Abstract
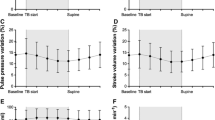
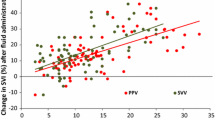
Non-invasive respiratory variations in arterial pulse pressure using infrared-plethysmography (PPVCNAP) are able to predict fluid responsiveness in mechanically ventilated patients. However, they cannot be continuously monitored. The present study evaluated a new algorithm allowing continuous measurements of PPVCNAP (PPVCNAPauto) (CNSystem, Graz, Austria). Thirty-five patients undergoing vascular surgery were studied after induction of general anaesthesia. Stroke volume was measured using the VigileoTM/FloTracTM. Invasive pulse pressure variations were manually calculated using an arterial line (PPVART) and PPVCNAPauto was continuously displayed. PPVART and PPVCNAPauto were simultaneously recorded before and after volume expansion (500 ml hydroxyethylstarch). Subjects were defined as responders if stroke volume increased by ≥15 %. Twenty-one patients were responders. Before volume expansion, PPVART and PPVCNAPauto exhibited a bias of 0.1 % and limits of agreement from −7.9 % to 7.9 %. After volume expansion, PPVART and PPVCNAPauto exhibited a bias of −0.4 % and limits of agreement from −5.3 % to 4.5 %. A 14 % baseline PPVART threshold discriminated responders with a sensitivity of 86 % (95 % CI 64–97 %) and a specificity of 100 % (95 % CI 77–100 %). Area under the receiver operating characteristic (ROC) curve for PPVART was 0.93 (95 % CI 0.79–0.99). A 15 % baseline PPVCNAPauto threshold discriminated responders with a sensitivity of 76% (95 % CI 53–92 %) and a specificity of 93 % (95 % CI 66–99 %). Area under the ROC curves for PPVCNAPauto was 0.91 (95 % CI 0.76–0.98), which was not different from that for PPVART. When compared with PPVART, PPVCNAPauto performs satisfactorily in assessing fluid responsiveness in hemodynamically stable surgical patients.




Similar content being viewed by others
References
Hamilton MA, Cecconi M, Rhodes A. A systematic review and meta-analysis on the use of preemptive hemodynamic intervention to improve postoperative outcomes in moderate and high-risk surgical patients. Anesth Analg. 2011;112:1392–402.
Vallet B, Blanloeil Y, Cholley B, Orliaguet G, Pierre S, Tavernier B. Socie te franc aise d’anesthe sie et de re a. Guidelines for perioperative haemodynamic optimization. Ann Fr Anesth Reanim. 2013;32:e151–8.
Marik PE, Cavallazzi R, Vasu T, Hirani A. Dynamic changes in arterial waveform derived variables and fluid responsiveness in mechanically ventilated patients: a systematic review of the literature. Crit Care Med. 2009;37:2642–7.
Benes J, Chytra I, Altmann P, Hluchy M, Kasal E, Svitak R, Pradl R, Stepan M. Intraoperative fluid optimization using stroke volume variation in high risk surgical patients: results of prospective randomized study. Crit Care. 2010;14:R118.
Lopes MR, Oliveira MA, Pereira VO, Lemos IP, Auler JO Jr, Michard F. Goal-directed fluid management based on pulse pressure variation monitoring during high-risk surgery: a pilot randomized controlled trial. Crit Care. 2007;11:R100.
Mayer J, Boldt J, Mengistu AM, Rohm KD, Suttner S. Goal-directed intraoperative therapy based on autocalibrated arterial pressure waveform analysis reduces hospital stay in high-risk surgical patients: a randomized, controlled trial. Crit Care. 2010;14:R18.
Ramsingh DS, Sanghvi C, Gamboa J, Cannesson M, Applegate RL 2nd. Outcome impact of goal directed fluid therapy during high risk abdominal surgery in low to moderate risk patients: a randomized controlled trial. J Clin Monit Comput. 2013;27:249–57.
Biais M, Ouattara A, Janvier G, Sztark F. Case scenario: respiratory variations in arterial pressure for guiding fluid management in mechanically ventilated patients. Anesthesiology. 2012;116:1354–61.
Cannesson M. Arterial pressure variation and goal-directed fluid therapy. J Cardiothorac Vasc Anesth. 2010;24:487–97.
Bogert LW, van Lieshout JJ. Non-invasive pulsatile arterial pressure and stroke volume changes from the human finger. Exp Physiol. 2005;90:437–46.
Penaz J, Voigt A, Teichmann W. Contribution to the continuous indirect blood pressure measurement. Z Gesamte Inn Med. 1976;31:1030–3.
Epstein RH, Bartkowski RR, Huffnagle S. Continuous noninvasive finger blood pressure during controlled hypotension. A comparison with intraarterial pressure. Anesthesiology. 1991;75:796–803.
Gibbs NM, Larach DR, Derr JA. The accuracy of Finapres noninvasive mean arterial pressure measurements in anesthetized patients. Anesthesiology. 1991;74:647–52.
Stokes DN, Clutton-Brock T, Patil C, Thompson JM, Hutton P. Comparison of invasive and non-invasive measurements of continuous arterial pressure using the Finapres. Br J Anaesth. 1991;67:26–35.
Hahn R, Rinosl H, Neuner M, Kettner SC. Clinical validation of a continuous non-invasive haemodynamic monitor (CNAP™ 500) during general anaesthesia. Br J Anaesth. 2012;108:581–5.
Ilies C, Kiskalt H, Siedenhans D, Meybohm P, Steinfath M, Bein B, Hanss R. Detection of hypotension during Caesarean section with continuous non-invasive arterial pressure device or intermittent oscillometric arterial pressure measurement. Br J Anaesth. 2012;109:413–9.
Schramm C, Huber A, Plaschke K. The accuracy and responsiveness of continuous noninvasive arterial pressure during rapid ventricular pacing for transcatheter aortic valve replacement. Anesth Analg. 2013;117:76–82.
Biais M, Stecken L, Ottolenghi L, Roullet S, Quinart A, Masson F, Sztark F. The ability of pulse pressure variations obtained with CNAP device to predict fluid responsiveness in the operating room. Anesth Analg. 2011;113:523–8.
Monnet X, Dres M, Ferre A, Le Teuff G, Jozwiak M, Bleibtreu A, Le Deley MC, Chemla D, Richard C, Teboul JL. Prediction of fluid responsiveness by a continuous non-invasive assessment of arterial pressure in critically ill patients: comparison with four other dynamic indices. Br J Anaesth. 2012;109:330–8.
Minto C, Schnider T, Egan T, Youngs E, Lemmens H, Gambus P, Billard V, Hoke J, Moore K, Hermann D, Muir K, Mandema J, Shafer S. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil. I. Model developpement. Anesthesiology. 1997;86:10–23.
Minto C, Schnider T, Shafer S. Pharmacokinetics and pharmacodynamics of remifentanil. II. Model application. Anesthesiology. 1997;86:24–33.
Schnider T, Minto C, Shafer S, Gambus P, Andresen C, Goodale D, Youngs E. The influence of age on propofol pharmacodynamics. Anesthesiology. 1999;90:1506–16.
Fortin J, Wellisch A, Maier K. CNAP - Evolution of Continuous Non-Invasive Arterial Blood Pressure Monitoring. Biomed Tech (Berl). 2013. doi:10.1515/bmt-2013-4179.
Michard F, Boussat S, Chemla D, Anguel N, Mercat A, Lecarpentier Y, Richard C, Pinsky MR, Teboul JL. Relation between respiratory changes in arterial pulse pressure and fluid responsiveness in septic patients with acute circulatory failure. Am J Respir Crit Care Med. 2000;162:134–8.
Stetz CW, Miller RG, Kelly GE, Raffin TA. Reliability of the thermodilution method in the determination of cardiac output in clinical practice. Am Rev Respir Dis. 1982;126:1001–4.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;327:307–10.
Hanley JA, McNeil BJ. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology. 1983;148:839–43.
Biais M, Bernard O, Ha JC, Degryse C, Sztark F. Abilities of pulse pressure variations and stroke volume variations to predict fluid responsiveness in prone position during scoliosis surgery. Br J Anaesth. 2010;104:407–13.
Ray P, Le Manach Y, Riou B, Houle TT. Statistical evaluation of a biomarker. Anesthesiology. 2010;112:1023–40.
Prys-Roberts C. Cardiovascular monitoring in patients with vascular disease. Br J Anaesth. 1981;53:767–76.
Van Bergen FH, Weatherhead DS, Treloar AE, Dobkin AB, Buckley JJ. Comparison of indirect and direct methods of measuring arterial blood pressure. Circulation. 1954;10:481–90.
Scheer B, Perel A, Pfeiffer UJ. Clinical review: complications and risk factors of peripheral arterial catheters used for haemodynamic monitoring in anaesthesia and intensive care medicine. Crit Care. 2002;6:199–204.
Cohen A, Reyes R, Kirk M, Fulks RM. Osler’s nodes, pseudoaneurysm formation, and sepsis complicating percutaneous radial artery cannulation. Crit Care Med. 1984;12:1078–9.
Evans PJ, Kerr JH. Arterial occlusion after cannulation. Br Med J. 1975;3:197–9.
McEllistrem RF, O’Toole DP, Keane P. Post-cannulation radial artery aneurysm–a rare complication. Can J Anaesth. 1990;37:907–9.
Slogoff S, Keats AS, Arlund C. On the safety of radial artery cannulation. Anesthesiology. 1983;59:42–7.
Lansdorp B, Ouweneel D, de Keijzer A, van der Hoeven JG, Lemson J, Pickkers P. Non-invasive measurement of pulse pressure variation and systolic pressure variation using a finger cuff corresponds with intra-arterial measurement. Br J Anaesth. 2011;107:540–5.
Cannesson M, Attof Y, Rosamel P, Desebbe O, Joseph P, Metton O, Bastien O, Lehot JJ. Respiratory variations in pulse oximetry plethysmographic waveform amplitude to predict fluid responsiveness in the operating room. Anesthesiology. 2007;106:1105–11.
Feissel M, Teboul JL, Merlani P, Badie J, Faller JP, Bendjelid K. Plethysmographic dynamic indices predict fluid responsiveness in septic ventilated patients. Intensive Care Med. 2007;33:993–9.
Cannesson M, Delannoy B, Morand A, Rosamel P, Attof Y, Bastien O, Lehot JJ. Does the Pleth variability index indicate the respiratory-induced variation in the plethysmogram and arterial pressure waveforms? Anesth Analg. 2008;106:1189–94.
Biais M, Cottenceau V, Petit L, Masson F, Cochard JF, Sztark F. Impact of norepinephrine on the relationship between pleth variability index and pulse pressure variations in ICU adult patients. Crit Care. 2011;15:R168.
Cannesson M, Desebbe O, Rosamel P, Delannoy B, Robin J, Bastien O, Lehot JJ. Pleth variability index to monitor the respiratory variations in the pulse oximeter plethysmographic waveform amplitude and predict fluid responsiveness in the operating theatre. Br J Anaesth. 2008;101:200–6.
Loupec T, Nanadoumgar H, Frasca D, Petitpas F, Laksiri L, Baudouin D, Debaene B, Dahyot-Fizelier C, Mimoz O. Pleth variability index predicts fluid responsiveness in critically ill patients. Crit Care Med. 2011;39:294–9.
Biais M, Nouette-Gaulain K, Cottenceau V, Vallet A, Cochard JF, Revel P, Sztark F. Cardiac output measurement in patients undergoing liver transplantation: pulmonary artery catheter versus uncalibrated arterial pressure waveform analysis. Anesth Analg. 2008;106:1480–6.
Biais M, Nouette-Gaulain K, Cottenceau V, Revel P, Sztark F. Uncalibrated pulse contour-derived stroke volume variation predicts fluid responsiveness in mechanically ventilated patients undergoing liver transplantation. Br J Anaesth. 2008;101:761–8.
Myburgh JA, Finfer S, Bellomo R, Billot L, Cass A, Gattas D, Glass P, Lipman J, Liu B, McArthur C, McGuinness S, Rajbhandari D, Taylor CB, Webb SA, Investigators C. Australian, New Zealand Intensive Care Society Clinical Trials G. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367:1901–11.
Perner A, Haase N, Guttormsen AB, Tenhunen J, Klemenzson G, Aneman A, Madsen KR, Moller MH, Elkjaer JM, Poulsen LM, Bendtsen A, Winding R, Steensen M, Berezowicz P, Soe-Jensen P, Bestle M, Strand K, Wiis J, White JO, Thornberg KJ, Quist L, Nielsen J, Andersen LH, Holst LB, Thormar K, Kjaeldgaard AL, Fabritius ML, Mondrup F, Pott FC, Moller TP, Winkel P, Wetterslev J, Group ST, Scandinavian Critical Care Trials G. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med 2012;367:124–34.
Acknowledgments
The authors thank Ray Cooke, Ph.D. (Assistant Professor and Director, Département Langues et Cultures, University of Bordeaux, Bordeaux, France) for reviewing this manuscript.
Author contribution
All authors have approved the submitted manuscript. MB conceived study design, performed data collection, data interpretation and analysis and wrote the manuscript. LS and AM: participated in the conception, recruitment, and interpretation of data. SR and AQ: participated in the recruitment and interpretation of data. FS participated in writing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in this study were in accordance with the ethical standard of the local ethic committee and have been performed in accordance with the ethical standard as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Conflicts of interest
M.B received honoraria from Edwards Lifesciences and Pulsion Medical System as a lecturer. Other authors declare that they have no competing interests.
Financial disclosure
Only departmental funds were used for this study. No external funds were obtained. The manufacturers (CNSystem, Graz, Austria) provided the material free of charge.
Rights and permissions
About this article
Cite this article
Biais, M., Stecken, L., Martin, A. et al. Automated, continuous and non-invasive assessment of pulse pressure variations using CNAP® system. J Clin Monit Comput 31, 685–692 (2017). https://doi.org/10.1007/s10877-016-9899-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-016-9899-4