Abstract
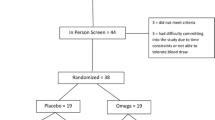
This pilot placebo-controlled trial (NCT00965068, 07/28/2009) explored effects of cholesterol supplementation on autism spectrum disorder (ASD) symptoms in children with abnormally low cholesterol. Fifteen children age 4–12 with ASD and cholesterol <5th percentile for age/sex were randomized to 225 mg cholesterol vs. matched placebo twice daily for 12 weeks, followed by 12-week open extension. The cholesterol daily dose approximated the amount in two eggs. Measures included the Ohio Autism Clinical Impression Scale (OACIS, primary outcome), and Peabody Picture Vocabulary Test (PPVT-4). Effect sizes were emphasized over statistical tests. On the OACIS-Improvement score for overall ASD symptoms, both groups improved, with cholesterol showing nominally (nonsignificantly) more improvement. The cholesterol group improved more than placebo by medium to large effects in OACIS-I nonverbal communication (d = −0.643); and OACIS-I problems with anxiety and fear (d = −0.671). Moreover, cholesterol showed significantly more improvement than placebo in PPVT-4 receptive language (p = 0.040). During the 12-week open extension, children originally assigned placebo improved significantly more in OACIS-I social interaction (p = 0.026, d = 1.170) from end of randomized phase than those receiving cholesterol throughout and improved more by large effects in OACIS-I aberrant behavior (d = 0.924), and anxiety/fear (d = 0.925). Graphs suggest continued open-trial improvement for the cholesterol group. Adverse events were similar between treatments. Cholesterol supplementation of children with ASD and cholesterol below the 5th percentile appears safe. The effect on communication, social interaction, and anxiety/fear looks encouraging; deserving further study, possibly over a longer period or at higher doses.


Similar content being viewed by others
References
Adams, J. B. (2015). Vitamin/mineral supplements for children and adults with autism. Vitamins & Minerals, 04(01). https://doi.org/10.4172/2376-1318.1000127.
Adams, J. B., & Holloway, C. (2004). Pilot study of a moderate dose multivitamin/mineral supplement for children with autistic Spectrum disorder. The Journal of Alternative and Complementary Medicine, 10(6), 1033–1039. https://doi.org/10.1089/acm.2004.10.1033.
Akshoomoff, N. (2006). Use of the Mullen scales of Early learning for the assessment of Young children with autism Spectrum disorders. Child Neuropsychology, 12(4–5), 269–277. https://doi.org/10.1080/09297040500473714.
American Psychiatric Association. (1994). Diagnostic and statistical manual of mental disorders (4th ed.). Arlington, VA: Author.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Arlington, VA: Author.
Aneja, A., & Tierney, E. (2008). Cholesterol deficit in autism: Insights from Smith–Lemli–Opitz syndrome. Autism, 69–79. https://doi.org/10.1007/978-1-60327-489-0_3.
Arnold, L. E., Aman, M. G., Hollway, J., Hurt, E., Bates, B., Li, X., Farmer, C., Anand, R., Thompson, S., Ramadan, Y., & Williams, C. (2012). Placebo-controlled pilot trial of Mecamylamine for treatment of autism Spectrum disorders. Journal of Child and Adolescent Psychopharmacology, 22(3), 198–205. https://doi.org/10.1089/cap.2011.0056.
Bochem, A. E., Holleboom, A. G., Romijn, J. A., Hoekstra, M., Dallinga-Thie, G. M., Motazacker, M. M., Hovingh, G. K., Kuivenhoven, J. A., & Stroes, E. S. G. (2013). High density lipoprotein as a source of cholesterol for adrenal steroidogenesis: A study in individuals with low plasma HDL-C. Journal of Lipid Research, 54(6), 1698–1704.
Borkowski, A. J., Levin, S., Delcroix, C., Mahler, A., & Verhas, V. (1967). Blood cholesterol and hydrocortisone production in man: Quantitative aspects of the utilization of circulating cholesterol by the adrenals at rest and under adrenocorticotropin stimulation. The Journal of Clinical Investigation, 46(5), 797–811.
Bracken, B. A., Prasse, D. P., & McCallum, R. S. (1984). Peabody picture vocabulary test-revised: An appraisal and review. School Psychology Review, 13(1), 49–60. https://doi.org/10.1080/02796015.1984.12085079.
Bukelis, I., Porter, F. D., Zimmerman, A. W., & Tierney, E. (2007). Smith-Lemli-Opitz syndrome and autism Spectrum disorder. American Journal of Psychiatry, 164(11), 1655–1661. https://doi.org/10.1176/appi.ajp.2007.07020315.
Butter, E., & Mulick, J. (2006). The Ohio autism clinical impressions scale (OACIS). Columbus, OH: Children’s Research Institute.
Campbell, J. (1998). Book review: Peabody picture vocabulary test, third edition. Journal of Psychoeducational Assessment, 16(4), 334–338. https://doi.org/10.1177/073428299801600405.
Cartocci, V., Catallo, M., Tempestilli, M., Segatto, M., Pfrieger, F. W., Bronzuoli, M. R., Scuderi, C., Servadio, M., Trezza, V., & Pallottini, V. (2018). Altered brain cholesterol/Isoprenoid metabolism in a rat model of autism Spectrum disorders. Neuroscience, 372, 27–37. https://doi.org/10.1016/j.neuroscience.2017.12.053.
Centers for Disease Control and Prevention (CDC). (2005). National health and nutrition examination survey data. Hyattsville, MD: US Department of Health and Human Services, Centers for Disease Control and Prevention, 2008.
Choque Olsson, N., & Bölte, S. (2013). Brief report: “Quick and (not so) dirty” assessment of change in autism: Cross-cultural reliability of the developmental disabilities CGAS and the OSU autism CGI. Journal of Autism and Developmental Disorders, 44(7), 1773–1778. https://doi.org/10.1007/s10803-013-2029-y.
Christensen, D. L., Van Naarden Braun, K., Baio, J., Bilder, D., Charles, J., Constantino, J. N., et al. (2018). Prevalence and characteristics of autism spectrum disorder among children aged 8 years — autism and developmental disabilities monitoring network, 11 sites, United States, 2012. MMWR. Surveillance Summaries, 65(13), 1–23. Available at: https://doi.org/10.15585/mmwr.ss6513a1.
Cohen, J. (1988). Statistical power analysis for the behavioral sciences (second ed.). New York: Lawrence Erlbaum Associates.
Cross, J. L., Iben, J., Simpson, C. L., Thurm, A., Swedo, S., Tierney, E., Bailey-Wilson, J. E., Biesecker, L. G., Porter, F. D., & Wassif, C. A. (2014). Determination of the allelic frequency in Smith-Lemli-Opitz syndrome by analysis of massively parallel sequencing data sets. Clinical Genetics, 87(6), 570–575. https://doi.org/10.1111/cge.12425.
Diaz-Stransky, A., & Tierney, E. (2012). Cognitive and behavioral aspects of Smith-Lemli-Opitz syndrome. American Journal of Medical Genetics Part C: Seminars in Medical Genetics, 160C(4), 295–300. https://doi.org/10.1002/ajmg.c.31342.
Dunn, L. M., & Dunn, D. M. (2007). PPVT-4: Peabody picture vocabulary test (4th ed.p. 2007). Minneapolis, MN: Pearson Assessments.
Elias, E. R., & Irons, M. (1995). Abnormal cholesterol metabolism in Smith-Lemli-Opitz syndrome. The Journal of Pediatrics, 7(6), 710–714. https://doi.org/10.1097/00008480-199512000-00015.
Elias, E. R., Irons, M. B., Hurley, A. D., Tint, G. S., & Salen, G. (1997). Clinical effects of cholesterol supplementation in six patients with the Smith-Lemli-Opitz syndrome (SLOS). American Journal of Medical Genetics, 68(3), 305–310. https://doi.org/10.1002/(sici)1096-8628(19970131)68:3<305::aid-ajmg11>3.0.co;2-x.
Frye, C. A., & Llaneza, D. C. (2010). Corticosteroid and neurosteroid dysregulation in an animal model of autism, BTBR mice. Physiology & Behavior, 100(3), 264–267. https://doi.org/10.1016/j.physbeh.2010.03.005.
Frye, R. E., DeLaTorre, R., Taylor, H. B., Slattery, J., Melnyk, S., Chowdhury, N., & James, S. J. (2013). Metabolic effects of sapropterin treatment in autism spectrum disorder: A preliminary study. Translational Psychiatry, 3(3), e237–e237. https://doi.org/10.1038/tp.2013.14.
Frye, R. E., Slattery, J., Delhey, L., Furgerson, B., Strickland, T., Tippett, M., Sailey, A., Wynne, R., Rose, S., Melnyk, S., Jill James, S., Sequeira, J. M., & Quadros, E. V. (2016). Folinic acid improves verbal communication in children with autism and language impairment: A randomized double-blind placebo-controlled trial. Molecular Psychiatry, 23(2), 247–256. https://doi.org/10.1038/mp.2016.168.
Gillberg, C., Fernell, E., Kočovská, E., Minnis, H., Bourgeron, T., Thompson, L., & Allely, C. S. (2017). The role of cholesterol metabolism and various steroid abnormalities in autism spectrum disorders: A hypothesis paper. Autism Research, 10(6), 1022–1044. https://doi.org/10.1002/aur.1777.
Gray, S., Plante, E., Vance, R., & Henrichsen, M. (1999). The diagnostic accuracy of four vocabulary tests administered to preschool-age children. Language, Speech, and Hearing Services in Schools, 30(2), 196–206. https://doi.org/10.1044/0161-1461.3002.196.
Irons, M., Roy Elias, E., Salen, G., Tint, G. S., & Batta, A. (1993). Defective cholesterol biosynthesis in Smith-Lemli-Opitz syndrome. The Lancet, 341(8857), 1414. https://doi.org/10.1016/0140-6736(93)90983-n.
Irons, M., Elias, E. R., Abuelo, D., Bull, M. J., Greene, C. L., Johnson, V. P., Keppen, L., Schanen, C., Tint, G. S., & Salen, G. (1997). Treatment of Smith-Lemli-Opitz syndrome: Results of a multicenter trial. American Journal of Medical Genetics, 68(3), 311–314. https://doi.org/10.1002/(sici)1096-8628(19970131)68:3<311::aid-ajmg12>3.0.co;2-w.
Korade, Z., Folkes, O. M., & Harrison, F. E. (2013). Behavioral and serotonergic response changes in the Dhcr7-HET mouse model of Smith–Lemli–Opitz syndrome. Pharmacology Biochemistry and Behavior, 106, 101–108. https://doi.org/10.1016/j.pbb.2013.03.007.
Laurent, J., Swerdlik, M., & Ryburn, M. (1992). Review of validity research on the Stanford-Binet intelligence scale: Fourth edition. Psychological Assessment, 4(1), 102–112. https://doi.org/10.1037/1040-3590.4.1.102.
Lee, R. W. Y., & Tierney, E. (2011). Hypothesis: The role of sterols in autism Spectrum disorder. Autism Research and Treatment, 2011, 1–7. https://doi.org/10.1155/2011/653570.
Moy, S. S., Nadler, J. J., Young, N. B., Nonneman, R. J., Grossman, A. W., Murphy, D. L., D’Ercole, A. J., Crawley, J. N., Magnuson, T. R., & Lauder, J. M. (2009). Social approach in genetically engineered mouse lines relevant to autism. Genes, Brain and Behavior, 8(2), 129–142. https://doi.org/10.1111/j.1601-183x.2008.00452.x.
Mullen, E. M. (1995). Mullen scales of early learning (pp. 58–64). Circle Pines, MN: American Guidance Service.
Nwokoro, N. A., & Mulvihill, J. J. (1997). Cholesterol and bile acid replacement therapy in children and adults with Smith-Lemli-Opitz (SLO/RSH) syndrome. American Journal of Medical Genetics, 68(3), 315–321. https://doi.org/10.1002/(sici)1096-8628(19970131)68:3<315::aid-ajmg13>3.0.co;2-w.
Opitz, J. M. (1999). RSH (so-called Smith-Lemli-Opitz) syndrome. Current Opinion in Pediatrics, 11(4), 353–362. https://doi.org/10.1097/00008480-199908000-00015.
Pauli, R. M., Williams, M. S., Josephson, K. D., & Tint, G. S. (1997). Smith-Lemli-Opitz syndrome: Thirty-year follow-up of “S” of “RSH” syndrome. American Journal of Medical Genetics, 68, 260–262. https://doi.org/10.1002/(sici)1096-8628(19970131)68:3<260::aid-ajmg3>3.0.co;2-q.
Porter, F. D., & Herman, G. E. (2010). Malformation syndromes caused by disorders of cholesterol synthesis. Journal of Lipid Research, 52(1), 6–34. https://doi.org/10.1194/jlr.r009548.
Restrepo, M. A., Schwanenflugel, P. J., Blake, J., Neuharth-Pritchett, S., Cramer, S. E., & Ruston, H. P. (2006). Performance on the PPVT–III and the EVT: Applicability of the measures with African American and European American preschool children. Language, Speech, and Hearing Services in Schools, 37(1), 17–27. https://doi.org/10.1044/0161-1461(2006/003).
Roid, G. H. (2003). Stanford-Binet intelligence scales interpretive manual: Expanded guide to the interpretation of SB5 test results. Itasca, IL: Riverside Publishing.
Ryan, A. K., Bartlett, K., Clayton, P., Eaton, S., Mills, L., Donnai, D., Winter, R. M., & Burn, J. (1998). Smith-Lemli-Opitz syndrome: A variable clinical and biochemical phenotype. Journal of Medical Genetics, 35(7), 558–565. https://doi.org/10.1136/jmg.35.7.558.
Segatto, M., Di Giovanni, A., Marino, M., & Pallottini, V. (2013). Analysis of the protein network of cholesterol homeostasis in different brain regions: An age and sex dependent perspective. Journal of Cellular Physiology, 228(7), 1561–1567. https://doi.org/10.1002/jcp.24315.
Sikora, D. M., Pettit-Kekel, K., Penfield, J., Merkens, L. S., & Steiner, R. D. (2006). The near universal presence of autism spectrum disorders in children with Smith–Lemli–Opitz syndrome. American Journal of Medical Genetics Part A, 140A(14), 1511–1518. https://doi.org/10.1002/ajmg.a.31294.
Singh, K., Connors, S. L., Macklin, E. A., Smith, K. D., Fahey, J. W., Talalay, P., & Zimmerman, A. W. (2014). Sulforaphane treatment of autism spectrum disorder (ASD). Proceedings of the National Academy of Sciences, 111(43), 15550–15555. https://doi.org/10.3410/f.722006003.793500809.
Sparrow, S., Balla, D., & Cicchetti, D. (1984). Vineland adaptive behavior scales: Expanded form manual. Circle Pines: American Guidance Service.
Strous, R. D., Golubchik, P., Maayan, R., Mozes, T., Tuati-Werner, D., Weizman, A., & Spivak, B. (2005). Lowered DHEA-S plasma levels in adult individuals with autistic disorder. European Neuropsychopharmacology, 15(3), 305–309. https://doi.org/10.1016/j.euroneuro.2004.12.004.
Thurm, A., Tierney, E., Farmer, C., Albert, P., Joseph, L., Swedo, S., Bianconi, S., Bukelis, I., Wheeler, C., Sarphare, G., Lanham, D., Wassif, C. A., & Porter, F. D. (2016). Development, behavior, and biomarker characterization of Smith-Lemli-Opitz syndrome: An update. Journal of Neurodevelopmental Disorders, 8(1), 12. https://doi.org/10.1186/s11689-016-9145-x.
Tierney, E., Nwokoro, N. A., Porter, F. D., Freund, L. S., Ghuman, J. K., & Kelley, R. I. (2001). Behavior phenotype in the RSH/Smith-Lemli-Opitz syndrome. American Journal of Medical Genetics, 98(2), 191–200. https://doi.org/10.1002/1096-8628(20010115)98:2<191::aid-ajmg1030>3.0.co;2-m.
Tierney, E., Bukelis, I., Thompson, R. E., Ahmed, K., Aneja, A., Kratz, L., & Kelley, R. I. (2006). Abnormalities of cholesterol metabolism in autism spectrum disorders. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics, 141B(6), 666–668. https://doi.org/10.1002/ajmg.b.30368.
Tierney, E., Conley, S. K., Goodwin, H., & Porter, F. D. (2010). Analysis of short-term behavioral effects of dietary cholesterol supplementation in Smith–Lemli–Opitz syndrome. American Journal of Medical Genetics Part A, 152(1), 91–95. https://doi.org/10.1002/ajmg.a.33148.
Waage-Baudet, H., Lauder, J. M., Dehart, D. B., Kluckman, K., Hiller, S., Tint, G. S., & Sulik, K. K. (2003). Abnormal serotonergic development in a mouse model for the Smith–Lemli–Opitz syndrome: Implications for autism. International Journal of Developmental Neuroscience, 21(8), 451–459. https://doi.org/10.1016/j.ijdevneu.2003.09.002.
Wang, H. (2014). Lipid rafts: A signaling platform linking cholesterol metabolism to synaptic deficits in autism spectrum disorders. Frontiers in Behavioral Neuroscience, 8. https://doi.org/10.3389/fnbeh.2014.00104.
Williams, K. T. (2007). Expressive vocabulary test. Minneapolis, MN: Pearson Assessments.
Wink, L. K., Early, M., Schaefer, T., Pottenger, A., Horn, P., McDougle, C. J., & Erickson, C. A. (2014). Body mass index change in autism Spectrum disorders: Comparison of treatment with Risperidone and Aripiprazole. Journal of Child and Adolescent Psychopharmacology, 24(2), 78–82. https://doi.org/10.1089/cap.2013.0099.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Nadja Ging-Jehli, Deepa Manda, Elizabeth Hurt, and Stacey Moone have no conflicts of interest to disclose. Dr. Arnold has received research funding from Curemark, Forest, Lilly, Neuropharm, Novartis, Noven, Shire, Supernus, Roche, and YoungLiving (as well as NIH and Autism Speaks), has consulted with Gowlings, Neuropharm, Organon, Pfizer, Sigma Tau, Shire, Tris Pharma, and Waypoint, and been on advisory boards for Arbor, Ironshore, Novartis, Noven, Otsuka, Pfizer, Roche, Seaside Therapeutics, Sigma Tau, Shire. Dr. Hollway has received research contracts, or research support from Forest Research Institute, Sunovion Pharmaceuticals, F. Hoffman-La Roche, Supernus, Young Living, Autism Speaks, and the NIH. She has served on the advisory board for YAMO Pharmaceuticals and has consulted with MedAvante-ProPhase.
Ethics Approval
The research was approved by The Ohio State University’s Institutional Review Board (IRB). The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Informed Consent
Written informed consent and assent (if the child was capable) was obtained from parents. The authors affirm that human research participants provided informed consent for the use and share of their de-identified information for future research and publication.
Consent to Participate
Informed consent from parents and assent (if the child was capable) from participants was obtained using forms and procedures approved by the Ohio State University IRB.
Consent to Publish
The authors affirm that human research participants provided informed consent for the use and share of their de-identified information for future research and publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Nadja Rita Ging-Jehli and Deepa Manda are Shared First Authorship
Electronic supplementary material
ESM 1
(DOCX 22.9 kb)
Rights and permissions
About this article
Cite this article
Ging-Jehli, N.R., Manda, D., Hollway, J. et al. A Placebo-Controlled Pilot Exploration of Cholesterol Supplementation for Autistic Symptoms in Children with Low Cholesterol. J Dev Phys Disabil 33, 819–837 (2021). https://doi.org/10.1007/s10882-020-09776-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10882-020-09776-4