Abstract
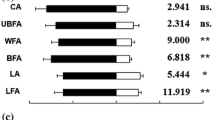
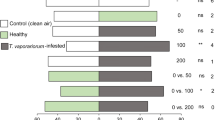
Kairomones are semiochemicals that are emitted by an organism and which mediate interspecific interaction that is of benefit to an organism of another species that receives these chemical substances. Parasitoids find and recognize their hosts through eavesdropping on the kairomones emitted from the by-products or the body of the host. Hemipteran insect pests feed on plant sap and excrete the digested plant materials as honeydew. Honeydew serves as a nutritional food source for parasitoids and a medium for micro-organisms whose activity induces the release of volatiles exploited by parasitoids for host location. The parasitoid Encarsia formosa preferentially parasitizes its host, the greenhouse whitefly, Trialeurodes vaporariorum, on tomato Solanum lycopersicum, but little is known about the chemicals that mediate these interactions. We investigated the olfactory responses of the parasitoid E. formosa to odours from honeydew and nymphs of T. vaporariorum in a Y-tube olfactometer. Arrestment behaviour of the parasitoid to honeydew and nymph extracts, as well as to synthetic hydrocarbons, was also observed in Petri-dish bioassays. We found that T. vaporariorum honeydew volatiles attracted the parasitoid E. formosa but odours from the whitefly nymphs did not. We also found that the parasitoid spent more time searching on areas treated with extracts of honeydew and nymphs than on untreated areas. Gas-chromatography-mass spectrometric analysis revealed that the honeydew volatiles contained compounds such as (Z)-3-hexenol, δ-3-carene, 3-octanone, α-phellandrene, methyl salicylate, β-ocimene, β-myrcene, and (E)-β-caryophyllene which are known to be attractive to E. formosa. The cuticular extracts of the nymphs predominantly contained alkanes, alkenes, and esters. Among the alkanes, synthetic nonacosane arrested the parasitoid. Our findings are discussed in relation to how the parasitoid E. formosa uses these chemicals to locate its host, T. vaporariorum.






Similar content being viewed by others

Data Availability
Data generated in the study are available upon request via the corresponding author.
References
Afsheen S, Wang X, Li R, Zhu C-S, Lou Y-G (2008) Differential attraction of parasitoids in relation to specificity of kairomones from herbivores and their by-products. Insect Sci 15:381–397. https://doi.org/10.1111/j.1744-7917.2008.00225.x
Ayelo PM, Pirk CWW, Yusuf AA, Chailleux A, Mohamed SA, Deletre E (2021a) Exploring the kairomone-based foraging behaviour of natural enemies to enhance biological control: A Review. Front Ecol Evol 9:641974. https://doi.org/10.3389/fevo.2021.641974
Ayelo PM, Yusuf AA, Pirk CWW, Chailleux A, Mohamed SA, Deletre E (2021b) The role of Trialeurodes vaporariorum-infested tomato plant volatiles in the attraction of Encarsia formosa (Hymenoptera: Aphelinidae). J Chem Ecol 47(2):192–203. https://doi.org/10.1007/s10886-021-01245-2
Burger JMS, Reijnen TM, van Lenteren JC, Vet LEM (2004) Host feeding in insect parasitoids: why destructively feed upon a host that excretes an alternative? Entomol Exp Appl 112(3):207–215. https://doi.org/10.1111/j.0013-8703.2004.00196.x
Baroni MV, Nores ML, Díaz MDP, Chiabrando GA, Fassano JP, Costa C, Wunderlin DA (2006) Determination of volatile organic compound patterns characteristic of five unifloral honey by solid-phase microextraction-gas chromatography-mass spectrometry coupled to chemometrics. J Agric Food Chem 54(19):7235–7241. https://doi.org/10.1021/jf061080e
Bénédet F, Leroy T, Gauthier N, Thibaudeau C, Thibout E, Renault S (2002) Gustatory sensilla sensitive to protein kairomones trigger host acceptance by an endoparasitoid. Proc R Soc B: Biol Sci 269:1879–1886. https://doi.org/10.1098/rspb.2002.2077
Birkett MA, Chamberlaln K, Guerrieri E, Pickett JA, Wadhams LJ, Yasuda T (2003) Volatiles from whitefly-infested plants elicit a host-locating response in the parasitoid, Encarsia formosa. J Chem Ecol 29:1589–1600. https://doi.org/10.1023/A:1024218729423
Bleeker MAK, Smid HM, van Aelst AC, van Loon JJA, Vet LEM (2004) Antennal sensilla of two parasitoid wasps: a comparative scanning electron microscopy study. Microsc Res Tech 63:266–273. https://doi.org/10.1002/jemt.20038
Buckner JS., Hagen MM, Nelson DR (1999) The composition of the cuticular lipids from nymphs and exuviae of the silverleaf whitefly, Bemisia argentifolii. Comp Bioch Physiol - Part B 124(2):201–207. 10.1016/S0305-0491(99)00117-0.
Buckner JS, Jones WA (2005) Transfer of methyl-branched hydrocarbons from the parasitoid, Eretmocerus mundus, to silverleaf whitefly nymphs during oviposition. Comp Bioch Physiol 140(1):59–65. https://doi.org/10.1016/j.cbpb.2004.11.001
Buckner JS, Nelson DR, Mardaus MC (1994) The composition of external lipids from adult whiteflies, Bemisia tabaci and Trialeurodes vaporariorum. Insect Biochem Mol Biol 24(10):977–987. https://doi.org/10.1016/0305-0491(94)90013-2
Budenberg WJ (1990) Honeydew as a contact kairomone for aphid parasitoids. Entomol Exp Appl 55(2):139–148. https://doi.org/10.1111/j.1570-7458.1990.tb01357.x
Budenberg WJ, Powell W (1992) The role of honeydew as an ovipositional stimulant for two species of syrphids. Entomol Exp Appl 64:57–61. https://doi.org/10.1111/j.1570-7458.1992.tb01594.x
Chen C-S, Zhao C, Wu Z-Y, Liu GF, Yu X-P, Zhang P-J (2021) Whitefly-induced tomato volatiles mediate host habitat location of the parasitic wasp Encarsia formosa, and enhance its efficacy as a bio-control agent. Pest Manag Sci 77(2):749–757. https://doi.org/10.1002/ps.6071
Darshanee HLC, Ren H, Ahmed N, Zhang Z-F, Liu Y-H, Liu T-X (2017) Volatile-mediated attraction of greenhouse whitefly Trialeurodes vaporariorum to tomato and eggplant. Front Plant Sci 8:1285. https://doi.org/10.3389/fpls.2017.01285
De Vis RMJ, van Lenteren JC (2008) Biological control of Trialeurodes vaporariorum by Encarsia formosa on tomato in unheated greenhouses in the high altitude tropics. Bull Insect 61:43–57.
Dicke M, Sabelis MW (1988) Infochemical terminology: based on cost-benefit analysis rather than origin of compounds? Funct Ecol 2:131–139. https://doi.org/10.2307/2389687
Du Y, Poppy GM, Powell W, Pickett JA, Wadhams LJ, Woodcock CM (1998) Identification of semiochemicals released during aphid feeding that attract parasitoid Aphidius ervi. J Chem Ecol 24:1355–1368. 10.1023/A.
El-Sayed AM, Heppelthwaite VJ, Manning LM, Gibb AR, Suckling DM (2005) Volatile constituents of fermented sugar baits and their attraction to lepidopteran species. J Agric Food Chem 53(4):953–958. https://doi.org/10.1021/jf048521j
Fand BB, Amala U, Yadav DS, Rathi G, Mhaske SH, Upadhyay A, Ahammed-Shabeer TP, Kumbhar DR (2020) Bacterial volatiles from mealybug honeydew exhibit kairomonal activity toward solitary endoparasitoid Anagyrus dactylopii. J Pest Sci 93(1):195–206. https://doi.org/10.1007/s10340-019-01150-4
Fatouros NE, Dicke M, Mumm R, Meiners T, Hilker M (2008) Foraging behavior of egg parasitoids exploiting chemical information. Beh Ecol 19(3):677–689. https://doi.org/10.1093/beheco/arn011
Gamarra H, Carhuapoma P, Mujica M, Kreuze J, Kroschel J (2016) Greenhouse Whitefly, Trialeurodes vaporariorum (Westwood 1956). In: Kroschel J, Mujica N, Carhuapoma P, Sporleder M (eds) Pest Distribution and Risk Atlas for Africa: Potential, Global and Regional Distribution and Abundance of Agricultural and Horticultural Pests and Associated Biocontrol Agents under Current and Future climates. Lima, Peru: International Potato Center (CIP). ISBN 978-92-9060-476-1. pp 154–168. 10.4160/9789290604761.
Gross HR, Lewis WJ, Jones RL, Nordlund DA (1975) Kairomones and their use for management of entomophagous insects: III. stimulation of Trichogramma achaeae, T. pretiosum, and Microplitis croceipes with host-seeking stimuli at time of release to improve their efficiency. J Chem Ecol 1(4):431–438. https://doi.org/10.1007/BF00988584
Gusmão MR, Picanço MC, Guedes RNC, Galvan TL, Pereira EJG (2006) Economic injury level and sequential sampling plan for Bemisia tabaci in outdoor tomato. J Appl Entomol 130(3):160–166. https://doi.org/10.1111/J.1439-0418.2005.01032.X
Hanssen IM, Lapidot M (2012) Major tomato viruses in the mediterranean basin. Adv Virus Res 84:31–66. https://doi.org/10.1016/B978-0-12-394314-9.00002-6
Henneberry TJ, Forlow-Jech L, de la Torre T (2002) Maurer J (2003) Sweetpotato whitefly oviposition and honeydew production following colonization and feeding on different host plants. Arthropod Manag Tests 28(1):8–9. https://doi.org/10.1093/amt/28.1.M8
Hirose Y, Mitsunaga T, Yano E, Goto C (2009) Effects of sugars on the longevity of adult females of Eretmocerus eremicus and Encarsia formosa (Hymenoptera: Aphelinidae), parasitoids of Bemisia tabaci and Trialeurodes vaporariorum (Hemiptera: Alyerodidae), as related to their honeydew feeding and host. Appl Entomol Zool 44(1):175–181. https://doi.org/10.1303/aez.2009.175
Hoddle MS, Van Driesche RG, Sanderson JP (1998) Biology and use of the whitefly parasitoid Encarsia formosa. Annu Rev Entomol 43:645–669. https://doi.org/10.1146/annurev.ento.43.1.645
Hu JS, Gelman DB, Blackburn MB (2002) Growth and development of Encarsia formosa (Hymenoptera: Aphelinidae) in the greenhouse whitefly, Trialeurodes vaporariorum (Homoptera: Aleyrodidae): effect of host age. Arch Insect Biochem Physiol 49:125–136. https://doi.org/10.1002/arch.10015
Hung KY, McElfresh JS, Zou Y, Wayadande A, Gerry AC (2019) Identification of volatiles from plants infested with honeydew-producing insects, and attraction of house flies (Diptera: Muscidae) to these volatiles. J Med Entomol 57(3):667–676. https://doi.org/10.1093/jme/tjz232
Iacovone A, French AS, Tellier F, Cusumano A, Clément G, Gaertner C, Conti E, Salerno G, Marion-Poll F (2016) The role of contact chemoreception in the host location process of an egg parasitoid. J Insect Physiol 91–92:63–75. https://doi.org/10.1016/j.jinsphys.2016.07.001
Inbar M, Gerling D (2008) Plant-mediated interactions between whiteflies, herbivores, and natural enemies. Annu Rev Entomol 53:431–448. https://doi.org/10.1146/annurev.ento.53.032107.122456
Kaiser L, Ode P, van Nouhuys S, Calatayud P-A, Colazza S, Cortesero AM, Thiel A, van Baaren J (2017) The plant as a habitat for entomophagous insects. In: Sauvion N, Calatayud P-A, and Thiéry D (eds) Insect-Plant Interactions in a Crop Protection Perspective. Advances in Botanical Research. Elsevier, Londres pp 179–223.. https://doi.org/10.1016/bs.abr.2016.09.006.
Kapantaidaki DE, Sadikoglou E, Tsakireli D, Kampanis V, Stavrakaki M, Schorn C, Ilias A et al (2018) Insecticide resistance in Trialeurodes vaporariorum populations and novel diagnostics for Kdr mutations. Pest Manag Sci 74(1):59–69. https://doi.org/10.1002/ps.4674
Kishinevsky M, Cohen N, Chiel E, Wajnberg E, Keasar T (2018) Sugar feeding of parasitoids in an agroecosystem: effects of community composition, habitat and vegetation. Insect Conserv Divers 11(1):50–57. https://doi.org/10.1111/icad.12259
Kost C (2008) Chemical Communication. In: Jorgensen SE, Fath BD (eds) Encyclopedia of Ecology. Elsevier, Oxford, pp 557–575 http://hdl.handle.net/11858/00-001M-0000-0012-A137-1
Leroy PD, Heuskin S, Sabri A, Verheggen FJ, Farmakidis J, Lognay G, Thonart P, Wathelet JP, Brostaux Y, Haubruge E (2012) Honeydew volatile emission acts as a kairomonal message for the Asian lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). Insect Sci 19(4):498–506. https://doi.org/10.1111/j.1744-7917.2011.01467.x
Lewis WJ, Jones RL, Nordlund DA, Sparks AN (1975) Kairomones and their use for management of entomophagous insects: I. evaluation for increasing rates of parasitization by Trichogramma spp. in the field. J Chem Ecol 1(3):343–347. https://doi.org/10.1007/BF00988836
Liu T-X, Stansly PA, Gerling D (2015) Whitefly parasitoids: distribution, life history, bionomics, and utilization. Annu Rev Entomol 60(15):1–15. https://doi.org/10.1146/annurev-ento-010814-021101
Liu W-X, Yang Y, Wan F-H, Jin D-C (2007) Comparative analysis of carbohydrates, amino acids and volatile components of honeydew produced by two whiteflies Bemisia tabaci B-biotype and Trialeurodes vaporariorum (Homoptera: Aleyrodidae) feeding cabbage and cucumber. Sheng Tai Xue Bao 50:850–857
López YIA, Martínez-Gallardo NA, Ramírez-Romero R, López MG, Sánchez-Hernández C, Délano-Frier JP (2012) Cross-kingdom effects of plant-plant signaling via volatile organic compounds emitted by tomato (Solanum lycopersicum) plants infested by the greenhouse whitefly (Trialeurodes vaporariorum). J Chem Ecol 38(11):1376–1386. https://doi.org/10.1007/s10886-012-0201-z
Machado AM, Miguel MG, Vilas-Boas M, Figueiredo AC (2020) Honey volatiles as a fingerprint for botanical origin—a review on their occurrence on monofloral honeys. Molecules 25(2):374. https://doi.org/10.3390/molecules25020374
Mandour NS, Ren SX, Qiu BL (2007) Effect of Bemisia tabaci honeydew and its carbohydrates on search time and parasitization of Encarsia bimaculata. J Appl Entomol 131:645–651. https://doi.org/10.1111/j.1439-0418.2007.01165.x
Mandour NS, Shunxiang R, Qiu B, Wackers FL (2005) Arrestment response of Eretmocerus species (Hymenoptera : Aphelinidae) near Furuhashii to honeydew of Bemisia tabaci (Homoptera : Aleyrodidae) and its component carbohydrayes. Proceedings of the Sixth Arabian Conference for Horticulture, Ismailia, Egypt, pp:311–319.
Mehrnejad MR, Copland MJW (2006) Behavioral responses of the parasitoid Psyllaephagus pistaciae (Hymenoptera : Encyrtidae) to host plant volatiles and honeydew. Entomol Sci 9(1):31–37. https://doi.org/10.1111/j.1479-8298.2006.00151.x
Morawo T, Fadamiro H (2016) Identification of key plant-associated volatiles emitted by Heliothis virescens larvae that attract the parasitoid, Microplitis croceipes: Implications for parasitoid perception of odor blends. J Chem Ecol 42(11):1112–1121. https://doi.org/10.1007/s10886-016-0779-7
Moreno C, Quiñones JR, Jiménez P (2016) Phenological growth stages of Solanum sessiliflorum according to BBCH scale. Ann Appl Biol 168(1):151–157. https://doi.org/10.1111/AAB.12251
Navas-Castillo J, López-Moya JJ, Aranda MA (2014) Whitefly-transmitted RNA viruses that affect intensive vegetable production. Ann Appl Biol 165(2):155–171. https://doi.org/10.1111/aab.12147
Pringle EG, Novo A, Ableson I, Barbehenn RV, Vannette RL (2014) Plant-derived differences in the composition of aphid honeydew and their effects on colonies of aphid-tending ants. Ecol Evol 4(21):4065–4079. https://doi.org/10.1002/ece3.1277
Qiu YT, van Lenteren JC, Drost YC, Posthuma-Doodeman CJAM (2004) Life-history parameters of Encarsia formosa, Eretmocerus eremicus and E. mundus, aphelinid parasitoids of bemisia argentifolii (Hemiptera: Aleyrodidae). Eur J Entomol 101:83–94
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org
Ramachandran R, Norris DM, Phillips JK, Phillips TW (1991) Volatiles mediating plant–herbivore–natural enemy interactions: soybean looper frass volatiles, 3-octanone and guaiacol, as kairomones for the parasitoid Microplitis demolitor. J Agric Food Chem 39(12):2310–2317. https://doi.org/10.1021/jf00012a044
Rand TA, Waters DK (2020) Aphid honeydew enhances parasitoid longevity to the same extent as a high-quality floral resource: implications for conservation biological control of the wheat stem sawfly (Hymenoptera: Cephidae). J Econ Entomol 113(4):2022–2025. https://doi.org/10.1093/jee/toaa076
Rodriguez-Saona C, Stelinski LL (2009) Behavior-modifying strategies in IPM: theory and practice. In: Peshin AKDR (ed) Integrated Pest Management: Innovation and Development. Springer, New York, pp 261–312. https://doi.org/10.1007/978-1-4020-8992-3_11
Romeis J, Zebitz CPW (1997) Searching behaviour of Encarsia formosa as mediated by colour and honeydew. Entomol Exp Appl 82:299–309. https://doi.org/10.1046/j.1570-7458.1997.00144.x
Roopa HK, Asokan R, Krishna-Kumar NK, Mahmood R, Bakthavatsalam N, Uprithi K, Raghavendra A (2016) Estimation of sugars and volatiles in the honeydew of Bemisia tabaci genetic groups Meam-I and Asia-I. J Biosci Biotechnol Discv 1:66–73. https://doi.org/10.31248/jbbd2016.015
Shonouda ML (1999) Aphid aqueous-extract as a source of host searching kairomones for the aphidophagous predator Coccinella septempunctata L. (Col., Coccinellidae). J Pest Sci 72:126–128
Tena A, Wackers FL, Heimpel GE, Urbaneja A, Pekas A (2016) Parasitoid nutritional ecology in a community context: the importance of honeydew and implications for biological control. Cur Opin Insect Sci 14:100–104. https://doi.org/10.1016/j.cois.2016.02.008
Thompson DA, Lehmler HJ, Kolpin DW, Hladik ML, Vargo JD, Schilling KE, Lefevre GH et al (2020) A critical review on the potential impacts of neonicotinoid insecticide use: current knowledge of environmental fate, toxicity, and implications for human health. Environ Sci: Processes Impacts 22(6):1315–1346. https://doi.org/10.1039/c9em00586b
Udayagiri S, Bigelow R (2000) Potential for biological control of the greenhouse whitefly in strawberry field by Encarsia formosa. In California conference on biological control II, the historic mission inn Riverside, California, USA, ed. by Hoddle MS, California, Center for Biological Control, College of Natural Resources, University of California. pp: 191–194.
Vandoorn A, De-Vries M, Kant MR, Schuurink RC (2015) Whiteflies glycosylate salicylic acid and secrete the conjugate via their honeydew. J Chem Ecol 41(1):52–58. https://doi.org/10.1007/s10886-014-0543-9
Vet LEM, Dicke M (1992) Ecology of infochemical use by natural enemies in a tritrophic context. Annu Rev Entomol 37:141–172. https://doi.org/10.1146/annurev.en.37.010192.001041
Vet LE, Wackers FL, Dicke M (1991) How to hunt for hiding hosts: the reliability-detectability problem in foraging parasitoids. Neth J Zool 41(2):202–213. https://doi.org/10.1163/156854291X00144
van Roermund HJW, van Lenteren JC (1995) Residence times of the whitefly parasitoid Encarsia formasa Gahan (Hym., Aphelinidae) on tomato leaflets. Entomol Exp Appl 119:465–471. https://doi.org/10.1111/j.1439-0418.1995.tb01319.x
Wang Z, Dai P, Yang X, Ruan CC, Biondi A, Desneux N, Zang LS (2019) Selectivity of novel and traditional insecticides used for management of whiteflies on the parasitoid Encarsia formosa. Pest Manag Sci 75(10):2716–2124. https://doi.org/10.1002/ps.5380
Wickremasinghe MGV, van Emden HF (1992) Reactions of adult female parasitoids, particularly Aphidius rhopalosiphi, to volatile chemical cues from the host plants of their aphid prey. Physiol Entomol 17(3):297–304. https://doi.org/10.1111/j.1365-3032.1992.tb01025.x
Wool D, Hendrix DL, Shukry O (2006) Seasonal variation in honeydew sugar content of galling aphids (Aphidoidea: Pemphigidae: Fordinae) feeding on Pistacia: host ecology and aphid physiology. Basic Appl Ecol 7(2):141–151. https://doi.org/10.1016/j.baae.2005.02.009
Zhang Q-Q, Chen H-Y, Qin Y-C (2011) Damage from Bemisia tataci affects its host plant’s volatiles and the parasitism of Encarsia formosa. Chinese. J Biol Control:105–112
Zhu J, Cossé AA, Obrycki JJ, Boo KS, Baker TC (1999) Olfactory reactions of the twelve-spotted lady beetle, Coleomegilla maculata and the green lacewing, Chrysoperla carnea to semiochemicals released from their prey and host plant: electroantennogram and behavioral responses. J Chem Ecol 25(5):1163–1177. https://doi.org/10.1023/A:1020846212465
Acknowledgments
The authors acknowledge the financial support provided by the French National Research Agency (ANR) through CIRAD (Award no. ANR-16-CE32-0010-01); the National Research Foundation through the NRF grants of A.A.Y (IFRR Grant no. 109380 and Y-rated Researchers Grant no. RDYR180504326262) and C.W.W.P (Grant no. CPRR160502163617); and the bursary offered to P.M.A. by the University of Pretoria. We also gratefully acknowledge the following icipe core funding donors: the UK’s Foreign, Commonwealth & Development Office (FCDO); the Swedish International Development Cooperation Agency (SIDA); the Swiss Agency for Development and Cooperation (SDC); the Federal Democratic Republic of Ethiopia; and the Kenyan Government. P.M.A. was supported by the University of Pretoria and the Deutscher Akademischer Austauschdienst (DAAD) In-Region Postgraduate Scholarship (Personal Grant no. 91672680). The views expressed herein do not necessarily reflect the official opinion of the donors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the research work. P.M.A. conducted the experiments, analysed the data, and wrote the first draft of the manuscript. All authors proofread the manuscript and approved the final version for submission.
Corresponding authors
Ethics declarations
Conflict of interest statement
The authors state that they have no conflict of interests to declare.
Ethical approval
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 875 kb)
Rights and permissions
About this article
Cite this article
Ayelo, P.M., Yusuf, A.A., Chailleux, A. et al. Chemical Cues From Honeydew and Cuticular Extracts of Trialeurodes Vaporariorum Serve as Kairomones for The Parasitoid Encarsia Formosa. J Chem Ecol 48, 370–383 (2022). https://doi.org/10.1007/s10886-022-01354-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-022-01354-6