Abstract
The cuticle of all insects is covered with hydrocarbons which have multiple functions. Cuticular hydrocarbons (CHCs) basically serve to protect insects against environmental harm and reduce dehydration. In many species, some CHCs also act as pheromones. CHCs have been intensively studied in Drosophila species and more especially in D. melanogaster. In this species, flies produce about 40 CHCs forming a complex sex- and species-specific bouquet. The quantitative and qualitative pattern of the CHC bouquet was characterized during the first days of adult life but remains unexplored in aging flies. Here, we characterized CHCs during the whole—or a large period of—adult life in males and females of several wild type and transgenic lines. Both types of lines included standard and variant CHC profiles. Some of the genotypes tested here showed very dramatic and unexpected aging-related variation based on their early days’ profile. This study provides a concrete dataset to better understand the mechanisms underlying the establishment and maintenance of CHCs on the fly cuticle. It could be useful to determine physiological parameters, including age and response to climate variation, in insects collected in the wild.



Similar content being viewed by others
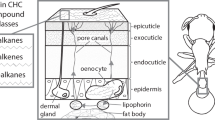
Data Availability
All data will be made available as supplementary material upon publication.
Code Availability
Not applicable.
References
Addinsoft (2021) XLSTAT 2021: data analysis and statistical solution for Microsoft excel. Addinsoft, Paris, France
Antony C, Jallon J-M (1982) The chemical basis for sex recognition in Drosophila melanogaster. J Insect Physiol 28:873–880
Bagnères A-G, Clément J-L, Blum MS, Severson RF, Joulie C, Lange C (1990) Cuticular hydrocarbons and defensive compounds of Reticulitermes flavipes (Kollar) and R. santonensis (Feytaud): polymorphism and chemotaxonomy. J Chem Ecol 16:3213–3244. https://doi.org/10.1007/BF00982094
Bagnères A-G, Darrouzet E, Christidès J-P, Landré X (2011) Endogenous synchronization of the chemical signature of Reticulitermes (Isoptera, Rhinotermitidae) worker termites. Ann Soc Entomol France 47:202–208. https://doi.org/10.1080/00379271.2011.10697712
Bagnères A-G, Killian A, Clément J-L, Lange C (1991) Interspecific recognition among termites of the genus Reticulitermes: evidence for a role for the cuticular hydrocarbons. J Chem Ecol 17:2397–2420. https://doi.org/10.1007/BF00994590
Bagnères A-G, Lorenzi MC, Dusticier G, Turillazzi S, Clément J-L (1996) Chemical usurpation of a nest by paper wasp parasites. Science 272:889–892. https://doi.org/10.1126/science.272.5263.889
Bagnères A-G, Rivière G, Clément J-L (1998) Artificial neural network modeling of caste odor discrimination based on cuticular hydrocarbons in termites. Chemoecology 8(4):201–209. https://doi.org/10.1007/s000490050026
Beani L, Bagnères A-G, Elia M, Petrocelli I, Cappa F, Lorenzi MC (2019) Cuticular hydrocarbons as cues of sex and health condition in Polistes dominula wasps. Insectes Soc 66:543–553. https://doi.org/10.1007/s00040-019-00721-z
Begun DJ, Aquadro CF (1993) African and north American populations of Drosophila melanogaster are very different at the DNA level. Nature 365:548–550
Benzer S (1967) Behavioral mutants of Drosophila isolated by countercurrent distribution. Proc Natl Acad Sci U S A 58:1112–1119. https://doi.org/10.1073/pnas.58.3.1112
Billeter J-C, Atallah J, Krupp JJ, Millar JG, Levine JD (2009) Specialized cells tag sexual and species identity in Drosophila melanogaster. Nature 461:987–991. https://doi.org/10.1038/nature08495
Blomquist GJ, Bagnères A-G (2010) Insect hydrocarbons: biology, biochemistry, and chemical ecology. Cambridge University Press
Bonavita-Cougourdan A, Rivière G, Provost E, Bagnères A-G, Roux M, Dusticier G, Clément J-L (1996) Selective adaptation of the cuticular hydrocarbon profiles of the slave-making ants Polyergus rufescens Latr. and their Formica rufibarbis Fab. and F. cunicularia Latr. slaves. Comp Biochem Physiol Part B Biochem Mol Biol 113:313–329. https://doi.org/10.1016/0305-0491(95)02029-2
Bousquet F et al (2012) Expression of a desaturase gene, desat1, in neural and nonneural tissues separately affects perception and emission of sex pheromones in Drosophila. Proc Natl Acad Sci U S A 109:249–254. https://doi.org/10.1073/pnas.1109166108
Bridges CB (1916) Non-disjunction as proof of the chromosome theory of heredity. Genetics 1:1–52
Caputo B et al (2005) Identification and composition of cuticular hydrocarbons of the major Afrotropical malaria vector Anopheles gambiae s.s. (Diptera: Culicidae): analysis of sexual dimorphism and age-related changes. J Mass Spectrom 40:1595–1604. https://doi.org/10.1002/jms.961
Carvalho GB et al (2017) The 4E-BP growth pathway regulates the effect of ambient temperature on Drosophila metabolism and lifespan. Proc Natl Acad Sci U S A 114:9737–9742. https://doi.org/10.1073/pnas.1618994114
Chertemps T, Duportets L, Labeur C, Ueda R, Takahashi K, Saigo K, Wicker-Thomas C (2007) A female-biased expressed elongase involved in long-chain hydrocarbon biosynthesis and courtship behavior in Drosophila melanogaster. Proc Natl Acad Sci 104:4273–4278
Chertemps T, Duportets L, Labeur C, Wicker-Thomas C (2005) A new elongase selectively expressed in Drosophila male reproductive system. Biochem Biophys Res Commun 333:1066–1072
Clark RI, Walker DW (2018) Role of gut microbiota in aging-related health decline: insights from invertebrate models. Cellular and Molecular Life Sciences: CMLS 75:93–101. https://doi.org/10.1007/s00018-017-2671-1
Cortot J, Farine J-P, Houot B, Everaerts C, Ferveur J-F (2019) Experimental introgression to evaluate the impact of sex specific traits on Drosophila melanogaster incipient speciation. G3: Genes|Genomes|Genetics 9:2561. https://doi.org/10.1534/g3.119.400385
Coyne JA (1996) Genetics of a difference in male cuticular hydrocarbons between two sibling species, Drosophila simulans and D sechellia. Genetics 143:1689–1698
Coyne JA, Crittenden AP, Mah K (1994) Genetics of a pheromonal difference contributing to reproductive isolation in Drosophila. Science 265:1461–1464
Coyne JA, Oyama R (1995) Localization of pheromonal sexual dimorphism in Drosophila melanogaster and its effect on sexual isolation. Proc Natl Acad Sci U S A 92:9505–9509. https://doi.org/10.1073/pnas.92.21.9505
Coyne JA, Wicker-Thomas C, Jallon J-M (1999) A gene responsible for a cuticular hydrocarbon polymorphism in Drosophila melanogaster. Genet Res 73:189–203
Crimm PD, Short DM (1934) Age variations in the polymorphonuclear leucocyte of the rat. American Journal of Physiology-Legacy Content 108:324–330. https://doi.org/10.1152/ajplegacy.1934.108.2.324
Curcillo PG, Tompkins L (1987) The ontogeny of sex appeal in Drosophila melanogaster males behavior. Genetics 17:81–86
Dallerac R, Labeur C, Jallon J-M, Knippie DC, Roelofs WL, Wicker-Thomas C (2000) A Delta 9 desaturase gene with a different substrate specificity is responsible for the cuticular diene hydrocarbon polymorphism in Drosophila melanogaster. Proc Natl Acad Sci U S A 97:9449–9454
Darrouzet E, Labédan M, Landré X, Perdereau E, Christidès J-P, Bagnères A-G (2014) Endocrine control of cuticular hydrocarbon profiles during worker-to-soldier differentiation in the termite Reticulitermes flavipes. J Insect Physiol 61:25–33. https://doi.org/10.1016/j.jinsphys.2013.12.006
Dembeck LM, Bööczky K, Huang W, Schal C, Anholt RHR, Mackay TFC (2015) Genetic architecture of natural variation in cuticular hydrocarbon composition in Drosophila melanogaster. eLife 4. https://doi.org/10.7554/eLife.09861
Diehl PA (1973) Paraffin synthesis in the oenocytes of the desert locust. Nature 243:468–470. https://doi.org/10.1038/243468a0
Dronnet S, Lohou C, Christidès J-P, Bagnères A-G (2006) Cuticular hydrocarbon composition reflects genetic relationship among colonies of the introduced termite Reticulitermes santonensis Feytaud. J Chem Ecol 32:1027–1042. https://doi.org/10.1007/s10886-006-9043-x
Etges WJ, De Oliveira CC, Ritchie MG, Noor MA (2009) Genetics of incipient speciation in Drosophila mojavensis: II. Host plants and mating status influence cuticular hydrocarbon QTL expression and G×E interactions. Evolution 63:1712–1730. https://doi.org/10.1111/j.1558-5646.2009.00661.x
Evans JJT (1967) Development and ultrastructure of the fat body cells and oenocytes of the Queensland fruit fly, Dacus tryoni (Frogg.). Z Zellforsch Mikrosk Anat 81:49–61. https://doi.org/10.1007/BF00344551
Everaerts C, Farine J-P, Cobb M, Ferveur J-F (2010) Drosophila cuticular hydrocarbons revisited: mating status alters cuticular profiles. PLoS One 5:e9607. https://doi.org/10.1371/journal.pone.0009607
Fan Y, Schal C, Vargo EL, Bagnères A-G (2004) Characterization of termite lipophorin and its involvement in hydrocarbon transport. J Insect Physiol 50:609–620. https://doi.org/10.1016/j.jinsphys.2004.04.007
Ferveur J-F, Cortot J, Rihani K, Cobb M, Everaerts C (2018) Desiccation resistance: effect of cuticular hydrocarbons and water content in Drosophila melanogaster adults. PeerJ 6:e4318–e4318. https://doi.org/10.7717/peerj.4318
Ferveur J-F (1991) Genetic-control of pheromones in Drosophila simulans .1. Ngbo, a locus on the 2nd chromosome. Genetics 128:293–301
Ferveur J-F (2005) Cuticular hydrocarbons: their evolution and roles in Drosophila pheromonal communication. Behav Genet 35:279–295
Ferveur J-F, Jallon J-M (1996) Genetic control of male cuticular hydrocarbons in Drosophila melanogaster Genet Res 67:211–218
Ferveur J-F, Savarit F, O'Kane CJ, Sureau G, Greenspan RJ, Jallon J-M (1997) Genetic feminization of pheromones and its behavioral consequences in Drosophila males. Science 276:1555–1558
Ferveur J-F, Stortkuhl KF, Stocker RF, Greenspan RJ (1995) Genetic feminization of brain structures and changed sexual orientation in male Drosophila. Science 267:902–905. https://doi.org/10.1126/science.7846534
Gibbs A, Pomonis JG (1995) Physical properties of insect cuticular hydrocarbons: the effects of chain length, methyl-branching and unsaturation. Comp Biochem Physiol B Biochem Mol Biol 112:243–249
Ginzel MD, Blomquist GJ (2016) Insect hydrocarbons: biochemistry and chemical ecology. In: Cohen E, Moussian B (eds) Extracellular composite matrices in arthropods. Springer, Cham, pp 221–252. https://doi.org/10.1007/978-3-319-40740-1_7
Gosden TP, Chenoweth SF (2011) On the evolution of heightened condition dependence of male sexual displays. J Evol Biol 24:685–692. https://doi.org/10.1111/j.1420-9101.2010.02205.x
Grangeteau C, Yahou F, Everaerts C, Dupont S, Farine J-P, Beney L, Ferveur J-F (2018) Yeast quality in juvenile diet affects Drosophila melanogaster adult life traits. Sci Rep 8:13070. https://doi.org/10.1038/s41598-018-31561-9
Greene MJ, Gordon DM (2003) Cuticular hydrocarbons inform task decisions. Nature 423:32–32. https://doi.org/10.1038/423032a
Greenspan RJ (2001) Opinion - the flexible genome. Nat Rev Genet 2:383–387. https://doi.org/10.1038/35072018
Greenspan RJ (2009) Selection, gene interaction, and flexible gene networks. Cold Spring Harb Symp Quant Biol 74:131–138. https://doi.org/10.1101/sqb.2009.74.029
Grillet M, Everaerts C, Houot B, Ritchie MG, Cobb M, Ferveur J-F (2012) Incipient speciation in Drosophila melanogaster involves chemical signals. Sci Rep 2:224. https://doi.org/10.1038/srep00224
Grillet M, Ferveur J-F, Everaerts C (2018) Behavioural elements and sensory cues involved in sexual isolation between Drosophila melanogaster strains. Royal Society Open Science 5. https://doi.org/10.1098/rsos.172060
Gutierrez E, Wiggins D, Fielding B, Gould AP (2007) Specialized hepatocyte-like cells regulate Drosophila lipid metabolism. Nature 445:275–280
Hadley NF (1981) Cuticular lipids of terrestrial plants and arthropods: a comparison of their structure, composition, and waterproofing function. Biological Reviews 56:23–47. https://doi.org/10.1111/j.1469-185X.1981.tb00342.x
Horst K, Mendel LB, Benedict FG (1934) The influence of previous dietgrowth and age upon the basal metabolism of the rat. J Nutr 8:139–162. https://doi.org/10.1093/jn/8.2.139
Horváth B, Kalinka AT (2016) Effects of larval crowding on quantitative variation for development time and viability in Drosophila melanogaster. Ecol Evol 6:8460–8473. https://doi.org/10.1002/ece3.2552
Houot B, Bousquet F, Ferveur J-F (2010) The consequences of regulation of desat1 expression for pheromone emission and detection in Drosophila melanogaster. Genetics 185:1297–1309. https://doi.org/10.1534/genetics.110.117226
Houot B, Fraichard S, Greenspan RJ, Ferveur J-F (2012) Genes involved in sex pheromone discrimination in Drosophila melanogaster and their background-dependent effect. PLoS One 7:e30799. https://doi.org/10.1371/journal.pone.0030799
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393
Hur JH, Stork DA, Walker DW (2014) Complex-I-ty in aging. J Bioenerg Biomembr 46:329–335. https://doi.org/10.1007/s10863-014-9553-0
Jallon J-M (1984) A few chemical words exchanged by Drosophila during courtship and mating. Behav Genet 14:441–478
Jallon J-M, Lauge G, Orssaud L, Antony C (1988) Female pheromones in Drosophila melanogaster are controlled by the doublesex locus. Genet Res 51:17–22
Jallon J-M, Pechiné J-M (1989) A novel chemical race of Drosophila melanogaster in Africa. Comptes Rendus de l'Academie des Sciences Serie Ii 309:1551–1556
Johnson MB, Butterworth FM (1985) Maturation and aging of adult fat body and oenocytes in Drosophila as revealed by light microscopic morphometry. J Morphol 184:51–59. https://doi.org/10.1002/jmor.1051840106
Kaib M, Eisermann B, Schoeters E, Billen J, Franke S, Francke W (2000) Task-related variation of postpharyngeal and cuticular hydrocarbon compositions in the ant Myrmicaria eumenoides. J Comp Physiol A 186(10):939–948. https://doi.org/10.1007/s003590000146
Klepsatel P, Procházka E, Gáliková M (2018) Crowding of Drosophila larvae affects lifespan and other life-history traits via reduced availability of dietary yeast. Exp Gerontol 110:298–308. https://doi.org/10.1016/j.exger.2018.06.016
Krupp JJ et al (2008) Social experience modifies pheromone expression and mating behavior in male Drosophila melanogaster. Curr Biol 18:1373–1383. https://doi.org/10.1016/j.cub.2008.07.089
Kühbandner S, Sperling S, Mori K, Ruther J (2012) Deciphering the signature of cuticular lipids with contact sex pheromone function in a parasitic wasp. J Exp Biol 215:2471–2478. https://doi.org/10.1242/jeb.071217
Kuo TH, Yew JY, Fedina TY, Dreisewerd K, Dierick HA, Pletcher SD (2012) Aging modulates cuticular hydrocarbons and sexual attractiveness in Drosophila melanogaster. J Exp Biol 215:814–821. https://doi.org/10.1242/jeb.064980
Lamb MJ (1968) Temperature and lifespan in Drosophila. Nature 220:808–809. https://doi.org/10.1038/220808a0
Láruson ÁJ, Reed FA (2016) Stability of underdominant genetic polymorphisms in population networks. J Theor Biol 390:156–163. https://doi.org/10.1016/j.jtbi.2015.11.023
Lazareva AA, Roman G, Mattox W, Hardin PE, Dauwalder B (2007) A role for the adult fat body in Drosophila male courtship behavior. PLoS Genet 3:e16–e16. https://doi.org/10.1371/journal.pgen.0030016
Le Conte Y, Hefetz A (2008) Primer pheromones in social hymenoptera. Annu Rev Entomol:523–542. https://doi.org/10.1146/annurev.ento.52.110405.091434
Le Conte Y, Huang ZY, Roux M, Zeng ZJ, Christidès J-P, Bagnères A-G (2015) Varroa destructor changes cuticular hydrocarbons to mimic its new host. Biol Lett 11:20150233. https://doi.org/10.1098/rsbl.2015.0233
Lebreton S, Chevrier C, Darrouzet E (2010) Sex allocation strategies in response to conspecifics’ offspring sex ratio in solitary parasitoids. Behav Ecol 21:107–112. https://doi.org/10.1093/beheco/arp156
Lebreton S, Labarussias M, Chevrier C, Darrouzet E (2009) Discrimination of the age of conspecific eggs by an ovipositing ectoparasitic wasp. Entomol Exp Appl 130:28–34. https://doi.org/10.1111/j.1570-7458.2008.00790.x
Lee KP et al (2008) Lifespan and reproduction in Drosophila: new insights from nutritional geometry. Proc Natl Acad Sci U S A 105:2498–2503. https://doi.org/10.1073/pnas.0710787105
Lengyel F, Westerlund SA, Kaib M (2007) Juvenile hormone III influences task-specific cuticular hydrocarbon profile changes in the ant Myrmicaria eumenoides. J Chem Ecol 33:167–181. https://doi.org/10.1007/s10886-006-9185-x
Lenoir A, Malosse C, Yamaoka R (1997) Chemical mimicry between parasitic ants of the genus Formicoxenus and their host Myrmica (Hymenoptera, Formicidae). Biochem Syst Ecol 25:379–389. https://doi.org/10.1016/S0305-1978(97)00025-2
Leonhardt SD, Menzel F, Nehring V, Schmitt T (2016) Ecology and evolution of communication in social insects. Cell 164:1277–1287. https://doi.org/10.1016/j.cell.2016.01.035
Liang D, Silverman J (2000) “You are what you eat”: diet modifies cuticular hydrocarbons and nestmate recognition in the argentine ant, Linepithema humile. Naturwissenschaften 87:412–416. https://doi.org/10.1007/s001140050752
Liebig J, Peeters C, Oldham NJ, Markstadter C, Holldobler B (2000) Are variations in cuticular hydrocarbons of queens and workers a reliable signal of fertility in the ant Harpegnathos saltator? Proc Natl Acad Sci U S A 97:4124–4131
Lindsley DL, Grell EH (1968) Genetic variations of Drosophila melanogaster, vol 627. Carnegie Institution of Washington, Washington, D.C.
Locke M (1965) Permeability of insect cuticle to water and lipids. Science 147:295–298. https://doi.org/10.1126/science.147.3655.295
Lushchak O et al (2018) Larval crowding results in hormesis-like effects on longevity in Drosophila: timing of eclosion as a model. Biogerontology 20:191–201
Mannino MC, Huarte-Bonnet C, Davyt-Colo B, Pedrini N (2019) Is the insect cuticle the only entry gate for fungal infection? Insights into alternative modes of action of entomopathogenic fungi. Journal of Fungi 5. https://doi.org/10.3390/jof5020033
Marcillac F, Bousquet F, Alabouvette J, Savarit F, Ferveur J-F (2005a) A mutation with major effects on Drosophila melanogaster sex pheromones. Genetics 171:1617–1628
Marcillac F, Grosjean Y, Ferveur J-F (2005b) A single mutation alters production and discrimination of Drosophila sex pheromones. Proc R Soc Biol Sci Ser B 272:303–309. https://doi.org/10.1098/rspb.2004.2971
Martins G, Ramalho-Ortigão JM (2012) Oenocytes in insects. ISJ-Invertebrate Survival Journal 9:139–152
McCay CM, Maynard LA, Sperling G, Barnes LL (1939) Retarded growth, life span, ultimate body size and age changes in the albino rat after feeding diets restricted in calories: four figures. J Nutr 18:1–13. https://doi.org/10.1093/jn/18.1.1
Menzel F, Zumbusch M, Feldmeyer B (2018) How ants acclimate: impact of climatic conditions on the cuticular hydrocarbon profile. Funct Ecol 32:657–666. https://doi.org/10.1111/1365-2435.13008
Michalak P, Malone JH, Lee IT, Hoshino D, Ma DN (2007) Gene expression polymorphism in Drosophila populations. Mol Ecol 16:1179–1189
Miller RS, Thomas JL (1958) The effects of larval crowding and body size on the longevity of adult Drosophila melanogaster. Ecology 39:118–125. https://doi.org/10.2307/1929973
Miquel J, Lundgren PR, Bensch KG, Atlan H (1976) Effects of temperature on the life span, vitality and fine structure of Drosophila melanogaster. Mech Ageing Dev 5:347–370. https://doi.org/10.1016/0047-6374(76)90034-8
Mołoń M et al (2020) Effects of temperature on lifespan of Drosophila melanogaster from different genetic backgrounds: links between metabolic rate and longevity. Insects 11:470. https://doi.org/10.3390/insects11080470
Moore HE, Adam CD, Drijfhout FP (2014) Identifying 1st instar larvae for three forensically important blowfly species using “fingerprint” cuticular hydrocarbon analysis. Forensic Sci Int 240:48–53. https://doi.org/10.1016/j.forsciint.2014.04.002
Mpuru S et al (2001) Effect of age and sex on the production of internal and external hydrocarbons and pheromones in the housefly, Musca domestica. Insect Biochem Mol Biol 31:139–155. https://doi.org/10.1016/S0965-1748(00)00098-9
Murgier J, Everaerts C, Farine J-P, Ferveur J-F (2019) Live yeast in juvenile diet induces species-specific effects on Drosophila adult behaviour and fitness. Scientific Reports 9. https://doi.org/10.1038/s41598-019-45140-z
Nunes TM, Turatti ICC, Lopes NP, Zucchi R (2009) Chemical signals in the stingless bee, Frieseomelitta varia, indicate caste, gender, age, and reproductive status. J Chem Ecol 35:1172. https://doi.org/10.1007/s10886-009-9691-8
Parra-Peralbo E, Culi J (2011) Drosophila lipophorin receptors mediate the uptake of neutral lipids in oocytes and imaginal disc cells by an endocytosis-independent mechanism. PLoS Genet 7:e1001297. https://doi.org/10.1371/journal.pgen.1001297
Partridge L, Tower J (2008) Yeast, a feast: the fruit fly Drosophila as a model organism for research into aging. In: Guarente LP, Partridge L, Wallace DC (eds) Molecular biology of aging. Cold Spring Harbor Laboratory Press, NY, pp 267–308
Pechal JL, Moore H, Drijfhout F, Benbow ME (2014) Hydrocarbon profiles throughout adult Calliphoridae aging: a promising tool for forensic entomology. Forensic Sci Int 245:65–71. https://doi.org/10.1016/j.forsciint.2014.10.019
Pho DB, Pennanech M, Jallon J-M (1996) Purification of adult Drosophila melanogaster lipophorin and its role in hydrocarbon transport. Arch Insect Biochem Physiol 31:289–303
Piper MD, Partridge L (2016) Protocols to study aging in Drosophila. Methods Mol Biol 1478. https://doi.org/10.1007/978-1-4939-6371-3_18
Pomonis JG (1989) Cuticular hydrocarbons of the screwworm, Cochliomyia hominivorox (Diptera: Calliphoridae). J Chem Ecol 15:2301–2317. https://doi.org/10.1007/BF01012083
Provost E, Rivière G, Roux M, Morgan ED, Bagnères A-G (1993) Change in the chemical signature of the ant Leptothorax lichtensteini Bondroit with time. Insect Biochem Mol Biol 23:945–957. https://doi.org/10.1016/0965-1748(93)90112-6
Qiu Y et al (2012) An insect-specific P450 oxidative decarbonylase for cuticular hydrocarbon biosynthesis. Proc Natl Acad Sci U S A 109:14858–14863
Rahman S, Hajong SR, Gévar J, Lenoir A, Darrouzet E (2016) Cuticular hydrocarbon compounds in worker castes and their role in nestmate recognition in Apis cerana indica. J Chem Ecol 42:44–451. https://doi.org/10.1007/s10886-016-0700-4
Rivault C, Cloarec A, Sreng L (2002) Are differences in hydrocarbon profiles able to mediate strain recognition in German cockroaches (Dictyoptera: Blattellidae)? EJE 99:437–444
Rose MR, Charlesworth B (1981) Genetics of life history in Drosophila melanogaster. II Exploratory selection experiments . Genetics 97:187–196
Savarit F, Ferveur J-F (2002a) Genetic study of the production of sexually dimorphic cuticular hydrocarbons in relation with the sex-determination gene transformer in Drosophila melanogaster. Genet Res 79:23–40
Savarit F, Ferveur J-F (2002b) Temperature affects the ontogeny of sexually dimorphic cuticular hydrocarbons in Drosophila melanogaster. J Exp Biol 205:3241–3249
Savarit F, Sureau G, Cobb M, Ferveur J-F (1999) Genetic elimination of known pheromones reveals the fundamental chemical bases of mating and isolation in Drosophila. Proc Natl Acad Sci U S A 96:9015–9020
Schal C, Sevala V, Capurro ML, Snyder TE, Blomquist GJ, Bagnères A-G (2001) Tissue distribution and lipophorin transport of hydrocarbons and sex pheromones in the house fly, Musca domestica. J Insect Sci 1:12
Seeholzer LF, Seppo M, Stern DL, Ruta V (2018) Evolution of a central neural circuit underlies Drosophila mate preferences. Nature 559:564–569. https://doi.org/10.1038/s41586-018-0322-9
Sevala V, Bagnères A-G, Kuenzli M, Blomquist GJ, Schal C (2000) Cuticular hydrocarbons of the termite Zootermopsis nevadensis (Hagen): caste differences and role of lipophorin in transport of hydrocarbons and hydrocarbon metabolites. J Chem Ecol 26:765–790. https://doi.org/10.1023/A:1005440624678
Skorupa DA, Dervisefendic A, Zwiener J, Pletcher SD (2008) Dietary composition specifies consumption, obesity, and lifespan in Drosophila melanogaster. Aging Cell 7:478–490. https://doi.org/10.1111/j.1474-9726.2008.00400.x
Smith RK, Taylor OR (1990) Unsaturated extracted hydrocarbon caste differences between European queen and worker honey bees, Apis mellifera L. (Hymenoptera: Apidae). J Kansas Entomol Soc 63:369–374
Steiner S, Hermann N, Ruther J (2006) Characterization of a female-produced courtship pheromone in the parasitoid Nasonia vitripennis. J Chem Ecol 32:1687–1702. https://doi.org/10.1007/s10886-006-9102-3
Stern C, Schaeffer EW (1943) On primary attributes of alleles in Drosophila melanogaster. Proc Natl Acad Sci U S A 29:351–361. https://doi.org/10.1073/pnas.29.11.351
Svetec N, Ferveur J-F (2005) Social experience and pheromonal perception can change male-male interactions in Drosophila melanogaster. J Exp Biol 208:891–898
Tatar M, Post S, Yu K (2014) Nutrient control of Drosophila longevity. Trends Endocrinol Metab 25:509–517. https://doi.org/10.1016/j.tem.2014.02.006
Thomas ML, Simmons LW (2008) Cuticular hydrocarbons are heritable in the cricket Teleogryllus oceanicus. J Evol Biol 21:801–806. https://doi.org/10.1111/j.1420-9101.2008.01514.x
Tompkins L (1984) Genetic analysis of sex appeal in Drosophila. Behav Genet 14:411–440. https://doi.org/10.1007/BF01065443
Tompkins L, McRobert SP (1995) Behavioral and pheromonal phenotypes associated with expression of loss-of-function mutations in the sex-lethal gene of Drosophila melanogaster. J Neurogenet 9:219–226. https://doi.org/10.3109/01677069509084158
Tower J (2015) Mitochondrial maintenance failure in aging and role of sexual dimorphism. Arch Biochem Biophys 576:17–31. https://doi.org/10.1016/j.abb.2014.10.008
Tower J (2017) Sex-specific gene expression and life span regulation trends in endocrinology and metabolism: TEM 28:735-747. https://doi.org/10.1016/j.tem.2017.07.002
Tower J (2019) Drosophila flies in the face of aging. J Gerontol A Biol Sci Med Sci 74:1539–1541. https://doi.org/10.1093/gerona/glz159
van Zweden JS, d’Ettorre P (2010) Nestmate recognition in social insects and the role of hydrocarbons. In: Blomquist GJ, Bagnères A-G (eds) Insect hydrocarbons: biology, biochemistry and chemical ecology. Cambridge University Press, Cambridge, pp 222–243
Vauchot B, Provost E, Bagnères A-G, Clément J-L (1996) Regulation of the chemical signatures of two termite species, Reticulitermes santonensis and Reticulitermes lucifugus grassei, living in mixed experimental colonies. J Insect Physiol 42:309–321. https://doi.org/10.1016/0022-1910(95)00102-6
Wang Y et al (2020) Dysfunction of Oskyddad causes Harlequin-type ichthyosis-like defects in Drosophila melanogaster. PLoS Genet 16:e1008363–e1008363. https://doi.org/10.1371/journal.pgen.1008363
Weaver LN, Ma T, Drummond-Barbosa D (2020) Analysis of Gal4 expression patterns in adult Drosophila females G3: Genes|Genomes|Genetics:g3.401676.402020. https://doi.org/10.1534/g3.120.401676
Wicker-Thomas C, Jallon J-M (2001) Control of female pheromones in Drosophila melanogaster by homeotic genes. Genet Res 78:235–242
Wigglesworth VB (1933) Memoirs: the physiology of the cuticle and of ecdysis in Rhodnius prolixus (Triatomidae, Hemiptera); with special reference to the function of the oenocytes and of the dermal glands. J Cell Sci s2-76:269–318. https://doi.org/10.1242/jcs.s2-76.302.269
Wigglesworth VB (1942) The storage of protein, fat, glycogen and uric acid in the fat body and other tissues of mosquito larvae. J Exp Biol 19:56–77. https://doi.org/10.1242/jeb.19.1.56
Wigglesworth VB (1988) The source of lipids and polyphenols for the insect cuticle: the role of fat body, oenocytes and oenocytoids. Tissue Cell 20:919–932. https://doi.org/10.1016/0040-8166(88)90033-x
Woodcox A (2018) Aristotle’s theory of aging. Cahiers des études anciennes, LV | 2018:65-78
Würf J, Pokorny T, Wittbrodt J, Millar JG, Ruther J (2020) Cuticular hydrocarbons as contact sex pheromone in the parasitoid wasp Urolepis rufipes. Front Ecol Evol 8. https://doi.org/10.3389/fevo.2020.00180
Zhu G-H, Yu X-J, Xie L-X, Luo H, Wang D, Lv J-Y, Xu X-H (2013) Time of death revealed by hydrocarbons of empty puparia of Chrysomya megacephala (Fabricius) (Diptera: Calliphoridae): a field experiment. PLoS One 8:e73043. https://doi.org/10.1371/journal.pone.0073043
Zhu GH, Ye GY, Hu C, Xu XH, Li K (2006) Development changes of cuticular hydrocarbons in Chrysomya rufifacies larvae: potential for determining larval age. Med Vet Entomol 20:438–444. https://doi.org/10.1111/j.1365-2915.2006.00651.x
Ziehm M, Piper MD, Thornton JM (2013) Analysing variation in Drosophila aging across independent experimental studies: a meta-analysis of survival data. Aging Cell 12:917–922. https://doi.org/10.1111/acel.12123
Acknowledgments
We thank Félicien Hua Vanié for his technical help in the preliminary experiments and the two reviewers for their helpful comments.
Funding
This work was financially supported by the Burgundy Regional Council (PARI2012), the CNRS (Insb), and the Burgundy University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest/Competing Interests
The authors have no conflict of interest or competing interest to declare.
Supplementary Information

Supplemental Figure 1.
Age-related variation of cuticular hydrocarbons in male and female flies of three D. melanogaster lines. Discriminant analysis of male (upper panel) and female (lower panel) flies of three D.melanogaster lines: the wild type Canton (Cs; black colored circles), the double transgenic 5670-Gal4 > UAS-traF (5670-tra; green colored circles) and homozygous mutant desat11573-Gal4 (desat1; purple colored circles). For each sex, a Factorial Discriminant Analysis (FDA) was carried out using ∑CHCs, Monoenes% and/or Dienes%, where appropriate, and LinCHCs% and BrCHCs% as quantitative variables and the “strain/age” as a qualitative variable. For the sake of clarity, the graphical representation of the results is simplified by using for each “strain/age” group the corresponding barycenter and equiprobable ellipse (p = 0.05) instead of individuals. Percentage in the axis tittle indicates the proportion of total variability explained by this axis. These data correspond to the analysis shown on Fig. 1. (PNG 14352 kb)

Supplemental Figure 2.
Age-related variation of cuticular hydrocarbons in male and female flies of various wild type and derived lines. Discriminant analysis of male (upper panel) and female (lower panel) flies of the following wild type strains: Cs (black colored circles), Dijon2000 (Di2; orange colored circles), Oregon-R (Or-R; pink colored circles) and Zimbabwe30 (Z30; green colored lines and circles, diamonds and triangles). These data correspond to the analysis shown on Fig. 2. For further explanation see Supp Fig. 1 legend. (PNG 14166 kb)

Supplemental Figure 3.
Age-related variation of cuticular hydrocarbons in male and female flies of oenocyteless lines. Discriminant analysis of male (upper panel) and female (lower panel) flies of control Cs flies (black colored circle) and of four oenocyteless genotypes (oe−) resulting of the following (females x males) crosses between two transgenic lines: #2 x #5 (2 × 5; light blue colored empty circles), #5 x #2 (5 × 2; light blue colored filled circles), #7 x #5 (7 × 5; dark blue colored empty circles) and #5 x #7 (5 × 7; dark blue colored filled circles). These data correspond to the analysis shown on Fig. 3. For further explanation see Supp Fig. 1 legend. Due to the weak strength, oe− crosses are only indicated by their centroïds. (PNG 14306 kb)

Supplemental Figure 4.
Age-related variation of desaturated cuticular hydrocarbons in male and female flies of Zimbabwe and derived lines. The variation of desaturated cuticular hydrocarbons (CHCs) was noted in males (top) and females (bottom), at the following ages: 4 days (4d), 8 days (8d), 12 days (12d), and 24 days (24d; indicated below the box plots), in the Z30 (left panels), 3 W1 (center panels) and 1 W14 (right panels). Each box plot represents the sum of isomers of CHCs with a desaturation on Carbon 9 (Iso 9), on Carbon 7 (Iso 7), or on Carbon 5 (Iso 5). These data correspond to the analysis shown on Fig. 3. For each sex and strain, we analysed the ontogeny of each CHC isomer using a Kruskall-Wallis test followed by Conover-Iman multiple pairwise comparisons (p = 0.05, with a Bonferroni correction). Different letters indicate significant differences (p < 0.05). For Z30 males, n = 11; for Z30 females, n = 16 except at 24d (n = 6). For 3 W1 males, n = 20; for 3 W1 females, n = 12–20. For 1 W14 8d flies, n = 20, and n = 2 in 2d flies, n = 8 for 4d males and n = 3 for 4d females. (PNG 7327 kb)
ESM 1
(XLSX 2898 kb)
ESM 2
(XLSX 518 kb)
ESM 3
(XLSX 409 kb)
Rights and permissions
About this article
Cite this article
Cortot, J., Farine, JP., Ferveur, JF. et al. Aging-Related Variation of Cuticular Hydrocarbons in Wild Type and Variant Drosophila melanogaster. J Chem Ecol 48, 152–164 (2022). https://doi.org/10.1007/s10886-021-01344-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01344-0



