Abstract
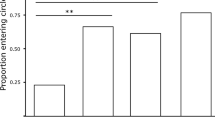
Female response to male sexual signals is context dependent and influenced by a variety of environmental factors. For instance, females often adjust mating preferences in response to predation risk. Social experiences as a juvenile, such as exposure to conspecific sexual signals, have also been shown to influence female mating preferences as adults. Experiments that examine the influence of both environmental factors and early social experiences on female preferences are needed to understand female mating behaviors in the wild. In Pacific field crickets (Teleogryllus oceanicus), males produce a calling song that females evaluate during mate choice. We examined the influence of both risk perception and juvenile exposure to calling song in shaping adult T. oceanicus female phonotaxis to either a high- or low-quality calling song. We predicted that females raised in song-less conditions would be more responsive to calling song to maximize mating opportunities, and that females would show reduced response to calling in a risky, open area. Females raised in a song-deprived environment were much more responsive to calling song compared to those reared with song. In contrast to our predictions, females raised in song-less conditions were more responsive to the high-quality song in the risky, open arena. These results suggest that juvenile acoustic rearing influences female responsiveness to male calling song, but it has less influence on strength of preference for signal quality.




Similar content being viewed by others
References
Adamo S, Robert D, Hoy R (1995) Effects of a tachinid parasitoid, Ormia ochracea, on the behavior and reproduction of its male and female field cricket hosts (Gryllus spp). J Insect Physiol 41:269–277
Alem S, Greenfield MD (2010) Economics of mate choice at leks: Do female waxmoths pay costs for indirect genetic benefits? Behav Ecol 21:615–625
Andersson MB (1994) Sexual selection. Princeton University Press
Atwell A, Wagner WE Jr (2015) Along came a spider who sat down beside her: Perceived predation risk, but not female age, affects female mate choosiness. Behav Process 115:143–148
Backwell PR, Christy J, Passmore N (1999) Female choice in the synchronously waving fiddler crab, Uca annulipes. Ethology 105:415–421
Bailey W, Haythornthwaite S (1998) Risks of calling by the field cricket, Teleogryllus oceanicus; potential predation by Australian long-eared bats. J Zool 244:505–513
Bailey NW, Zuk M (2008) Acoustic experience shapes female mate choice in field crickets. Proc R Soc B 275:2645–2650
Bailey NW (2008) Love will tear you apart: different components of female choice exert contrasting selection pressures on male field crickets. Behav Ecol 19:960–966
Bailey NW, Gray B, Zuk M (2010) Acoustic experience shapes alternative mating tactics and reproductive investment in male field crickets. Curr Biol 20:845–849
Bakker TC, Milinski M (1991) Sequential female choice and the previous male effect in sticklebacks. Behav Ecol Sociobiol 29:205–210
Balenger SL, Zuk M (2015) Roaming Romeos: male crickets evolving in silence show increased locomotor behaviors. Anim Behav 101:213–219
Ball E, Oldfield B, Rudolph KM (1989) Auditory organ structure, development, and function. In: Huber F, Moore T, Loher W (eds) Cricket behavior and neurobiology. Comstock Pub. Associates, New York, pp 391–422
Bateman PW, Gilson LN, Ferguson J (2001) Male size and sequential mate preference in the cricket. Gryllus bimaculatus Anim Behav 61:631–637
Cade W (1975) Acoustically orienting parasitoids: Fly phonotaxis to cricket song. Science 190:1312–1313
Csada RD, Neudorf DL (1995) Effects of predation risk on mate choice in female Acheta domesticus crickets. Ecol Entomol 20:393–395
Fowler-Finn KD, Rodríguez RL (2012) Experience‐mediated plasticity in mate preferences: mating assurance in a variable environment. Evolution 66:459–468
Grant JW, Noakes DL (1987) Escape behavior and use of cover by young-of-the-year brook trout, Salvelinus fontinalis. Can J Fish 44:1390–1396
Hebets EA (2003) Subadult experience influences adult mate choice in an arthropod: Exposed female wolf spiders prefer males of a familiar phenotype. Proc Natl Acad Sci USA 100:13390–13395
Hebets EA, Wesson J, Shamble PS (2008) Diet influences mate choice selectivity in adult female wolf spiders. Anim Behav 76:355–363
Hedrick AV, Dill LM (1993) Mate choice by female crickets is influenced by predation risk. Anim Behav 46:193–196
Hedrick A, Weber T (1998) Variance in female responses to the fine structure of male song in the field cricket, Gryllus integer. Behav Ecol 9:582–591
Heinen-Kay JL, Strub DB, Zuk M (2018) Limited flexibility in female Pacific field cricket (Teleogryllus oceanicus) exploratory behaviors in response to perceived social environment. Ethology 124:650–656
Helfman G (1989) Threat-sensitive predator avoidance in damselfish-trumpetfish interactions. Behav Ecol Sociobiol 24:47–58
Jennions MD, Petrie M (1997) Variation in mate choice and mating preferences: a review of causes and consequences. Biol Rev 72:283–327
Kibedi J (2016) An investigation on the ecological significance of the terrestrial context in predator-prey interactions between echolocating bats and the Australian field cricket (Teleogryllus spp.). Thesis, The University of Queensland
Kirkpatrick M (1996) Good genes and direct selection in the evolution of mating preferences. Evolution 50:2125–2140
Kramer DL, Bonenfant M (1997) Direction of predator approach and the decision to flee to a refuge. Anim Behav 54:289–295
Labadessa R, Todisco S (2016) Patterns of ecology and distribution of the tree crickets, Oecanthus dulcisonans and O. pellucens (orthoptera: Gryllidae; Oecanthinae), in southern Italy. Zootaxa. https://doi.org/10.11646/zootaxa.4169.3.10
Lafaille M, Bimbard G, Greenfield MD (2010) Risk trading in mating behavior: forgoing anti-predator responses reduces the likelihood of missing terminal mating opportunities. Behav Ecol Sociobiol 64:1485–1494
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640
Matsumoto Y, Mizunami M (2000) Olfactory learning in the cricket, Gryllus bimaculatus. J Exp Biol 203:2581–2588
Milinski M, Bakker TC (1992) Costs influences sequential mate choice in sticklebacks, Gasterosteus aculeatus. Proc Roy Soc B 250:229–233
Pilakouta N, Alonzo SH (2013) Predator exposure leads to a short-term reversal in female mate preferences in the green swordtail, Xiphophorus helleri. Behav Ecol 25:306–312
Pomiankowski A (1987) The costs of choice in sexual selection. J Theor Biol 128:195–218
Real L (1990) Search theory and mate choice. I. models of single-sex discrimination. Am Nat 136:376–405
Simmons LW, Zuk M, Rotenberry JT (2001) Geographic variation in female preference functions and male songs of the field cricket, Teleogryllus oceanicus. Evolution 55:1386–1394
Swanger E, Zuk M (2015) Cricket responses to sexual signals are influenced more by adult than juvenile experiences. J Insect Behav 28:328–337
Vraec V, Kočárek P (2005) The observation of saga Hellenica kaltenbach, 1967 (Orthoptera) on Corfu Island. Entomofauna Carpathica 17:11–13
Ydenberg RC, Dill LM (1986) The economics of fleeing from predators. Adv Study Behav 16:229–249
Zuk M, Simmons LW, Cupp L (1993) Calling characteristics of parasitized and unparasitized populations of the field cricket, Teleogryllus oceanicus. Behav Ecol Sociobiol 33:339–343
Zuk M, Rotenberry J, Simmons L (2001) Geographical variation in calling song of the field cricket, Teleogryllus oceanicus: the importance of spatial scale. J Evol Biol 14:731–741
Zuk M, Rotenberry JT, Tinghitella RM (2006) Silent night: adaptive disappearance of a sexual signal in a parasitized population of field crickets. Biol Lett 2:521–524
Zuk M, Bailey NW, Gray B, Rotenberry JT (2018) Sexual signal loss: the link between behavior and rapid evolutionary dynamics in a field cricket. J Anim Ecol 87:623–633
Acknowledgements
We wish to thank Daina Strub, Kristine Grab, Mounica Kota, and Adam Hartman for their help in maintaining cricket colonies and aiding in the experiments. Dr. David Stephens and Dr. Emilie Snell-Rood provided insightful comments that improved the paper.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 98 kb)
Rights and permissions
About this article
Cite this article
Ghalichi, N.S., Heinen-Kay, J.L. & Zuk, M. Acoustic Experience Interacts with Perceived Risk of Predation in Shaping Female Response in Crickets. J Insect Behav 33, 38–47 (2020). https://doi.org/10.1007/s10905-020-09744-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-020-09744-y