Abstract
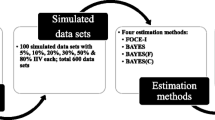
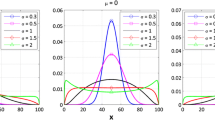
In a nonlinear mixed-effects modeling (NLMEM) approach of pharmacokinetic (PK) and pharmacodynamic (PD) data, two levels of random effects are generally modeled: between-subject variability (BSV) and residual unexplained variability (RUV). The goal of this simulation-estimation study was to investigate the extent to which PK and RUV model misspecification, errors in recording dosing and sampling times, and variability in drug content uniformity contribute to the estimated magnitude of RUV and PK parameter bias. A two-compartment model with first-order absorption and linear elimination was simulated as a true model. PK parameters were clearance 5.0 L/h; central volume of distribution 35 L; inter-compartmental clearance 50 L/h; peripheral volume of distribution 50 L. All parameters were assumed to have a 30% coefficient of variation (CV). One hundred in-silico subjects were administered a labeled dose of 120 mg under 4 sample collection designs. PK and RUV model misspecifications were associated with relatively larger increases in the magnitude of RUV compared to other sources for all levels of sampling design. The contribution of dose and dosing time misspecifications have negligible effects on RUV but result in higher bias in PK parameter estimates. Inaccurate sampling time data results in biased RUV and increases with the magnitude of perturbations. Combined perturbation scenarios in the studied sources will propagate the variability and accumulate in RUV magnitude and bias in PK parameter estimates. This work provides insight into the potential contributions of many factors that comprise RUV and bias in PK parameters.






Similar content being viewed by others
References
US Pharmacopeia (2011) <905> Uniformity of dosage units. https://www.usp.org/sites/default/files/usp/document/harmonization/gen-method/q0304_stage_6_monograph_25_feb_2011.pdf. Accessed Aug 2020
Wang J, Gao P, Zhang H et al (2020) Evaluation of concentration errors and inappropriate dose tailoring of tacrolimus caused by sampling-time deviations in pediatric patients with primary nephrotic syndrome. Ther Drug Monit 42:392–399. https://doi.org/10.1097/FTD.0000000000000717
Santalo O, Baig U, Poulakos M, Brown D (2016) Early vancomycin concentrations and the applications of a pharmacokinetic extrapolation method to recognize sub-therapeutic outcomes. Pharmacy 4:37. https://doi.org/10.3390/pharmacy4040037
Proost JH (2019) Calculation of the coefficient of variation of log-normally distributed parameter values. Clin Pharmokinet 58:1101–1102
Baron KT (2022) mrgsolve: simulate from ODE-based models
Lavielle M, Aarons L (2016) What do we mean by identifiability in mixed effects models? J Pharmacokinet Pharmacodyn 43:111–122. https://doi.org/10.1007/s10928-015-9459-4
Irby DJ, Ibrahim ME, Dauki AM et al (2021) Approaches to handling missing or “problematic” pharmacology data: pharmacokinetics. CPT Pharmacometr Syst Pharmacol 10:291–308. https://doi.org/10.1002/psp4.12611
Alihodzic D, Broeker A, Baehr M et al (2020) Impact of inaccurate documentation of sampling and infusion time in model-informed precision dosing. Front Pharmacol 11:172. https://doi.org/10.3389/fphar.2020.00172
Karlsson MO, Jonsson EN, Wiltse CG, Wade JR (1998) Assumption testing in population pharmacokinetic models: illustrated with an analysis of moxonidine data from congestive heart failure patients. J Pharmacokinet Biopharm 26:207–246
Choi L, Crainiceanu CM, Caffo BS (2013) Practical recommendations for population PK studies with sampling time errors. Eur J Clin Pharmacol 69:2055–2064. https://doi.org/10.1007/s00228-013-1576-7
Sun H, Ette EI, Ludden TM (1996) On the recording of sample times and parameter estimation from repeated measures pharmacokinetic data. J Pharmacokinet Biopharm 24:637–650
Silber HE, Kjellsson MC, Karlsson MO (2009) The impact of misspecification of residual error or correlation structure on the type I error rate for covariate inclusion. J Pharmacokinet Pharmacodyn 36:81–99
Al-Sallami HS, Wright DF, Duffull SB (2022) The propagation of between-subject variability from dose to response. Br J Clin Pharmacol 88:1414–1417
Ette EI, Kelman AW, Howie CA, Whiting B (1993) Interpretation of simulation studies for efficient estimation of population pharmacokinetic parameters. Ann Pharmacother 27(9):1034–1039
Al-Banna MK, Kelman AW, Whiting B (1990) Experimental design and efficient parameter estimation in population pharmacokinetics. J Pharmacokinet Biopharm 18:347–360
Acknowledgements
The authors acknowledge the Minnesota Supercomputing Institute (MSI) at the University of Minnesota for providing resources that contributed to the research results reported within this paper. URL: http://www.msi.umn.edu.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data analysis were performed by Mutaz Jaber and Richard Brundage. The first draft of the manuscript was written by Mutaz Jaber and Richard Brundage. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests. The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaber, M.M., Brundage, R.C. Investigating the contribution of residual unexplained variability components on bias and imprecision of parameter estimates in population pharmacokinetic mixed-effects modeling. J Pharmacokinet Pharmacodyn 50, 123–132 (2023). https://doi.org/10.1007/s10928-022-09837-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-022-09837-5