Abstract
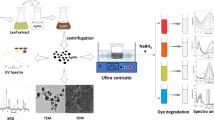
The present study reports the green synthesis of Silver nanoparticles (AgNPs) using Azadirachta indica (neem) leaf broth as reducing and capping agent in aqueous solution. The effect of different temperature on the morphology of dispersed AgNPs was studied. Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM) analysis results indicate that optimum temperature is 30 °C for the synthesis of nanoparticles. Fourier Transform Infrared Spectroscopy (FTIR) studies explain the presence of biomolecules such as terpenoids and flavanones responsible for capping and stabilizing of nanoparticles (NPs). The synthesized AgNPs showed excellent catalytic activity in oxidative degradation of Acid orange 10 (AO10) and Acid orange 52 (AO52) by peroxomonosulphate (PMS) in an aqueous medium. It was observed that the synthesized Nano-catalyst could effectively decompose oxidant to generate sulphate radicals (SRs) and degrade both dyes in aqueous solution by advanced oxidation process (AOP). The maximum degradation efficiency of AO10 reached 84% in 32 min and 90% in 15 min for AO52. The effect of several parameters such as the different concentration of dye, PMS, AgNPs and pH on the degradation efficiency of the process was investigated and Liquid Chromatography-Mass Spectrometry (LC-MS) analyses used for determining intermediates and end products during the degradation process. The AgNPs are expected to be a suitable alternative and play an important role in the fields of catalysis and environmental remediation.













Similar content being viewed by others
References
N. Yan, Y. Yuanb, P.J. Dyson, Dalton Trans. 42, 13294–13304 (2013). https://doi.org/10.1039/C3DT51180D
R.A. Soomro, A. Nafady, N. Memon, T.H. Sherazi, N.H. Kalwar, Talanta 130, 415–422 (2014). https://doi.org/10.1016/j.talanta.2014.07.023
A. Goel, R. Bhatt, Neetu, Int. J. Res. Chem. Environ. 2, 210–217 (2012)
N. Nagar, V. Devra, J. Envion. Chem. Eng. 5, 5793–5800 (2017). https://doi.org/10.1016/j.jece.2017.11.014
Y. Konishi, K. Ohno, N. Saitoh, T. Nomura, S. Nagamine, H. Hishida, Y. Takahashi, T. Uruga, J. Biotechnol. 128, 648–653 (2007). https://doi.org/10.1016/j.jbiotec.2006.11.014
I. Willner, R. Baron, B. Willner, Adv. Mater. 18, 1109–1120 (2006). https://doi.org/10.1002/adma.200501865
B.H. Patel, M.Z. Channiwala, S.B. Chaudhari, A.A. Mandot, J. Envion. Chem. Eng. 4, 2163–2169 (2016). https://doi.org/10.1016/j.jece.2016.03.046
S. Ahmed, S. Ullah, M. Ahmad, B.L. Swami, S. Ikram, J. Radiat. Res. Appl. Sci. 9, 1–7 (2016). https://doi.org/10.1016/j.jrras.2015.06.006
N. Nagar, S. Jain, P. Kachhawah, V. Devra, Korean J. Chem. Eng. 33, 2990–2997 (2016). https://doi.org/10.1007/s11814-016-0156-9
K. Khaldi, M. Hadjel, A. Benyoucef, Surf. Eng. Appl. Electrochem. 54, 194–202 (2018). https://doi.org/10.3103/S1068375518020084
A. Belalia, A. Zehhaf, A. Benyoucef, Polym. Sci. Ser, B+ 60, 816–824 (2018). https://doi.org/10.1134/S1560090418060039
A. Ahmad, A. Idris, B. Hameed, Desalin. Water Treat. 41, 224–231 (2012). https://doi.org/10.1080/19443994.2012.664717
L. Ahmad, W.A. Harris, B.S. Ooi, J. Teknol. 36, 31–44 (2012). https://doi.org/10.11113/jt.v36.581
R. Singh, M. Kumar, L. Tashi, H. Khajuria, H.N. Sheikh, Nanochem. Res. 3, 149–159 (2018). https://doi.org/10.22036/ncr.2018.02.004
Y. Yuan, T. Luo, J. Xu, J. Li, F. Wu, M. Brigante, G. Mailhot, Chem. Eng. J. 362, 183–189 (2019). https://doi.org/10.1016/j.cej.2019.01.010
F. Zhu, C. Liu, H. Ling, H. Jiang, A. Wu, Li, Appl. Catal. B 242, 238–248 (2019). https://doi.org/10.1016/j.apcatb.2018.09.088
S. Rodriguez, L. Vasquez, D. Costa, A. Romero, A. Santos, Chemosphere. 101, 86–92 (2014). https://doi.org/10.1016/j.chemosphere.2013.12.037
J. Madhavan, P. Maruthamuthu, S. Murugesan, M. Ashokkumar, Appl. Catal. A 368, 35–39 (2009). https://doi.org/10.1016/j.apcata.2009.08.008
J. Madhavan, B. Muthuraaman, S. Murugesan, S. Anandan, P. Maruthamuthu, Sol. Energy Mater.Sol. Cells. 90, 1875–1887 (2006). https://doi.org/10.1016/j.solmat.2005.12.001
J. Zhang, M. Chen, L. Zhu, RSC Adv. 6, 758–768 (2016). https://doi.org/10.1039/C5RA22457H
X.Y. Chen, J.W. Chen, X.L. Qiao, D.G. Wang, X.Y. Cai, Appl. Catal. B 80, 116–121 (2008). https://doi.org/10.1016/j.chemosphere.2006.10.032
Z. Huixuan, L. Huarui, W. Zhongjuan, L. Bo, C. Xiuwen, C. Qingfeng, J. Nanosci. Nanotechnol. 18, 6942–6948 (2018). https://doi.org/10.1166/jnn.2018.15800
A. Meetani, M.A. Rauf, S. Hisaindee, A. Khaleel, A. Alzamly, A. Ahmad, RSC Adv. 1, 490–497 (2011). https://doi.org/10.1039/C1RA00177A
A. Obeid, D. Bée, S.B. Talbot, V. Jaafar, S. Dupuis, V. Abramson, M. Cabuil, Welschbillig, J. Colloid Interface Sci. 410, 52–58 (2013). https://doi.org/10.1016/j.jcis.2013.07.057
P. Banerjee, M. Satapathy, A. Mukhopahayay, P. Das, Bioresourc. Bioprocess. 1, 1–10 (2014). https://doi.org/10.1186/s40643-014-0003-y
S. Jain, N. Nagar, V. Devra, Int. J. Curr. Eng. Technol. 5, 966–973 (2015). E-ISSN 2277–4106
S.S. Shankar, A. Rai, A. Ahmad, M. Sastry, J. Colloid Interface Sci. 275, 496–502 (2004). https://doi.org/10.1016/j.jcis.2004.03.003
S. Hisaindee, M.A. Meetani, M.A. Rauf, Trends Anal. Chem. 49, 31–44 (2013). https://doi.org/10.1016/j.trac.2013.03.011
J. Zhang, M. Chen, L. Zhu, RSC Adv. 6, 47562–47569 (2016). https://doi.org/10.1039/C6RA07231C
T. Chen, Y. Zheng, J.-M. Lin, G. Chen, J. Am. Soc. Mass Spectrom. 19, 997–1003 (2008). https://doi.org/10.1016/j.jasms.2008.03.008
Q. Cai, Y.-Z. Zhu, Z.-S. Wei, J.-Q. Hu, S.-D. Pan, R.-Y. Jin, C.-Y. Xiao, M.-C. Dong, Sci. Total Environ. 580, 966–973 (2017). https://doi.org/10.1016/j.scitotenv.2016.12.047
N. Nagar, V. Devra, Environ. Technol. Innov. 10, 281–289 (2018). https://doi.org/10.1016/j.eti.2018.03.005
R. Xiao, Z. Luo, Z. Wei, S. Luo, R. Spinney, W. Yang, D.D. Dionysiou, Curr. Opin. Chem. Eng. 19, 51–58 (2018). https://doi.org/10.1016/j.coche.2017.12.005
J. Santhanalakshmi, V. Dhanalakshmi, Indian J. Sci. Technol. 5, 3234–3838. ISSN:0974–6846 (2012)
K. Lal, A. Garg, Process Saf. Environ. Prot. (PSEP) 111, 766–774 (2017). https://doi.org/10.1016/j.psep.2017.09.005
R.J. Dougherty, J. Singh, V.V. Krishnan, J. Mol. Struct. 1131, 196–200 (2017). https://doi.org/10.1016/j.molstruc.2016.11.038
C. Tan, N. Gao, Y. Deng, J. Deng, S. Zhou, J. Li, X. Xin, J. Hazard. Mater. 276, 452–460 (2014). https://doi.org/10.1016/j.jhazmat.2014.05.068
C. Zhu, F. Zhu, D.D. Dionysiou, D. Zhou, F. Fang, J. Gao, Water Res. 139, 66–73 (2018). https://doi.org/10.1016/j.watres.2018.03.069
Acknowledgements
This work was supported in part by Department of Science and Technology sponsored FIST Laboratory of our institution for experimental work, MNIT Jaipur for SEM and TEM analysis of synthesis of AgNPs, SAIF (Punjab University, Chandigarh) for LC-MS analysis and University Grants Commission for financial support as JRF (Ref. No: 22/12/2013(ii)EU-V).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nagar, N., Devra, V. Textile Dyes Degradation from Activated Peroxomonosulphate by Green synthesize Silver Nanoparticles: A Kinetic Study. J Inorg Organomet Polym 29, 1645–1657 (2019). https://doi.org/10.1007/s10904-019-01127-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01127-x