Abstract
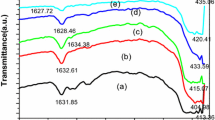
Agglomerates of \(\text{Co}_{3}\text{O}_{4}\) were obtained using the sol–gel method for the synthesis with subsequent calcination of the samples up to 550 °C. Through X-rays it was observed that the samples presented the pure spinel phase with a crystallite size between 33.73 and 41.45 nm. In the thermogravimetric measurement from 262 °C high structural stability is presented with phase change at 917 °C. As the temperature increases, the particles increase in size, observing agglomerated nanometer particles that increase with temperature (3.5–3.8 μm). The 683 cm\(^{-1}\) Raman mode for 550 °C clearly presents an shifted compared to the other samples. The band gap for the samples under study varied with the temperature change of 1.77–1.81 eV. The FWHM decreases at a higher temperature, this confirms the larger crystallite size and the higher degree of sintering.








Similar content being viewed by others
References
P. Yang, F. Wu, L. Wang, X. Chen, J. Xie, Nanostructuring \(\text{Co}_{3}\text{O}_{4}\) to tune capacitive behaviors: from low to high dimensions. Chem. Select. 5, 3638–3643 (2020)
L. Cardenas, A. Raba, R.M. Joya, Synthesis and evaluation of nickel doped \(\text{Co}_{3}\text{O}_{4}\) produced through hydrothermal technique. Dynamics 87, 184–191 (2020)
H. Sahan, H. Goktepe, S. Yildiz, C. Caymaz, S. Patat, A novel and green synthesis of mixed phase CoO@Co3O4@C anode material for lithium ion batteries. Ionics 25, 447–455 (2019)
R.M. Kakhki, M. Bina, Dispersive micro-solid phase extraction based on \(\text{Co}_{3}\text{O}_{4}\) modified nanoclinoptilolite for fast determination of malachite green in the environmental water samples. J. Inorg. Organomet. Polym. 30, 2475–2479 (2020)
M. Li, C. Han, Y. Zhang, X. Bo, L. Guo, Facile synthesis of ultrafine \(\text{Co}_{3}\text{O}_{4}\) nanocrystals embedded carbon matrices with specific skeletal structures as efficient non-enzymatic glucose sensors. Anal. Chim. Acta 861, 25–35 (2015)
J. Cao, S. Wang, X. Zhao, Y. Xing, J. Li, D. Li, Facile synthesis and enhanced toluene gas sensing performances of \(\text{Co}_{3}\text{O}_{4}\) hollow nanosheets. Mater. Lett. 263, 127215 (2020)
R. Kumar, Ni\(\text{Co}_{3}\text{O}_{4}\) nano-/microstructures as high-performance biosensors: a review. Nano-Micro Lett. 12–122, 1–52 (2020)
M.M. Abdullah, M.S. Akhtar, S.M. Al-Abbas, Facile growth and promising applications of cobalt oxide (\(\text{Co}_{3}\text{O}_{4}\)) nanoparticles as chemi-sensor and dielectric material. Curr. Nanosci. 14, 343–351 (2018)
K. Choi, H. Kim, K.M. Kim, D.W. Liu, G.Z. Cao, J.H. Le, \(\text{C}_{2}\text{H}_{5}\text{OH }\) sensing characteristics of various \(\text{Co}_{3}\text{O}_{4}\) nanostructures prepared by solve thermal reaction. Sensors Actuators B 146, 183–189 (2010)
B. Zhang, X. Zhou, C. Jiang, F. Qu, M. Yang, Facile synthesis of mesoporous \(\text{Co}_{3}\text{O}_{4}\) nanofans as gas sensing materials for selective detection of xylene vapor. Mater. Lett. 128, 127–130 (2018)
C. Yan, Y. Zhu, Y. Li, Z. Fang, L. Peng, X. Zhou, G. Yu, Built-in electric field enabled in carbon-doped \(\text{Co}_{3}\text{O}_{4}\) nanocrystals for superior lithium-ion storage. Adv. Funct. Mater. 28, 1705951 (2018)
S.L. Zhang, B.Y. Guan, X.F. Lu, S. Xi, Y. Du, X.W.D. Lou, Metal atom-doped \(\text{Co}_{3}\text{O}_{4}\) hierarchical nanoplates for electrocatalytic oxygen evolution***. Adv. Mater. 1000, 10000 (2020). https://doi.org/10.1002/adma.20200223
X. Xie, C. Ni, Z. Lin, D. Wu, X. Sun, Y. Zhang, W. Du, Phase and morphology evolution of high dielectric CoO/\(\text{Co}_{3}\text{O}_{4}\) particles with \(\text{Co}_{3}\text{O}_{4}\) nanoneedles on surface for excellent microwave absorption application. Chem. Eng. J. 396, 125205 (2020)
T. He, D. Chen, X. Jiao, Controlled synthesis of \(\text{Co}_{3}\text{O}_{4}\) nanoparticles through oriented aggregation. Chem. Mater. 16, 737–743 (2004)
B. Raghavendra, T. Sankarappa, A. Malge, Structural, optical absorption and conductivity of PIn/\(\text{Co}_{3}\text{O}_{4}\) composites******. J. Inorg. Organomet. Polym. 10000, 10000 (2020). https://doi.org/10.1007/s10904-020-01589-4
A. Lakehal, B. Bedhiaf, A. Bouaza, H. Benhebal, A. Ammari, C. Dalache, Structural, optical and electrical properties of Ni-doped \(\text{Co}_{3}\text{O}_{4}\) prepared via sol–gel technique. Mater. Res. 21, 1–8 (2018)
L. Qiao, H.Y. Xiao, H.M. Meyer, J.N. Sun, C.M. Rouleau, A.A. Puretzky, M.D. Biegalski, Nature of the band gap and origin of the electro-/photo-activity of \(\text{Co}_{3}\text{O}_{4}\). J. Mater. Chem. C. 1, 4628 (2013)
W. Shi, F. Guo, C. Zhu, H. Wang, H. Li, H. Huang, Y. Liu, Z. Kang, Carbon dots anchored on octahedral CoO as a stable visible-light-responsive composite photocatalyst for overall water splitting. J. Mater. Chem. A 5, 19800 (2017)
L. Chao, C. Taiqiang, X. Weijing, L. Xiaobing, P. Likun, C. Qun, H. Bingwen, Mesoporous nanostructured \(\text{Co}_{3}\text{O}_{4}\) derived from MOF template: a high-performance anode material for lithium-ion batteries. J. Mater. Chem. A 3, 5585–5591 (2015)
S. Jinxiao, Z. Hu, Z. Meizhou, F. Jianhui, Y. Aihua, Facile synthesis of metal-organic framework-derived \(\text{Co}_{3}\text{O}_{4}\) with different morphologies coated graphene foam as integrated anodes for lithium-ion. J. Alloys Compd. 768, 1049–1057 (2018)
B. Liu, X. Zhang, H. Shioyama, T. Mukai, T. Sakai, Q. Xu, Converting cobalt oxide subunits in cobalt metal-organic framework into agglomerated \(\text{Co}_{3}\text{O}_{4}\) nanoparticles as an electrode material for lithium ion battery. J. Power Sources 195, 857861 (2010)
R. Gao, Z. Yang, L. Zheng, L. Gu, L. Liu, Y. Lee, Z. Hu, X. Liu, Enhancing the catalytic activity of \(\text{Co}_{3}\text{O}_{4}\) for LiO\(_2\) batteries through the synergy of surface/interface/doping engineering. ACS Catal. 8, 1955 (2018)
Y. Li, W. Qiu, F. Qin, H. Fang, V.G. Hadjiev, D. Litvinov, J. Bao, Identification of cobalt oxides with Raman scattering and Fourier transform infrared spectroscopy. J. Phys. Chem. C. 120, 4511–4516 (2016)
A. Raba, J. BarbaOrtega, M. Joya, Effects of metal doping agent on the properties of \(\text{Nb}_{2-x}\)\(\text{M}_{x}\)\(\text{O}_{5}\) (M = Mn, Fe, and Ni) system. Int. J. Appl. Ceram. Technol. 15, 1577–1583 (2018)
J. Jiang, L. Li, Synthesis of sphere-like \(\text{Co}_{3}\text{O}_{4}\) nanocrystals via a simple polyol route. Mater. Lett. 61, 4894–4896 (2007)
R. Tummala, R.K. Guduru, P.S. Mohanty, Nanostructured \(\text{Co}_{3}\text{O}_{4}\) electrodes for supercapacitor applications from plasma spray technique. J. Power Sources 209, 44–51 (2012)
R. Tummala, R.K. Guduru, P.S. Mohanty, Binder free, porous and nanostructured \(\text{Co}_{3}\text{O}_{4}\) anode for Li-ion batteries from solution precursor plasma deposition. J. Power Sources 199, 270–277 (2012)
Y. Mussa, F. Ahmed, M. Arsalan, E. Alsharaeh, Two dimensional (2D) reduced graphene oxide (RGO)/hexagonal boron nitride (h-BN) based nanocomposites as anodes for high temperature rechargeable lithium-ion batteries. Sci. Rep. 10, 1882 (2020)
R. Itteboina, T.K. Sau, Sol–gel synthesis and characterizations of morphology-controlled \(\text{Co}_{3}\text{O}_{4}\) particles. Mater. Today. Proc. 9, 458467 (2019)
B. Tomić-Tucaković, D. Majstorovic, D. Jelic, S. Mentus, Thermogravimetric study of the kinetics of \(\text{Co}_{3}\text{O}_{4}\) reduction by hydrogen. Thermochim. Acta. 541, 15–24 (2012)
X. Deng, Z. Huang, W. Wang, R.N. Dav, Investigation of nanoparticle agglomerates properties using Monte Carlo simulations. Adv. Powder Technol. 27, 19711979 (2016)
A.M. Tonelli, J. Venturini, S. Arcaro, J.G. Henn, D.J. Moura, A.D.C. Viegas, C.P. Bergmann, Novel coreshell nanocomposites based on \(\text{TiO}_{2}\) covered magnetic \(\text{Co}_{3}\text{O}_{4}\) for biomedical applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 108, 1879–1887 (2020)
A. Takafumi, I. Toshio, M. Yoshitake, S. Woosuck, M. Ichiro, K. Masahito, Gas sensor properties of nanopore-bearing \(\text{Co}_{3}\text{O}_{4}\) particles containing Pt or Pd particles. J. Asian Ceram. Soc. 8(1), 138–148 (2020)
D. Trpkov, M. Panjan, L. Kopanja, M. Tadi, Hydrothermal synthesis, morphology, magnetic properties and self-assembly of hierarchical \(\alpha -\text{Fe}_{2}\text{O}_{3}\) (hematite) mushroom-, cube-and sphere-like superstructures. Appl. Surf. Sci. 457, 427–438 (2018)
I. Lorite, J.J. Romero, J.F. Fernndez, Effects of the agglomeration state on the Raman properties of \(\text{Co}_{3}\text{O}_{4}\) nanoparticles. J. Raman Spectrosc. 43, 14431448 (2012)
A. Diallo, A.C. Beye, T.B. Doyle, E. Park, M. Maaza, Green synthesis of \(\text{Co}_{3}\text{O}_{4}\) nanoparticles via Aspalathus linearis: physical properties. Green Chem. Lett. Rev. 8, 30–36 (2015)
G. George, S. Anandhan, A comparative study on the physicochemical properties of sol–gel electrospun cobalt oxide nanofibres from two different polymeric binders. RSC Adv. 5, 81429–81437 (2015)
V. Singh, M. Kosa, K. Majhi, D.T. Major, Putting DFT to the test: a first-principles study of electronic, magnetic, and optical properties of \(\text{Co}_{3}\text{O}_{4}\). J. Chem. Theory Comput. 11, 64–72 (2014)
R. Drasovean, S. Condurache-Bota, Structural characterization and optical properties of \(\text{Co}_{3}\text{O}_{4}\) and CoO films. J. Optoelectron. Adv. Mater. 11, 2141 (2009)
C.M. Jiang, L.R. Baker, J.M. Lucas, J. Vura-Weis, A.P. Alivisatos, S.R. Leone, Characterization of photo-induced charge transfer and hot carrier relaxation pathways in spinel cobalt oxide (\(\text{Co}_{3}\text{O}_{4}\)). J. Phys. Chem. C. 118, 22774–22784 (2014)
A.K. Sarfraz, S.K. Hasanain, Size dependence of magnetic and optical properties of \(\text{Co}_{3}\text{O}_{4}\) nanoparticles. Act. Phys. Pol. A. 125, 139–144 (2014)
J. Pal, P. Chauhan, Study of physical properties of cobalt oxide (\(\text{Co}_{3}\text{O}_{4}\)) nanocrystals. Mater. Charact. 61, 575–579 (2010)
H. Zhang, W. Tian, L. Zhou, H. Sun, M. Tade, S. Wang, Monodisperse \(\text{Co}_{3}\text{O}_{4}\) quantum dots on porous carbon nitride nanosheets for enhanced visible-light-driven water oxidation. Appl. Catal. B Environ. 223, 2–9 (2018)
Acknowledgements
The authors acknowledge the support of Universidad Nacional de Colombia in Bogotá. Edwin Murillo professor at Universidad Francisco de Paula Santander in Cúcuta, Colombia, for the laboratory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors certify that they have not signed an agreement with any sponsor of the research reported.
Rights and permissions
About this article
Cite this article
Cardenas-Flechas, L.J., Raba, A.M., Barba-Ortega, J. et al. Effect of Calcination Temperature on the Behavior of the Agglomerated \(\text{Co}_{3}\text{O}_{4}\) Nanoparticles Obtained Through the Sol–Gel Method. J Inorg Organomet Polym 31, 121–128 (2021). https://doi.org/10.1007/s10904-020-01685-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01685-5