Abstract
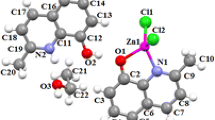
A new zinc complex, [Zn(mq)2], (mq = 2-methyl-8-hydroxyquinoline) was prepared via an electrochemical route from the oxidation of zinc metal in the presence of 2-methyl-8-hydroxyquinoline in a fast and facile process. The complex was fully characterized by means of NMR and IR spectra and elemental analysis. The nanostructure of the prepared compound was obtained by sonoelectrochemical process and studied by scanning electron microscopy, X-ray powder diffraction, IR spectroscopy and elemental analysis. Thermal stability of crystalline bulk and nano-size samples of the prepared compound was studied by thermal gravimetric and differential thermal analysis. The photoluminescence properties of the prepared compounds, as crystalline bulk and as nano-structure, have been investigated. The results showed a good correlation between the size and the shape of the complex particle and emission wavelength. The prepared complexes, as bulk and as nano-particles, were utilized as a precursor for preparation of ZnO nanoparticles by direct thermal decomposition at 550 °C in air. The nano-structures of ZnO were characterized by scanning electron microscopy, X-ray powder diffraction and IR spectroscopy.












Similar content being viewed by others
References
Semaltianos NG, Logothetidis S, Perrie W, Romani S, Potter RJ, Sharp M, French P, Dearden G, Watkins KG (2009) II–VI semiconductor nanoparticles synthesized by laser ablation J. Appl Phys A Mater Sci Process 94(3):641–647
Gaponenko SV (1998) Optical properties of semiconductor nanocrystals. Cambridge Univ. Press, Cambridge
Lin J, Lim SF, Mahmood S, Tan TL, Springham SV, Lee P, Rawat RS, Synthesis and characterization of FeCo nanoparticle colloid by pulsed laser ablation in distilled water. 33rd EPS Q22 Conference on Plasma Phys., Rome, Italy, 19–23 June 2006, ECA 301, 2087–2090.
Sylvestre JP, Kabashin AV, Sacher E, Meunier M (2005) Femtosecond laser ablation of gold in water: influence of the laser-produced plasma on the nanoparticle size distribution. J Appl Phys A: Mater Sci Process 80(4):753–758
Shen E, Wang C, Wang E, Kang Z, Gao L, Hu C, Xu L (2004) PEG-assisted synthesis of SnO2 nanoparticles. Mater Lett 58:3761–3764
Carbone L, Kudera S, Carlino E, Parak WJ, Giannini C, Cingolani R, Manna L (2006) Multiple wurtzite twinning in CdTe nanocrystals induced by methylphosphonic acid. J Am Chem Soc 128(3):748–755
Xie J, Cao X, Li J, Zhan H, Xia Y, Zhou Y (2005) Multiple wurtzite twinning in CdTe nanocrystals induced by methylphosphonic acid. Ultrason Sonochem 12(4):289–293
Regragui M, Addou M, Outzourhit A, Bernede JC, Idrissi EE, Benseddik E, Kachouane A (2000) Preparation and characterization of pyrolytic spray deposited electrochromic tungsten trioxide. Thin Solid Films 358(1):40–45
Ghanbari K, Mousavi MF, Shamsipur M (2006) Preparation of polyaniline nanofibers and their use as a cathode of aqueous rechargeable batteries. Electrochim Acta 52:1514–1522
Suslick KS, Choe SB, Cichowlas AA, Grinstaff MW (1991) Sonochemical synthesis of amorphous iron. Nature 353:414–416
Aslani A, Morsali A (2009) Hydrothermal and sonochemical synthesis of a nano- sized 2D lead(II) coordination polymer: a precursor for nano-structured PbO and PbBr2. J Mol Struct 929:187–192
Zhang KL, Zhou F, Gao HY, Pan ZC, Lin JG, Guo R (2008) Preparation and characterization of a metal-organic three-dimensional framework based on isonicotinate [Pb(INA)2] (INA = isonicotinate, NC5H4-4-CO2−). J Coord Chem 61:1494–1502
Wang SF, Gu F, Lu MK (2006) Sonochemical synthesis of hollow PbS nanospheres. Langmuir 22(1):398–401
Xu JZ, Xu S, Geng J, Li GX, Zhu JJ (2006) The fabrication of hollow spherical copper sulfide nanoparticle assemblies with 2-hydroxypropyl-β-cyclodextrin as a template under sonication. Ultrason Sonochem 13:451–454
Zhang SY, Liu Y, Ma X, Chen HY (2006) Rapid large-scale synthesis and single crystalline selenium nanotubes. J Phys Chem B 110:9041–9047
Lei H, Tang YJ, Wei JJ, Li J, Li XB, Shi HL (2007) Synthesis of tungsten nanoparticles by sonoelectrochemistry. Ultrason Sonochem 14(1):81–83
Kumar VG, Kim KB (2006) Organized and highly dispersed growth of MnO2 nano-rods by sonochemical hydrolysis of Mn3acetate. Ultrason Sonochem 13:549–556
Askarinezhad A, Morsali A (2008) Syntheses and characterization of CdCO3 and CdO nanoparticles by using a sonochemical method. Mater Lett 62:478–482
Wang ZL (2004) Zinc oxide nanostructures: growth, properties, and applications. J Phys Condens Matter 16:R829–R858
Hoffmann MR, Martin ST, Choi W, Bahnemann DW (1995) Environmental applications of semiconductor Photocatalysis. Chem Rev 95(1):69–96
Habeeb JJ, Tuck DC, Walters FH (1978) Direct electrochemical synthesis of Some metal Chelate complexes. J Coord Chem 8:27–33
Jiang P, Zhu W, Gan Z, Huang W, Li J, Zeng H, Shi J (2009) Electron transport properties of an ethanol-soluble AlQ3-based coordinationpolymer and its applications in OLED devices. J Mater Chem 19:4551–4556
Gupta P, Ramrakhiani M (2009) Influence of the particle size on the optical properties of CdSe nanoparticles. Open Nanosci J 3:15–19
Kowshik M, Vogel W, Urban J, Kulkarni SK, Paknikar KM (2002) Microbial synthesis of semiconductor PbS Nanocrystallites. Adv Mater 14:815–818
Xiong SL, Xi BJ, Xu DC, Wang CM, Feng XM, Zhou HY, Qian YT (2007) l-cysteine-assisted tunable synthesis of PbS of various morphologies. J Phys Chem C 111:16761–16767
Liu X, Wu X, Cao H, Chang RPH (2004) Growth mechanism and properties of ZnO nanorods synthesized by plasma-enhanced chemical vapor deposition. J Appl Phys 95:3141–3147
Mills G, Li ZG, Meisel D (1988) Photochemistry and spectroscopy of colloidal arsenic Sesquisulfide. J Phys Chem 92:822–828
Acknowledgments
The authors thank the Vice-President’s Office for Research Affairs of Islamic Azad University of Islam Shahr branch and Shahr-e- Ghods branch for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jodaian, V., Shahrjerdi, A. & Mirahmadpour, P. Sonoelectrochemical Synthesis of Nano Zinc(II) Complex with 2-Methyl-8-Hydroxyquinoline Ligand: a Precursor to Produce Pure Phase Nano-Sized Zinc(II) Oxide. J Fluoresc 27, 715–722 (2017). https://doi.org/10.1007/s10895-016-2000-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-2000-3