Abstract
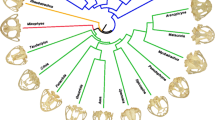
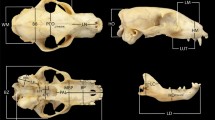
Although didelphid marsupials are considered to have a conservative body shape, they show a considerable amount of size variation. They also have different diets (from frugivore to animalivore), but none of the species are specialized. Didelphid marsupials also have a certain degree of specialization in vertical habitat use, from ground-dwellers to canopy-dwellers. Several authors have shown in other mammals that diet, activity patterns, locomotion, and habitat use influence skull shape in different groups of mammals, but also phylogenetic legacy plays a major role in skull evolution. Up till now, there are no published studies that investigate the form-function relationship in didelphid skulls; so the aim of this study is to analyze to what extent diet and vertical habitat use influence skull shape and override the influence of size and phylogeny. We used 2D geometric morphometry data from the skull and by analyzing the phylomorphospace, GLS, and PGLS we studied the effect of diet, vertical habitat use, allometry, and phylogenetic legacy on shape. Our results show that there are almost no shape differences between species of different diets and use of the vertical habitat, while allometry shows a strong correlation with shape, and also there is an evident effect of phylogenetic history. As didelphimorphians are not highly specialized, it is possible they developed a more generalized skull shape, flexible enough to adapt to different pressures. Moreover, as they have a highly integrated skull with few modules, it is expected that they respond to selection pressures by changing their size.





Similar content being viewed by others
References
Adams DC (2014a) A generalized K statistic for estimating phylogenetic signal from shape and other high-dimensional multivariate data. Syst Biol 63:685–697. https://doi.org/10.1093/sysbio/syu030
Adams DC (2014b) A method for assessing phylogenetic least squares models for shape and other high-dimensional multivariate data. Evolution 68:2675–2688. https://doi.org/10.1111/evo.12463
Adams DC, Collyer ML (2016) On the comparison of the strength of morphological integration across morphometric datasets. Evolution 70:2623–2631. https://doi.org/10.1111/evo.13045
Adams DC, Collyer ML, Kaliontzopoulou A (2018) Geomorph: software for geometric morphometric analyses. R package version 3.1.0.” https://cran.r-project.org/package=geomorph.
Alhajeri BH (2018) Craniomandibular variation in the taxonomically problematic gerbil genus Gerbillus (Gerbillinae, Rodentia): assessing the influence of climate, geography, phylogeny, and size. J Mammal Evol 25:261–276. https://doi.org/10.1007/s10914-016-9377-2
Amador LI, Giannini NP (2016) Phylogeny and evolution of body mass in didelphid marsupials (Marsupialia: Didelphimorphia: Didelphidae). Org Divers Evol 16:641–657. https://doi.org/10.1007/s13127-015-0259-x
Astúa D (2015a) Family Didelphidae (opossums). In: Wilson DE, Mittermeier RA (eds) Handbook of the Mammals of the World. Volume 5. Monotremes and Marsupials. Lynx Edicions, Barcelona, pp 70–186
Astúa D (2015b) Morphometrics of the largest New World marsupials, opossums of the genus Didelphis (Didelphimorphia, Didelphidae). Oecologia Aust 19:117–142. https://doi.org/10.4257/oeco.2015.1901.08
Astúa D (2009) Evolution of scapula size and shape in didelphid marsupials (Didelphimorphia: Didelphidae). Evolution 63:2438–2456. https://doi.org/10.1111/j.1558-5646.2009.00720.x
Astúa D (2010) Cranial sexual dimorphism in New World marsupials and a test of Rensch’s rule in Didelphidae. J Mammal 91:1011–1024. https://doi.org/10.1644/09-MAMM-A-018.1
Astúa D, Hinghst-Zaher E, Marcus LF, Cerqueira R (2000) A geometric morphometric analysis of cranial and mandibular shape variation of didelphid marsupials. Hystrix (n.s.) 10: 115–130
Birney EC, Monjeau JA, Phillips CJ, Sikes RS, Kim I (1996) Lestodelphis halli: new information on a poorly known argentine marsupial. Mastozool Neotropical 3: 171–181.
Blomberg SP, Garland T Jr, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x
Bookstein FL (1997) Landmark methods for forms without landmarks: morphometrics of group differences in outline shape. Med Image Anal 1:225–243
Cáceres NC, de Moraes Weber M, Melo GL, Meloro C, Sponchiado J, dos Santos Carvalho R, de Moura Bubadué J (2016) Which factors determine spatial segregation in the South American opossums (Didelphis aurita and D. albiventris)? An ecological niche modelling and geometric morphometrics approach. PLoS One 11:e0157723. https://doi.org/10.1371/journal.pone.0157723
Camargo NF, Machado LF, Mendonça AF, Vieira EM (2019) Cranial shape predicts arboreal activity of Sigmodontinae rodents. J Zool 1–11. https://doi.org/10.1111/jzo.12659
Cardini A (2017) Left, right or both? Estimating and improving accuracy of one-side-only geometric morphometric analyses of cranial variation. J Zool Syst Evol Res 55:1–10. https://doi.org/10.1111/jzs.12144
Chemisquy MA (2015) Peramorphic males and extreme sexual dimorphism in Monodelphis dimidiata (Didelphidae). Zoomorphology 134:587–599. https://doi.org/10.1007/s00435-015-0274-7
Chemisquy MA, Prevosti FJ, Martin G, Flores DA (2015) Evolution of molar shape in didelphid marsupials (Marsupialia: Didelphidae): analysis of the influence of ecological factors and phylogenetic legacy. Zool J Linn Soc 173:217–235. https://doi.org/10.1111/zoj.12205
Dumont M, Wall CE, Botton-Divet L, Goswami A, Peigné S, Fabre A-C (2016) Do functional demands associated with locomotor habitat, diet, and activity pattern drive skull shape evolution in musteloid carnivorans? Biol J Linn Soc 117:858–878. https://doi.org/10.1111/bij.12719
Eisenberg JF, Wilson DE (1981) Relative brain size and demographic strategies in didelphid marsupials. Am Nat 118: 1–15
Emerson SB, Radinsky L (1980) Functional analysis of sabertooth cranial morphology. Paleobiology 6: 295–312
Fox RC (1987) Paleontology and the early evolution of marsupials. In: Archer M (ed) Possums and Opossums: Studies in Evolution, Volume 1. Surrey Beatty & Sons, Chipping Norton, pp 161–169
Giarla TC, Jansa SA (2014) The role of physical geography and habitat type in shaping the biogeographical history of a recent radiation of Neotropical marsupials (Thylamys: Didelphidae). J Biogeogr 41:1547–1558. https://doi.org/10.1111/jbi.12320
Goin FJ, Woodburne MO, Zimicz AN, Martin G, Chornogbusky L (2016) A Brief History of South American Metatherians: Evolutionary Contexts and Intercontinental Dispersals. Springer, Dordrecht
Goodall C (1991) Procrustes methods in the statistical analysis of shape. J R Stat Soc Ser B 53:285–339
Grafen A (1989) The phylogenetic regression. Philos Trans R Soc B Biol Sci 326:119–157. https://doi.org/10.1098/rstb.1989.0106
Hansen TF (2003) Is modularity necessary for evolvability? Biosystems 69:83–94. https://doi.org/10.1016/s0303-2647(02)00132-6
Jansa SA, Barker FK, Voss RS (2013) The early diversification history of didelphid marsupials: a window into South America’s “splendid isolation.” Evolution 68:684–695. https://doi.org/10.1111/evo.12290
Law CJ, Duran E, Hung N, Richards E, Santillan I, Mehta RS (2018) Effects of diet on cranial morphology and biting ability in musteloid mammals. J Evol Biol 31:1918–1931. https://doi.org/10.1111/jeb.13385
Lee AK, Cockburn A (1985) Evolutionary Ecology of Marsupials. Cambridge University Press, Cambridge
Lima Silva LG, Ferreira GC, Vieira Rossi R (2019) Species diversity of Marmosa subgenus Micoureus (Didelphimorphia, Didelphidae) and taxonomic evaluation of the white-bellied woolly mouse opossum, Marmosa constantiae. Zool J Linn Soc 187: 240–277. https://doi.org/10.1093/zoolinnean/zlz023
Magnus LZ, Cáceres N (2017) Phylogeny explains better than ecology or body size the variation of the first lower molar in didelphid marsupials. Mammalia 81:119–133. https://doi.org/10.1515/mammalia-2015-0070
Magnus LZ, Machado RF, Cáceres N (2017) Comparative ecogeographical variation in skull size and shape of two species of woolly opossums (genus Caluromys). Zool Anz 267:139–150. https://doi.org/10.1016/j.jcz.2017.03.003
Marroig G, Shirai LT, Porto A, de Oliveira FB, De Conto V (2009) The evolution of modularity in the mammalian skull II: Evolutionary consequences. Evol Biol 36:136–148. https://doi.org/10.1007/s11692-009-9051-1
Martin GM, Udrizar Sauthier DE (2011) Observations on the captive behavior of the rare Patagonian opossum Lestodelphys halli (Thomas, 1921) (Marsupialia, Didelphimorphia, Didelphidae). Mammalia 75: 281–286. https://doi.org/10.1515/MAMM.2011.032
Martins EP, Hansen TF (1997) Phylogenies and the comparative method: a general approach to incorporating phylogenetic information into the analysis of interspecific data. Am Nat 149:646–667
Meloro C, Clauss M, Raia P (2015) Ecomorphology of Carnivora challenges convergent evolution. Org Divers Evol 15:711–720. https://doi.org/10.1007/s13127-015-0227-5
Pilatti P, Astúa D (2017) Orbit orientation in didelphid marsupials (Didelphimorphia: Didelphidae). Current Zool 63: 403–415. https://doi.org/10.1093/cz/zow068
Porto A, de Oliveira FB, Shirai LT, De Conto V, Marroig G (2009) The evolution of modularity in the mammalian skull I: Morphological integration patterns and magnitudes. Evol Biol 36:118–135. https://doi.org/10.1007/s11692-008-9038-3
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, URL http://www.R-project.org/
Radinsky LB (1981a) Evolution of skull shape in carnivores 1. Representative modern carnivores. Biol J Linn Soc 15: 369–388
Radinsky LB (1981b) Evolution of skull shape in carnivores 2. Additional modern carnivores. Biol J Linn Soc 16: 337–355
Radinsky LB (1982) Evolution of skull shape in carnivores 3. The origin and early radiation of the modern carnivore families. Paleobiology 8: 177–195
Reig OA, Kirsch JAW, Marshall LG (1987) Systematic relationships of the living and neocenozoic American “opossum–like” marsupials (suborder Didelphimorphia), with comments on the classification of these and the cretaceous and Paleogene New World and European metatherians. In: Archer M (ed) Possums and Opossums: Studies in Evolution. Surrey Beatty & Sons, Chipping Norton, pp 1–89
Rohlf FJ (2015) The tps series of software. Hystrix 26: 9–12. https://doi.org/10.4404/hystrix-26.1-11264
Rohlf FJ (1999) Shape statistics: Procrustes superimpositions and tangent spaces. J Classif 16:197–223. https://doi.org/10.1007/s003579900054
Sebastião H, Marroig G (2013) Size and shape in cranial evolution of 2 marsupial genera: Didelphis and Philander (Didelphimorphia, Didelphidae). J Mammal 94:1424–1437. https://doi.org/10.1644/11-MAMM-A-349.1
Shirai LT, Marroig G (2010) Skull modularity in Neotropical marsupials and monkeys: size variation and evolutionary constraint and flexibility. J Exp Zool Part B Mol Dev Evol 314 B:663–683. https://doi.org/10.1002/jez.b.21367
Teta P, Abba A, Cassini GH, Flores DA, Galliari CA, Lucero SO, Ramírez M (2018) Lista revisada de los mamíferos de Argentina. Mastozool Neotropical 25: 163–198. https://doi.org/10.31687/saremMN.18.25.1.0.15
Vieira EM, Astúa de Moraes D (2003) Carnivory and insectivory in Neotropical marsupials. In: Jones ME, Dickman CR, Archer M (eds) Predators with Pouches. CSIRO Publishing, Collingwood, pp 271–284
Vieira EM, Camargo NF (2012) Uso do espaço vertical por marsupiais brasileiros. In: Cáceres NC (ed) Os marsupiais do Brasil: Biologia, ecologia e conservação, 2o Edition. Editora UFMS, Campo Grande, pp 345–362
Voss RS, Días-Nieto JF, Jansa SA (2018) A revision of Philander (Marsupialia: Didelphidae), part 1: P. quica, P. canus, and a new species from Amazonia. Am Mus Novitates 3891:1–70
Voss RS, Gutiérrez EE, Solari S, Rossi RV, Jansa SA (2014) Phylogenetic relationships of mouse opossums (Didelphidae, Marmosa) with a revised subgeneric classification and notes on sympatric diversity. Am Mus Novitates 3817:1–27
Voss RS, Jansa SA (2009) Phylogenetic relationships and classification of didelphid marsupials, an extant radiation of New World metatherian mammals. Bull Am Mus Nat Hist 322:1–177. https://doi.org/10.1206/322.1
Acknowledgments
To G. Cassini, N. Toledo, and S. Vizcaíno for inviting us to contribute to this Special Issue - Tribute to Radinsky within the framework of the Symposium: “El paradigma de correlación formafunción en mastozoología: un tributo a Leonard Radinsky (1937–1985),” which took place during the XXXI Jornadas Argentinas de Mastozoología, in La Rioja, Argentina. 25 October 2018. To the curators of the following collections that allowed us to study the materials: P. Teta, G. Cassini, and S. Lucero (MACN, Argentina); E. González (MNHN, Uruguay); R. Maneyro (ZVC, Uruguay); M. de Vivo and J. Gualda (MZUSP, Brazil); S. Bogan (CFA); I. Olivares (MLP-Ma); D. Romero (MMP-Ma). To two anonymous reviewers who helped improve this manuscript. This is a contribution to PICT 2015-966 and PICT 2016-3151 by the Agencia Nacional de Promoción Científica y Tecnológica.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chemisquy, M.A., Tarquini, S.D., Romano Muñoz, C.O. et al. Form, Function and Evolution of the Skull of Didelphid Marsupials (Didelphimorphia: Didelphidae). J Mammal Evol 28, 23–33 (2021). https://doi.org/10.1007/s10914-019-09495-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-019-09495-4