Abstract
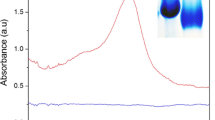
In this paper, a fluorescent aptamer sensor was constructed based on the carbon dots (CDs) and graphene oxide (GO). This sensor combines the excellent fluorescence performance of CDs with the high specificity of aptamer, which can detect progesterone (P4) with high sensitivity and selectivity. In the absence of P4, the CDs-aptamer system and GO form a fluorescence resonance energy transfer process (FRET), which quenches the fluorescence of the CDs. When P4 is added, the aptamer specifically binds to it, resulting the fluorescence of the CDs is recovered. At optimal conditions, the fluorescence intensity recovered by the CDs has a linear relationship with the concentration of P4 in the range of 0.1–120 nM and the detection limit is 3.3 × 10–11 M. Besides, the sensor has satisfactory detection results of P4 in milk, indicating that constructed method has enormous potential for application in food safety.









Similar content being viewed by others
Availability of Data and Material
Unpublished data.
Code Availability
Not applicable.
References
Ynsa MD, Ager FJ, Millan JC, Gomez-Zubelbia MA, Pinheiro T (2004) Effect of hormone replacement therapy on the elemental contents of uterine tissue. Biol Trace Elem Res 101(1):37–46. https://doi.org/10.1385/bter:101:1:37
Lishko PV, Botchkina IL, Kirichok Y (2011) Progesterone activates the principal Ca2+ channel of human sperm. Nature 471(7338):387–391. https://doi.org/10.1038/nature09767
Ricanyova J, Gadzala-Kopciuch R, Reiffova K, Bazel Y, Buszewski B (2010) Molecularly imprinted adsorbents for preconcentration and isolation of progesterone and testosterone by solid phase extraction combined with HPLC. Adsorption J Int Adsorption Soc 16(4–5):473–483. https://doi.org/10.1007/s10450-010-9265-7
Sherwin BB (1999) Progestogens used in menopause - Side effects mood and qualify of life. J Reprod Med 44(2):227–232
Greendale GA, Reboussin BA, Hogan P, Barnabei VM, Shumaker S, Johnson S, Barrett-Connor E, Estrogen P, Postmenopausal Estrogen/Progestin Interventions Trial Investigators (1998) Symptom relief and side effects of postmenopausal hormones: Results from the postmenopausal estrogen/progestin interventions trial. Obstet Gynecol 92(6):982–988. https://doi.org/10.1016/s0029-7844(98)00305-6
Key TJ, Appleby PN, Reeves GK, Roddam AW, Helzlsouer KJ, Alberg AJ, Rollison DE, Dorgan JF, Brinton LA, Overvad K, Kaaks R, Trichopoulou A, Clavel-Chapelon F, Panico S, Duell EJ, Peeters PHM, Rinaldi S, Riboli E, Fentiman IS, Dowsett M, Manjer J, Lenner P, Hallmans G, Baglietto L, English DR, Giles GG, Hopper JL, Severi G, Morris HA, Koenig K, Zeleniuch-Jacquotte A, Arslan AA, Toniolo P, Shore RE, Krogh V, Micheli A, Berrino F, Muti P, Barrett-Connor E, Laughlin GA, Kabuto M, Akiba S, Stevens RG, Neriishi K, Land CE, Cauley JA, Lui LY, Cummings SR, Gunter MJ, Rohan TE, Strickler HD, Endogenous Hormones and Breast Cancer Collaborative Group (2011) Circulating sex hormones and breast cancer risk factors in postmenopausal women: reanalysis of 13 studies. Br J Cancer 105(5):709–722. https://doi.org/10.1038/bjc.2011.254
Andersson AM, Skakkebaek NE (1999) Exposure to exogenous estrogens in food: possible impact on human development and health. Eur J Endocrinol 140(6):477–485. https://doi.org/10.1530/eje.0.1400477
Roelofs JB, Van Eerdenburg F, Hazeleger W, Soede NM, Kemp B (2006) Relationship between progesterone concentrations in milk and blood and time of ovulation in dairy cattle. Anim Reprod Sci 91(3–4):337–343. https://doi.org/10.1016/j.anireprosci.2005.04.015
Farlow DW, Xu X, Veenstra TD (2010) Consumption of Cow’s Milk and Possible Risk of Breast Cancer. Breast Care 5(1):44–46. https://doi.org/10.1159/000277938
Decheng S, Xia F, Zhiming X, Shulin W, Shi W, Peilong W (2021) Trace analysis of progesterone and 21 progestins in milk by ultra-performance liquid chromatography coupled with high-field quadrupole-orbitrap high-resolution mass spectrometry. Food Chem 361:130115. https://doi.org/10.1016/j.foodchem.2021.130115
Tschmelak J, Kappel N, Gauglitz G (2005) TIRF-based biosensor for sensitive detection of progesterone in milk based on ultra-sensitive progesterone detection in water. Anal Bioanal Chem 382(8):1895–1903. https://doi.org/10.1007/s00216-005-3261-x
Contreras-Jimenez G, Eissa S, Ng A, Alhadrami H, Zourob M, Siaj M (2015) Aptamer-based label-free impedimetric biosensor for detection of progesterone. Anal Chem 87(2):1075–1082. https://doi.org/10.1021/ac503639s
Kreuzer MP, McCarthy R, Pravda M, Guilbault GG (2007) Development of Electrochemical Immunosensor for Progesterone Analysis in Milk. Anal Lett 37(5):943–956. https://doi.org/10.1081/al-120030289
Zan GT, Wu T, Zhang ZL, Li J, Zhou JC, Zhu F, Chen HX, Wen M, Yang XC, Peng XJ, ChenJ, Wu QS (2022) Bioinspired Nanocomposites with Self-Adaptive Stress Dispersion for Super-Foldable Electrodes. Adv Sci 9(3):2103714. https://doi.org/10.1002/advs.202103714
Sun Y-P, Zhou B, Lin Y, Wang W, Fernando KAS, Pathak P, Meziani MJ, Harruff BA, Wang X, Wang H, Luo PG, Yang H, Kose ME, Chen B, Veca LM, Xie S-Y (2006) Quantum-sized carbon dots for bright and colorful photoluminescence. J Am Chem Soc 128(24):7756–7757. https://doi.org/10.1021/ja062677d
Zan GT, Wu T, Zhu F, He PF, Cheng YP, Chai SS, Wang Y, Huang XF, Zhang WX, Wan Y, Peng XJ, Wu QS (2021) A Biomimetic Conductive Super-foldable Material. Matter 4(10):3232–3247. https://doi.org/10.1016/j.matt.2021.07.021
Zhang Y-F, Maimaiti H, Zhang B (2017) Preparation of cellulose-based fluorescent carbon nanoparticles and their application in trace detection of Pb(II). RSC Adv 7(5):2842–2850. https://doi.org/10.1039/c6ra26684c
Chai SS, Zan GT, Dong KZ, Wu T, Wu QS (2021) Approaching Superfoldable Thickness-Limit Carbon Nanofiber Membranes Transformed from Water-Soluble PVA. Nano Lett 21(20):8831–8838. https://doi.org/10.1021/acs.nanolett.1c03241
Trapiella-Alfonso L, Costa-Fernandez JM, Pereiro R, Sanz-Medel A (2011) Development of a quantum dot-based fluorescent immunoassay for progesterone determination in bovine milk. Biosens Bioelectron 26(12):4753–4759. https://doi.org/10.1016/j.bios.2011.05.044
Cao L, Yu L, Yue J, Zhang Y, Ge M, Li L, Yang R (2020) Yellow-emissive carbon dots for “off-and-on” fluorescent detection of progesterone. Mater Lett 271:127760. https://doi.org/10.1016/j.matlet.2020.127760
Ma H, Liu J, Ali MM, Mahmood MAI, Labanieh L, Lu M, Iqbal SM, Zhang Q, Zhao W, Wan Y (2015) Nucleic acid aptamers in cancer research, diagnosis and therapy. Chem Soc Rev 44(5):1240–1256. https://doi.org/10.1039/c4cs00357h
Mairal T, Oezalp VC, Sanchez PL, Mir M, Katakis I, O’Sullivan CK (2008) Aptamers: molecular tools for analytical applications. Anal Bioanal Chem 390(4):989–1007. https://doi.org/10.1007/s00216-007-1346-4
Ellington AD, Szostak JW (1990) In vitro selection of RNA molecules that bind specific ligands. Nature 346(6287):818–822. https://doi.org/10.1038/346818a0
Li J, Yan H, Wang K, Tan W, Zhou X (2007) Hairpin fluorescence DNA probe for real-time monitoring of DNA methylation. Anal Chem 79(3):1050–1056. https://doi.org/10.1021/ac061694i
Tuerk C, Gold L (1990) Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249(4968):505–510. https://doi.org/10.1126/science.2200121
Siriangkhawut W, Sittichan P, Ponhong K, Chantiratikul P (2017) Stripping voltammetric determination of trace cadmium and lead in Thai organic unpolished rice after ultrasound-assisted digestion. J Food Compos Anal 59:145–152. https://doi.org/10.1016/j.jfca.2017.02.018
Yao D, Liang A, Yin W, Jiang Z (2014) Resonance light scattering determination of trace bisphenol A with signal amplification by aptamer-nanogold catalysis. Luminescence 29(5):516–521. https://doi.org/10.1002/bio.2578
Li P, Zhou L, Wei J, Yu Y, Yang M, Wei S, Qin Q (2016) Development and characterization of aptamer-based enzyme-linked apta-sorbent assay for the detection of Singapore grouper iridovirus infection. J Appl Microbiol 121(3):634–643. https://doi.org/10.1111/jam.13161
Wu X, Zhao Z, Bai H, Fu T, Yang C, Hu X, Liu Q, Champanhac C, Teng IT, Ye M, Tan W (2015) DNA Aptamer Selected against Pancreatic Ductal Adenocarcinoma for in vivo Imaging and Clinical Tissue Recognition. Theranostics 5(9):985–994. https://doi.org/10.7150/thno.11938
Zhu S, Meng Q, Wang L, Zhang J, Song Y, Jin H, Zhang K, Sun H, Wang H, Yang B (2013) Highly photoluminescent carbon dots for multicolor patterning, sensors and bioimaging. Angew Chem Int Ed Engl 52(14):3953–3957. https://doi.org/10.1002/anie.201300519
Chu BC, Wahl GM, Orgel LE (1983) Derivatization of unprotected polynucleotides. Nucleic Acids Res 11(18):6513–6529. https://doi.org/10.1093/nar/11.18.6513
Gao F, Zhang Q, Li X, Zhang Q, Mao T, Lu Y, Zhang W, Li H (2016) Comparison of standard addition and conventional isotope dilution mass spectrometry for the quantification of endogenous progesterone in milk. Accreditation Qual Assur 21(6):395–401. https://doi.org/10.1007/s00769-016-1236-6
Dong Y, Pang H, Yang HB, Guo C, Shao J, Chi Y, Li CM, Yu T (2013) Carbon-based dots co-doped with nitrogen and sulfur for high quantum yield and excitation-independent emission. Angew Chem Int Ed Engl 52(30):7800–7804. https://doi.org/10.1002/anie.201301114
Dong H, Gao W, Yan F, Ji H, Ju H (2010) Fluorescence Resonance Energy Transfer between Quantum Dots and Graphene Oxide for Sensing Biomolecules. Anal Chem 82(13):5511–5517. https://doi.org/10.1021/ac100852z
Xu Z-Q, Lan J-Y, Jin J-C, Gao T, Pan L-L, Jiang F-L, Liu Y (2015) Mechanistic studies on the reversible photophysical properties of carbon nanodots at different pH. Colloids Surf B 130:207–214. https://doi.org/10.1016/j.colsurfb.2015.04.012
Zhu Y, Xu Z, Gao J, Ji W, Zhang J (2020) An antibody-aptamer sandwich cathodic photoelectrochemical biosensor for the detection of progesterone. Biosens Bioelectron 160:112210. https://doi.org/10.1016/j.bios.2020.112210
Daems D, Lu J, Delport F, Marien N, Orbie L, Aernouts B, Adriaens I, Huybrechts T, Saeys W, Spasic D, Lammertyn J (2017) Competitive inhibition assay for the detection of progesterone in dairy milk using a fiber optic SPR biosensor. Anal Chim Acta 950:1–6. https://doi.org/10.1016/j.aca.2016.11.005
Jimena Monerris M, Javier Arevalo F, Fernandez H, Alicia Zon M, Gabriela Molina P (2012) Integrated electrochemical irnmunosensor with gold nanoparticles for the determination of progesterone. Sens Actuators B Chem 166:586–592. https://doi.org/10.1016/j.snb.2012.03.015
Funding
The authors thank the financial support by the National Nature Science Foundation of China (No. 21671132).
Author information
Authors and Affiliations
Contributions
The first author HYC performed the design, operation and data processing of the experiment, and wrote the manuscript. HL carried out the assistance of the experiment and the inspection and modification of the manuscript. YF completed the inspection and modification of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics
The authors declare that they have read the Ethical Responsibilities of Authors carefully, the authors meets its requirements.
Consent to Participate
The authors declare that they consent to participate.
Consent for Publication
The authors declare that they consent to publication.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cui, H., Lu, H., Yang, J. et al. A Significant Fluorescent Aptamer Sensor Based on Carbon Dots and Graphene Oxide for Highly Selective Detection of Progesterone. J Fluoresc 32, 927–936 (2022). https://doi.org/10.1007/s10895-022-02896-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-022-02896-4