707
D.T. Ma, Y.L. Li, J.B. Yang, H.W. Mi, S. Luo, L.B. Deng, C.Y. Yan, P.X. Zhang, Z.Q. Lin, X.Z. Ren, Nano Energy 43, 317–325 (2018). https://doi.org/10.1016/j.nanoen.2017.11.042
X.T. Cao, Y.H. Kim, J.M. Park, K.T. Lim, Eur. Polym. J. 78, 264–273 (2016). https://doi.org/10.1016/j.eurpolymj.2016.03.039
S.A. Salma, M.P. Patil, D.W. Kim, C.M.Q. Le, B.H. Ahn, G.D. Kim, K.T. Lim, Polym. Chem. 9, 4813–4823 (2018). https://doi.org/10.1039/C8PY00961A
Y.S. Birhan, B.Z. Hailemeskel, T.W. Mekonnen, E.Y. Hanurry, H.F. Darge, A.T. Andrgie, H.Y. Chou, J.Y. Lai, G.H. Hsiue, H.C. Tsai, Int. J. Pharm. 567, 118486 (2019). https://doi.org/10.1016/j.ijpharm.2019.118486
B. Urban-Klein, S. Werth, S. Abuharbeid, F. Czubayko, A. Aigner, Gene Ther. 12, 461–466 (2005). https://doi.org/10.1038/sj.gt.3302425
J.C. Yang, S.J. Pan, S.Q. Gao, T.Y. Li, H.P. Xu, Biomaterials 271, 120721 (2021). https://doi.org/10.1016/j.biomaterials.2021.120721
X.L. Zhang, X.Z. Su, Y.R. Zheng, S.J. Hu, L. Shi, F.Y. Gao, P.P. Yang, Z.Z. Niu, Z.Z. Wu, S. Qin, Angew. Chem. Int. Ed. 60, 26922–26931 (2021). https://doi.org/10.1002/anie.202111075
Y.F. Yuan, R.H. Zhou, T. Li, S. Qu, H. Bai, J.W. Liang, X.X. Cai, B. Guo, Cell Prolif. 54, e13008 (2021). https://doi.org/10.1111/cpr.13008
R. Pecora, J Nanopart Res 2, 123–131 (2000). https://doi.org/10.1023/A:1010067107182
M. Thommes, K. Kaneko, A.V. Neimark, J.P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol, K.S.W. Sing, Pure Appl. Chem. 87, 1051–1069 (2015). https://doi.org/10.1515/pac-2014-1117
A. Mohan, J. Peter, L. Rout, A.M. Thomas, S. Nagappan, S. Parambadath, W.J. Zhang, M. Selvaraj, C.S. Ha, Colloids Surf. A Physicochem. Eng. Asp. 611, 125846 (2021). https://doi.org/10.1016/j.colsurfa.2020.125846
S. Ravi, S.Q. Zhang, Y.R. Lee, K.K. Kang, J.M. Kim, J.W. Ahn, W.S. Ahn, J Ind Eng Chem 67, 210–218 (2018). https://doi.org/10.1016/j.jiec.2018.06.031
G.D. Park, J.S. Cho, J.K. Lee, Y.C. Kang, Sci. Rep. 6, 1–10 (2016). https://doi.org/10.1038/srep22432
W. Liu, Y.C. Zhu, F. Wang, X. Li, X.J. Liu, J.J. Pang, W.S. Pan, Royal Soc. Open Sci. 5, 181027 (2018). https://doi.org/10.1098/rsos.181027
P.R.S. Reddy, K.M. Rao, K.K. Rao, Y. Shchipunov, C.S. Ha, Macromol Res 22, 832–842 (2014). https://doi.org/10.1007/s13233-014-2117-7
R.W. Korsmeyer, N.A. Peppas, J Membr Sci 9, 211–227 (1981). https://doi.org/10.1016/S0376-7388(00)80265-3
M.R. Abukhadra, N.M. Refay, A.M. El-Sherbeeny, M.A. El-Meligy, ACS Omega 5, 11745–11755 (2020). https://doi.org/10.1021/acsomega.0c01078
J. Siepmann, N.A. Peppas, Int J Pharm 418, 6–12 (2011). https://doi.org/10.1016/j.ijpharm.2011.03.051
Acknowledgements
The work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Ministry of Science and ICT, Korea (2021R1I1A3060098, NRF-2021R1I1A3059777 and Brain Korea 21 Plus Program (4199990414196)) and by the Korea Institute for Advancement of Technology funded by the Ministry of Trade, Industry and Energy (P0017531).
Funding
National Research Foundation of Korea, 2021R1I1A3060098,NRF-2021R1I1A3059777 and Brain Korea 21 Plus Program (4199990414196), Korea Institute for Advancement of Technology, P0017531
Author information
Authors and Affiliations
Contributions
WJZ carried out main experimental works and wrote the manuscript text, AB performed cell cytotoxicity experiments. SSP did project administration, YZY and NJ helped measurements and discussion. IC and SKA supervised this work, IKP helped interpreted biological data. CSH reviewed and edited this manuscript as well as supervised and got fund for this work. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, WJ., Babu, A., Yan, YZ. et al. ROS/GSH dual-responsive selenium-containing mesoporous silica nanoparticles for drug delivery. J Porous Mater 30, 1469–1484 (2023). https://doi.org/10.1007/s10934-023-01430-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-023-01430-6