Abstract
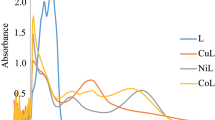
A series of chelated metal complexes, [Co(LI)2] (1), [Ni(LI)2] (2), [Cu(LI)2] (3) [Co(LII)2] (4), [Ni(LII)2] (5) and [Cu(LII)2] (6) were designed and synthesized from newly synthesized Schiff bases, LI = 2-((E)-(5-(4-fluorophenyl)isoxazol-3-ylimino)methyl)-5-methylphenol and LII = 2-((E)-(5-(4-fluorophenyl)isoxazol-3-ylimino)methyl)-4-chlorophenol. The synthesized compounds were characterized by elemental analysis, nuclear magnetic resonance spectroscopy (NMR), electronic spectroscopy (UV–Vis), infrared spectroscopy (FT-IR), magnetic susceptibility (µeff), electron spin resonance spectroscopy (ESR), Thermogravimetric analysis (TGA), scanning electron microscopy (SEM), energy dispersive X-ray analysis (EDX), and powder X-ray diffraction analysis (P-XRD). The spectral investigations have been clearly suggested 1:2 (metal: ligand) stoichiometric complexes with square planar geometrical arrangement around the metal ion. The thermal gravimmetric analysis (TGA) of these complexes indicates greater thermal stability and various steps involved in thermal decomposition of metal complexes. The binding ability between these metal complexes and calf thymus DNA (CT-DNA) was investigated by UV–Vis, fluorescence spectroscopy and viscometric experiments, which disclosed that, the complexes interacted to CT-DNA via an intercalation binding mode. The cleavage property of metal complexes against pBR322 DNA has been explored by gel electrophoresis technique mediated by UV-illumination and H2O2, showed momentous cleavage activity. Antioxidant activity of all complexes was determined by DPPH free radical scavenging experiment and showed prominent antioxidant activity. Further, the antibacterial and antifungal activities of all compounds were screened against bacterial and fungal strains via in-vitro disc diffusion method. These studies revealed that the complexes showed comparatively more antimicrobial activity than free ligands against tested microbial strains.
Graphical Abstract
















Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study were included in the manuscripts butany additional data needed are will be available from the corresponding author on reasonable request.
References
Saleem M, Hanif M, Rafiq M, Hassan M, Tahir T (2023) Synthesis, Characterization, Optical Properties, Molecular Modeling and Urease Inhibition Analysis of Organic Ligands and Their Metal Complexes. J Fluoresc 33(1):113–124
More MS, Joshi PG, Mishra YK, Khanna PK (2019) Metal complexes driven from Schiff bases and semicarbazones for biomedical and allied applications. Mater Today Chem 14:100–195
Sunitha N, Raj CI, Kumari BS (2023) Synthesis, spectral studies, biological evaluation and molecular docking studies of metal complexes from coumarin derivative. J Mol Struct 5(1285):135443
Azadbakht R, Chidan N, Menati S, Koolivand M (2023) A new Azo-Schiff base dual-mode chemosensor: Colorimetric detection of cobalt ions and fluorometric detection of aluminum ions in aqueous ethanol solution. J Fluoresc 33(2):527–538
Gou Y, Li J, Fan B, Xu B, Zhou M, Yang F (2017) Structure and biological properties of mixed-ligand Cu(II) Schiff base complexes as potential anticancer agents. Eur J Med Chem 134:207–217
Kateshiya MR, Desai ML, Malek NI, Kailasa SK (2023) Advances in Ultra-small Fluorescence Nanoprobes for Detection of Metal Ions, Drugs, Pesticides and Biomarkers. J Fluoresc 33(3):775–798
Alizadeh R, Yousuf I, Afzal M, Srivastav S, Srikrishna S, Arjmand F (2015) Enantiomeric fluoro-substituted benzothiazole Schiff base-valine Cu(II)/Zn(II) complexes as chemotherapeutic agents: DNA binding profile, cleavage activity, MTT assay and cell imaging studies. J Photochem Photobiol B 143:61–73
Ramesh G, Kumar MP, Rambabu A, Vamsikrishna N, Daravath S (2018) Shivaraj, Synthesis, spectral characterization, DNA binding, cleavage and biological evaluation on Co(II), Ni(II) and Cu(II) complexes of substituted isoxazole Schiff bases, Asian. J Chem Sci 4:1–20
Metcalfe C, Thomas JA (2003) Kinetically inert transition metal complexes that reversibly bind to DNA. Chem Soc Rev 32:215–224
Rad FV, Housaindokht MR, Jalal R, Hosseini HE, Doghaei AV, Goghari SS (2014) Spectroscopic and molecular modelling based approaches to study on the binding behavior of DNA with a copper(II) complex. J Fluoresc 24:1225–1234
Ganji N, Chityala VK, Kumar MP, Rambabu A, Vamsikrishna N, Daravath S (2017) Shivaraj, DNA incision evaluation, binding investigation and biocidal screening of Cu(II), Ni(II) and Co(II) complexes with isoxazole Schiff bases. J Photochem Photobiol B 175:132–140
Kumar MP, Vamsikrishna N, Ramesh G, Subhashini NJP, Nanubolu JB (2017) Shivaraj, Cu(II) complexes with 4-amino-3,5-dimethyl isoxazole and substituted aromatic aldehyde Schiff bases: synthesis, crystal structure, antimicrobial activity, DNA binding and cleavage studies. J Coord Chem 70:1368–1388
Ramesh G, Daravath S, Ganji N, Rambabu A, Venkateswarlu K (2020) Shivaraj, Facile synthesis, structural characterization, DNA binding, incision evaluation, antioxidant and antimicrobial activity studies of Cobalt(II), Nickle(II) and Copper(II) complexes of 3-amino-5-(4-fluorophenyl)isoxazole derivatives. J Mol Struct 1202:127338
Shafaatian B, Soleymanpour A, Oskouei NK, Notash B, Rezvani SA (2014) Synthesis, crystal structure, fluorescence and electrochemical studies of a new tridentate Schiff base ligand and its nickel(II) and palladium(II)complexes. Spectrochim Acta A 128:363–369
Daravath S, Rambabu A, Shankar DS (2019) Shivaraj, Structure elucidation of copper (II), cobalt (II) and nickel (II) complexes of benzothiazole derivatives: Investigation of DNA binding, nuclease efficacy, free radical scavenging and biocidal properties. Chem Data Collect 24
Kamal A, Bharathi EV, Reddy JS, Ramaiah MJ, Dastagiri D, Reddy MK, Viswanath A, Reddy TL, Shaik TB, Pushpavalli SNCVL, Bhadra MP (2011) Synthesis and biological evaluation of 3,5-diaryl isoxazoline/isoxazole linked 2,3-dihydroquinazolinone hybrids as anticancer agents. Eur J Med Chem 46:691–703
Burcu C, Sinoplu E, Ibis K, Guzelcan EA, Atalay RC, Banoglua E (2018) Synthesis and cellular bioactivities of novel isoxazole derivatives incorporating an arylpiperazine moiety as anticancer agents. J Enzyme Inhib Med Chem 33:1352–1361
Kumar MP, Tejaswi S, Rambabu A, Kalalbandi VKA (2015) Shivaraj, Synthesis, crystal structure, DNA binding and cleavage studies of copper(II) complexes with isoxazole Schiff bases. Polyhedron 102:111–120
Ramesh G, Daravath S, Swathi M, Sumalatha V, Shanker DS (2020) Shivaraj, Investigation on Co(II), Ni(II), Cu(II) and Zn(II) complexes derived from quadridentate salen-type Schiff base: Structural characterization, DNA interactions, antioxidant proficiency and biological evaluation. Chem Data Collect 28
Protogeraki C, Andreadou EG, Perdih F, Turel I, Pantazaki AA, Psomas G (2014) Cobalt(II) complexes with the antimicrobial drug enrofloxacin: Structure, antimicrobial activity, DNA- and albumin-binding. Eur J Med Chem 86:189–201
Manikandan RB, Viswanathamurthi P, Velmurugan K, Nandhakumar R, Hashimoto T, Endo A (2014) Synthesis, characterization and crystal structure of cobalt(III) complexes containing 2-acetylpyridine thiosemicarbazones: DNA/protein interaction, radical scavenging and cytotoxic activities. J Photochem Photobiol B 130:205–216
Tabrizi L, McArdle P, Erxleben A, Chiniforoshan H (2015) Nickel(II) and cobalt(II) complexes of lidocaine: Synthesis, structure and comparative in vitro evaluations of biological perspectives. Eur J Med Chem 103:516–529
Wei Q, Dong J, Zhao P, Li M, Cheng F, Kong J, Li L (2016) DNA binding, BSA interaction and SOD activity of two new nickel(II) complexes with glutamine Schiff base ligands. J Photochem Photobiol B 161:355–367
Rambabu A, Ganji N, Daravath S, Ramesh G, Shivaraj (2020) An investigation on DNA binding, cleavage and antimicrobial properties of mononuclear Co (II), Ni (II) and Cu (II) complexes derived from N, O Donor Schiff Bases. J Fluore 30:1397–1410
Manikandamathavan VM, Nair BU (2013) DNA binding and cytotoxicity of copper (II) imidazole terpyridine complexes: Role of oxyanion, hydrogen bonding and π-π* interaction. Eur J Med Chem 68:244–252
Kyhoiesh HA, Al-Adilee KJ (2021) Synthesis, spectral characterization, antimicrobial evaluation studies and cytotoxic activity of some transition metal complexes with tridentate (N, N, O) donor azo dye ligand. Res Chem 3
Kyhoiesh HAK, Al-Adilee KJ (2022) Synthesis, spectral characterization and biological activities of Ag(I), Pt(IV) and Au(III) complexes with novel azo dye ligand (N, N, O) derived from 2-amino-6-methoxy benzothiazole. Chem Pap 76:2777–2810
Kyhoiesh HA, Al-Adilee KJ (2023) Pt (IV) and Au (III) complexes with tridentate-benzothiazole based ligand: synthesis, characterization, biological applications (antibacterial, antifungal, antioxidant, anticancer and molecular docking) and DFT calculation. Inorg Chim Acta 555
Kyhoiesh HA, Hassan HM (2024) Synthesis, Characterization, in silico DFT, Molecular docking, ADMET Profiling Studies and Toxicity Predictions of Ag (I) Complex Derived from 4-Aminoacetophenone. Chem Select 9
Jayakumar S, Mahendiran D, Srinivasan T, Mohanraj G, Rahiman AK (2016) Theoretical investigation, biological evaluation and VEGFR2 kinase studies of metal(II) complexes derived from hydrotris(methimazolyl)borate. J Photochem Photobiol B 155:66–77
Bauer AW, Kirby WMM, Sherries JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45:493–496
Thompson RS, Rivara FP, Thompson DC (1989) A case-control study of the effectiveness of bicycle safety helmets. New Eng J Med 320:1361–1367
Agraharia B, Layeka S, Ganguly R, Pathak DD (2018) Synthesis and crystal structures of salen-type Cu(II) and Ni(II) Schiff base complexes: application in [3+2]-cycloaddition and A3-coupling reactions. New J Chem 42:13754–13762
Pandiyan RP, Raman N (2016) Biological screening and DNA nuclease activity of transition metal complexes of N2O2type of Knoevenagel condensate Schiff base. Appl Organomet Chem 30:531–539
Daravath S, Rambabu A, Vamsikrishna N, Ganji N (2019) Shivaraj, Synthesis, structural characterization, antioxidant, antimicrobial, DNA incision evaluation and binding investigation studies on copper(II) and cobalt(II) complexes of benzothiazole cored Schiff bases. J Coord Chem 72:1973–1993
Daravath S, Rambabu A, Ganjia N, Ramesh G, Lakshmi PVA (2022) Shivaraj, Spectroscopic, quantum chemical calculations, antioxidant, anticancer, antimicrobial, DNA binding and photo physical properties of bioactive Cu (II) complexes obtained from trifluoromethoxy aniline Schiff bases. J Mol Stru 1249:131601
Reddy PR, Rajeshwar S, Satyanarayana B (2016) Synthesis, characterization of new copper (II) Schiff base and 1,10 phenanthroline complexes and study of their bio-properties. J Photochem Photobiol B 160:217–224
Kneubuhl FK (1960) Line shapes of slectron paramagnetic resonance signals produced by powders, glasses, and viscous liquids. J Chem Phys 33:1074–1078
Hathaway BJ, Billing DE (1970) The electronic properties and stereochemistry of mononuclear complexes of the copper(II) ion. Coord Chem Rev 5:143–207
Kareem A, Arshad M, Nami SA, Nishat N (2016) Herbo-mineral based Schiff base ligand and its metal complexes: Synthesis, characterization, catalytic potential and biological applications. J Photochem Photobiol B 160:163–171
Vamsikrishna N, Kumar MP, Ramesh G, Ganji N, Daravath S, Shivaraj (2017) DNA interactions and biocidal activity of metal complexes of benzothiazole Schiff bases: Synthesis, characterization and validation. J Chem Sci 129:609–622.
M Shamsi S Yadav F Arjmand 2014 Synthesis and characterization of new transition metal Cu(II), Ni(II) and Co(II) L–phenylalanine–DACH conjugate complexes: In vitro DNA binding, cleavage and molecular docking studies J Photochem Photobiol B 136 1 11
Lepecqc JB, Paoletti CJ (1967) A Fluorescent Complex between Ethidium Bromide and Nucleic Acids Physical-Chemical Characterization. J Mol Biol 27:87–106
Tabassum S, Zaki M, Afzal M, Arjmand F (2013) New modulated design and synthesis of quercetin–Cu II/Zn II–Sn 2 IV scaffold as anticancer agents: in vitro DNA binding profile, DNA cleavage pathway and Topo-I activity. Dalton Trans 42:10029–10041
Arshad N, Abbas N, Bhatti MH, Rashid N, Tahir MN, Saleem S, Mirza B (2012) Synthesis, crystal structure, DNA binding and in vitro biological studies of Ni(II), Cu(II) and Zn(II) complexes of N-phthaloylglycine. J Photochem Photobiol B 117:228–239
Kawade VA, Kumbhar AA, Kumbhar AS, Nather C, Erxleben A, Sonawaned UB, Joshi RR (2011) Mixed ligand cobalt(II) picolinate complexes: synthesis, characterization, DNA binding and photocleavage. Dalton Trans 40:639–650
Kathiresan S, Anand T, Mugesh S, Annaraj J (2015) Synthesis, Spectral Characterization and DNA bindings of Tridentate N2O donor Schiff base Metal(II) Complexes. J Photochem Photobiol B 148:290–301
Merkel PB, Kearns DR (1972) Remarkable solvent effects on the lifetime of 1.DELTA.g oxygen. J Am Chem Soc 94:1029–1030
Sumalatha V, Daravath S, Rambabu A, Ramesh G (2021) Antioxidant, antimicrobial, DNA binding and cleavage studies of novel Co (II), Ni (II) and Cu (II) complexes of N, O donor Schiff bases: Synthesis and spectral characterization. J Mol Struct 1229:29606
Overton CE (1901) Studien über die Narkose: zugleich ein Beitrag zur allgemeinen Pharmakologie Switzerland. G Fischer 195
Anjaneyulu Y, Rao RP (1986) Preparation, characterization and antimicrobial activity studies on some ternary complexes of Cu(II) with Acetylacetone and various salicylic acids. Synth React Inorg Met Org Chem 16:257–272
Tweedy BG (1964) Plant extracts with metal ions as potential antimicrobial agents. Phytopathology 55:910–914
Rehman W, Hassan Z, Rashid U, Rahim F, Abid OR, Waseem Md (2014) Synthesis, characterization antibacterial and antifungal activity of some transition metal complexes. Med Chem Res 23:2207–2211
Acknowledgements
The authors are acknowledged to the Head, Department of Chemistry, University College of Science and Director-CFRD, Osmania University, Hyderabad for providing laboratory facilities and spectral analysis. We are conveying sincere thanks to the Head, SAIF, IIT Bombay for analyzing the ESR spectral data.
Funding
DST − SERB (SB/EMEQ-141/2014), UGC − UPE (FAR) (002/OU/UPE/FAR/CO/2013), OU-DST-PURSE-II (C-DST-PURSE-II/9/2017) and DST − INSPIRE (DST/INSPIRE Fellowship/2014/167), New Delhi, India for providing financial assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Conflicts of Interest
The author declares they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramesh, G., Daravath, S., Babu, K.J. et al. Design, Synthesis, Structural Investigation and Photo Induced Biological Investigations of Co(II), Ni(II) and Cu(II) Complexes Derived from N,O Donor Schiff Bases. J Fluoresc (2024). https://doi.org/10.1007/s10895-024-03657-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10895-024-03657-1