Abstract
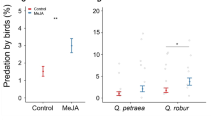
To what extent particular plant defences against herbivorous insects are constitutive or inducible will depend on the costs and benefits in their neighbourhood. Some defensive chemicals in leaves are thought to be costly and hard to produce rapidly, while others, including volatile organic compounds that attract natural enemies, might be cheaper and can be released rapidly. When surrounding tree species are more closely related, trees can face an increased abundance of both specialist herbivores and their parasitoids, potentially increasing the benefits of constitutive and inducible defences. To test if oaks (Quercus robur) respond more to herbivore attacks with volatile emission than with changes in leaf phenolic chemistry and carbon to nitrogen ratio (C: N), and whether oaks respond to the neighbouring tree species, we performed an experiment in a forest in Poland. Oak saplings were placed in neighbourhoods dominated by oak, beech, or pine trees, and half of them were treated with the phytohormone methyl jasmonate (elicitor of anti-herbivore responses). Oaks responded to the treatment by emitting a different volatile blend within 24 h, while leaf phenolic chemistry and C: N remained largely unaffected after 16 days and multiple treatments. Leaf phenolics were subtly affected by the neighbouring trees with elevated flavan-3-ols concentrations in pine-dominated plots. Our results suggest that these oaks rely on phenols as a constitutive defence and when attacked emit volatiles to attract natural enemies. Further studies might determine if the small effect of the neighbourhood on leaf phenolics is a response to different levels of shading, or if oaks use volatile cues to assess the composition of their neighbourhood.



Similar content being viewed by others
References
Agrawal AA, Fishbein M (2006) Plant defense syndromes. Ecology 87(S132–S149). https://doi.org/10.1890/0012-9658(2006)87[132:PDS]2.0.CO;2
Agrawal AA, Hastings AP (2019) Trade-offs constrain the evolution of an inducible defense within but not between plant species. Ecology 100:e02857. https://doi.org/10.1002/ecy.2857
Amo L, Jansen JJ, van Dam NM, Dicke M, Visser ME (2013) Birds exploit herbivore-induced plant volatiles to locate herbivorous prey. Ecol Lett 16:1348–1355. https://doi.org/10.1111/ele.12177
Amo L, Mrazova A, Saavedra I, Sam K (2022) Exogenous application of methyl jasmonate increases emissions of volatile organic compounds in pyrenean oak trees, Quercus pyrenaica. Biology 11:84
Åström M, Lundberg P (1994) Plant defence and stochastic risk of herbivory. Evol Ecol 8:288–298
Barbehenn RV, Jones CP, Karonen M, Salminen J-P (2006) Tannin composition affects the oxidative activities of tree leaves. J Chem Ecol 32:2235–2251
Barbier S, Gosselin F, Balandier P (2008) Influence of tree species on understory vegetation diversity and mechanisms involved—a critical review for temperate and boreal forests. Forest Ecol Manag 254:1–15
Bernays E, Chapman R (1994) Behavior: the process of host-plant selection. In: Host-plant selection by phytophagous insects. pp 95–165
Bertić M et al (2021) European oak chemical diversity – from ecotypes to herbivore resistance. New Phytol 232:818–834. https://doi.org/10.1111/nph.17608
Brosset A, Blande JD (2021) Volatile-mediated plant–plant interactions: volatile organic compounds as modulators of receiver plant defence, growth, and reproduction. J Exp Bot 73:511–528. https://doi.org/10.1093/jxb/erab487
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 410:577–580. https://doi.org/10.1038/35069058
Dicke M (2016) Plant phenotypic plasticity in the phytobiome: a volatile issue. Curr Opin Plant Biol 32:17–23
Dicke M, Hilker M (2003) Induced plant defences: from molecular biology to evolutionary ecology. Basic Appl Ecol 4:3–14
Ekholm A, Tack AJ, Pulkkinen P, Roslin T (2020) Host plant phenology, insect outbreaks and herbivore communities: the importance of timing. J Anim Ecol 89:829–841
Feeny PP (1968) Effect of oak leaf tannins on larval growth of the winter moth Operophtera brumata. J Insect Physiol 14:805–817
Feeny PP (1970) Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecol 51:565–581
Feeny P (1976) Plant apparency and chemical defense. Biochemical interaction between plants and insects. Springer, pp 1–40
Forkner RE, Marquis RJ, Lill JT (2004) Feeny revisited: condensed tannins as anti-herbivore defences in leaf‐chewing herbivore communities of Quercus. Ecol Entomol 29:174–187
Freitas AV, Oliveira PS (1996) Ants as selective agents on herbivore biology: effects on the behaviour of a non-myrmecophilous butterfly. J Anim Ecol :205–210
Frost CJ, Hunter MD (2008) Herbivore-induced shifts in carbon and nitrogen allocation in red oak seedlings. New Phytol 178:835–845. https://doi.org/10.1111/j.1469-8137.2008.02420.x
Ghirardo A, Heller W, Fladung M, Schnitzler JP, Schroeder H (2012) Function of defensive volatiles in pedunculate oak (Quercus robur) is tricked by the moth Tortrix Viridana. Plant Cell Environ 35:2192–2207. https://doi.org/10.1111/j.1365-3040.2012.02545.x
Glinwood R, Ninkovic V, Pettersson J (2011) Chemical interaction between undamaged plants – effects on herbivores and natural enemies. Phytochemistry 72:1683–1689. https://doi.org/10.1016/j.phytochem.2011.02.010
Goulas Y, Cerovic ZG, Cartelat A, Moya I (2004) Dualex: a new instrument for field measurements of epidermal ultraviolet absorbance by chlorophyll fluorescence. Appl Opt 43:4488–4496
Gripenberg S, Salminen J-P, Roslin T (2007) A tree in the eyes of a moth? Temporal variation in oak leaf quality and leaf-miner perform. Oikos 116:592–600. https://doi.org/10.1111/j.2007.0030-1299.15415.x
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference–performance relationships in phytophagous insects. Ecol Lett 13:383–393
Haukioja E, Hanhimäki S (1985) Rapid wound-induced resistance in white birch (Betula pubescens) foliage to the geometrid Epirrita autumnata: a comparison of trees and moths within and outside the outbreak range of the moth. Oecologia 65:223–228
Heil M (2004) Direct defense or ecological costs: responses of herbivorous beetles to volatiles released by wild lima bean (Phaseolus lunatus). J Chem Ecol 30:1289–1295
Hernández I, Alegre L, Munné-Bosch S (2011) Plant aging and excess light enhance flavan-3-ol content in cistus clusii. J Plant Physiol 168:96–102
Hoballah ME, Köllner TG, Degenhardt J, Turlings TCJ (2004) Costs of induced volatile production in maize. Oikos 105:168–180
Jactel H, Birgersson G, Andersson S, Schlyter F (2011) Non-host volatiles mediate associational resistance to the pine processionary moth. Oecologia 166:703–711. https://doi.org/10.1007/s00442-011-1918-z
Janssen A, Sabelis MW, Bruin J (2002) Evolution of herbivore-induced plant volatiles. Oikos 97:134–138
Kallenbach M, Oh Y, Eilers E, Veit D, Baldwin IT, Schuman MC (2014) A robust, simple, high-throughput technique for time‐resolved plant volatile analysis in field experiments. Plant J 78:1060–1072
Karban R (2011) The ecology and evolution of induced resistance against herbivores. Functional Ecology 25:339–347
Karban R (2021) Plant communication. Annu Rev Ecol Evol S 52:1–24
Karban R, Baldwin IT (2007) Induced responses to herbivory. University of Chicago Press
Karban R, Takabayashi J (2018) Chewing and other cues induce grass spines that protect meristems. Arthropod-Plant Interact 13:541–550. https://doi.org/10.1007/s11829-018-9666-1
Karban R, Baldwin IT, Baxter KJ, Laue G, Felton GW (2000) Communication between plants: induced resistance in wild tobacco plants following clipping of neighboring sagebrush. Oecologia 125:66–71. https://doi.org/10.1007/PL00008892
Keefover-Ring K, Trowbridge A, Mason CJ, Raffa KF (2016) Rapid induction of multiple terpenoid groups by Ponderosa pine in response to bark beetle-associated fungi. J Chem Ecol 42:1–12. https://doi.org/10.1007/s10886-015-0659-6
Kessler A, Halitschke R, Diezel C, Baldwin IT (2006) Priming of plant defense responses in nature by airborne signaling between Artemisia tridentata and Nicotiana attenuata. Oecologia 148:280–292 https://doi.org/10.1007/s00442-006-0365-8
Kondoh M, Williams IS (2001) Compensation behaviour by insect herbivores and natural enemies: its influence on community structure. Oikos 93:161–167
Kovács B, Tinya F, Ódor P (2017) Stand structural drivers of microclimate in mature temperate mixed forests. Agric for Meteorol 234:11–21
Kumar H (1992) Inhibition of ovipositional responses of Chilo partellus (Lepidoptera: Pyralidae) by the trichomes on the lower leaf surface of a maize cultivar. J Econ Entomol 85:1736–1739
Mallick S, Molleman F, Yguel B, Bailey R, Müller J, Jean F, Prinzing A (2023) Ectophagous folivores do not profit from rich resources on phylogenetically isolated trees. Oecologia 201:1–18
Martemyanov VV et al (2012) Rapid induced resistance of silver birch affects both innate immunity and performance of gypsy moths: the role of plant chemical defenses. Arthropod-Plant Interact 6:507–518. https://doi.org/10.1007/s11829-012-9202-7
Mertens D, Boege K, Kessler A, Koricheva J, Thaler JS, Whiteman NK, Poelman EH (2021) Predictability of biotic stress structures plant defence evolution. Trends Ecol Evol 36:444–456
Monson RK, Trowbridge AM, Lindroth RL, Lerdau MT (2022) Coordinated resource allocation to plant growth–defense trade-offs. New Phytol 233:1051–1066. https://doi.org/10.1111/nph.17773
Molleman F, Walczak U, Melosik I, Baraniak E, Piosik Ł, Prinzing A (2022) What drives caterpillar guilds on a tree: enemy pressure, leaf or tree growth, genetic traits, or phylogenetic neighbourhood? Insects 13(4):367. https://doi.org/10.3390/insects13040367
Moreira X, Mooney KA, Rasmann S, Petry WK, Carrillo-Gavilán A, Zas R, Sampedro L (2014) Trade‐offs between constitutive and induced defences drive geographical and climatic clines in pine chemical defences. Ecol Lett 17:537–546
Mrazova A, Sam K (2019) Exogenous application of methyl jasmonate to Ficus hahliana attracts predators of insects along an altitudinal gradient in Papua New Guinea. J Trop Ecol 35:157–164. https://doi.org/10.1017/s0266467419000117
Mrazova A, Houska Tahadlova M, Řehová V, Sam K (2023) The specificity of induced chemical defence of two oak species affects differently arthropod herbivores and arthropod and bird predation. Arthropod-Plant Interact 17:141–155
Ninkovic V, Markovic D, Dahlin I (2016) Decoding neighbour volatiles in preparation for future competition and implications for tritrophic interactions perspectives in plant ecology. Evol Syst 23:11–17. https://doi.org/10.1016/j.ppees.2016.09.005
Nykänen H, Koricheva J (2004) Damage-induced changes in woody plants and their effects on insect herbivore performance: a meta-analysis. Oikos 104:247–268. https://doi.org/10.1111/j.0030-1299.2004.12768.x
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MHH, Oksanen MJ, Suggests M (2007) The vegan package. Community ecology package 10:719
Pearse IS et al (2020) Generalising indirect defence and resistance of plants. Ecol Lett 23:1137–1152. https://doi.org/10.1111/ele.13512
Pellissier L, Moreira X, Danner H, Serrano M, Salamin N, van Dam NM, Rasmann S (2016) The simultaneous inducibility of phytochemicals related to plant direct and indirect defences against herbivores is stronger at low elevation. J Ecol 104:1116–1125. https://doi.org/10.1111/1365-2745.12580
Pihain M, Gerhold P, Ducousso A, Prinzing A (2019) Evolutionary response to coexistence with close relatives: increased resistance against specialist herbivores without cost for climatic-stress resistance. Ecol Lett 22:1285–1296
Price PW, Bouton CE, Gross P, McPheron BA, Thompson JN, Weis AE (1980) Interactions among three trophic levels: influence of plants on interactions between insect herbivores and natural enemies. Annu Rev Ecol Syst 11:41–65
R_Core_Team (2023) R: a language and environment for statistical computing, 4.3 edn. R Foundation for Statistical Computing, Vienna, Austria
Renwick JAA, Chew FS (1994) Oviposition behavior in lepidoptera annual. Rev Entomol 39:377–400. https://doi.org/10.1146/annurev.en.39.010194.002113
Rieske L, Dillaway D (2008) Response of two oak species to extensive defoliation: tree growth and vigor, phytochemistry, and herbivore suitability. Forest Ecol Manag 256:121–128
Robert CAM et al (2013) Genetically engineered maize plants reveal distinct costs and benefits of constitutive volatile emissions in the field. Plant Biotechnol J 11:628–639
Rosenkranz M, Chen Y, Zhu P, Vlot AC (2021) Volatile terpenes – mediators of plant-to-plant communication. Plant J 108:617–631. https://doi.org/10.1111/tpj.15453
Roslin T, Gripenberg S, Salminen JP, Karonen M, O’Hara B, Pihlaja R, Pulkkinen K P (2006) Seeing the trees for the leaves–oaks as mosaics for a host-specific moth. Oikos 113:106–120
Salminen JP, Karonen M (2011) Chemical ecology of tannins and other phenolics: we need a change in approach. Funct Ecol 25:325–338. https://doi.org/10.1111/j.1365-2435.2010.01826.x
Šamec D, Karalija E, Šola I, Vujčić Bok V, Salopek-Sondi B (2021) The role of polyphenols in abiotic stress response: the influence of molecular structure. Plants 10:118
Semiz G, Blande JD, Heijari J, Işık K, Niinemets Ü, Holopainen JK (2012) Manipulation of VOC emissions with methyl jasmonate and carrageenan in the evergreen conifer Pinus sylvestris and evergreen broadleaf Quercus ilex. Plant Biol 14:57–65. https://doi.org/10.1111/j.1438-8677.2011.00485.x
Simard SW (2018) Mycorrhizal Networks Facilitate Tree Communication, Learning, and memory. In: Baluska F, Gagliano M, Witzany G (eds) Memory and learning in plants. Springer International Publishing, Cham, pp 191–213. https://doi.org/10.1007/978-3-319-75596-0_10
Sokół-Łętowska A, Kucharska AZ, Hodun G, Gołba M (2020) Chemical composition of 21 cultivars of sour cherry (Prunus cerasus) fruit cultivated in Poland. Molecules 25:4587
Soler R, Harvey JA, Bezemer TM (2007) Foraging efficiency of a parasitoid of a leaf herbivore is influenced by root herbivory on neighbouring plants. Funct Ecol 21:969–974
Southwood T (1961) The number of species of insect associated with various trees. J Anim Ecol 1–8
Stamp N (2003) Out of the quagmire of plant defense hypotheses. Q Rev Biol 78:23–55
Šušić N, Milić Komić S, Živanović B, Jelušić A, Marković S, Sedlarevic Zoric A, Veljovic Jovanovic S (2023) Acclimation of pedunculate oak seedlings to different light conditions in the first months after germination. In: 30th International Conference Ecological Truth And Environmental Research–EcoTER’23. University of Belgrade, Technical Faculty in Bor, pp 135–140
Tollrian R, Harvell CD (1999) The ecology and evolution of inducible defenses. Princeton University Press
Tscharntke T, Thiessen S, Dolch R, Boland W (2001) Herbivory, induced resistance, and interplant signal transfer in Alnus glutinosa. Biochem Syst Ecol 29:1025–1047. https://doi.org/10.1016/S0305-1978(01)00048-5
Valdés-Correcher E et al (2021) Search for top‐down and bottom‐up drivers of latitudinal trends in insect herbivory in oak trees in Europe. Global Ecol Biogeogr 30:651–665
Vialatte A et al (2010) Phylogenetic isolation of host trees affects assembly of local Heteroptera communities. Proc Royal Soc B: Biol Sci 277:2227–2236. https://doi.org/10.1098/rspb.2010.0365
Visakorpi K, Riutta T, Malhi Y, Salminen JP, Salinas N, Gripenberg S (2020) Changes in oak (Quercus robur) photosynthesis after winter moth (Operophtera brumata) herbivory are not explained by changes in chemical or structural leaf traits. PLoS ONE 15:e0228157. https://doi.org/10.1371/journal.pone.0228157
Volf M et al (2021) Branch-localized induction promotes efficacy of volatile defences and herbivore predation in trees. J Chem Ecol 47:99–111
Volf M et al (2022) A mosaic of induced and non-induced branches promotes variation in leaf traits, predation and insect herbivore assemblages in canopy trees. Ecol Lett 25: 729–739
Wäckers FL, van Rijn PCJ, Bruin J (2005) Plant-provided food for carnivorous insects: a protective mutualism and its applications. Cambridge University Press
Walter DE (1996) Living on leaves: mites, tomenta, and leaf domatia. Annu Rev Entomoll 41:101–114
War AR, Hussain B, Sharma HC (2013) Induced resistance in groundnut by jasmonic acid and salicylic acid through alteration of trichome density and oviposition by Helicoverpa armigera (Lepidoptera: Noctuidae). AoB Plants 5:plt053
Yguel B et al (2011) Phytophagy on phylogenetically isolated trees: why hosts should escape their relatives. Ecol Lett 14:1117–1124. https://doi.org/10.1111/j.1461-0248.2011.01680.x
Zhang Z-Z, Che X-N, Pan Q-H, Li X-X, Duan C-Q (2013) Transcriptional activation of flavan-3-ols biosynthesis in grape berries by UV irradiation depending on developmental stage. Plant Sci 208:64–74
Acknowledgements
Fieldwork was carried out with the kind permission of the Forestry Experimental Station in Murowana Goślina (Poznań University of Life Sciences). We are grateful to Fatmanur Selvi for her help in the field. C: N analysis was performed at the Institute of Dendrology of the Polish Academy of Sciences in Kórnik.
Funding
The research was funded by grant No. 2018/29/B/NZ8/00112 to Freerk Molleman from the National Science Centre (NCN, Poland), and Martin Volf acknowledges the Grant Agency of the Czech Republic (grant number 19-28126X).
Author information
Authors and Affiliations
Contributions
Freerk Molleman developed the initial concept of the study, led the fieldwork, analysed the data and wrote the first draft of the manuscript, Manidip Mandal participated in the planning and fieldwork, performed induction, collected the volatile samples, and participated in the VOC analysis, Anna Sokół-Łętowska performed phenolics analyses, Urszula Walczak participated in developing the concept, planning of the study, and in fieldwork, Martin Volf participated in developing the concepts and planning of the study, Soumen Mallick participated in fieldwork and collection of volatile samples, Martin Moos and Petr Vodrážka advised on volatile sample collection and performed volatile measurements, Andreas Prinzing participated in developing the concept, Priscila Mezzomo participated in developing the concept, provided training in VOC sample collection and interpretation of VOC measurements. All authors contributed to the manuscript writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Molleman, F., Mandal, M., Sokół-Łętowska, A. et al. Simulated Herbivory Affects the Volatile Emissions of Oak Saplings, while Neighbourhood Affects Flavan-3-ols Content of Their Leaves. J Chem Ecol (2024). https://doi.org/10.1007/s10886-024-01471-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10886-024-01471-4