Abstract
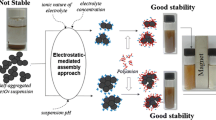
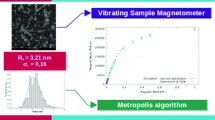
Here, we used experiments and Monte Carlo simulations (MC) to identify appropriate poly (ethylene) glycol (PEG) polymer molecular weight (Mw) as the coating agent for iron oxide nanoparticles (IONPs) resulting in individual particles in water. IONPs coated with PEG Mw’s ranging from 1500 to 6000 Da showed highly stable dispersions for more than a year. Transmission electron microscopy (TEM) results of the dispersions revealed that particles coated with lower PEG Mw were aggregated, which shifted to individual form with increasing PEG Mw used for coating. We find that this transformation is due to increased PEG layer shell thickness (from 1.47 to 6.46 nm) with increasing PEG Mw (from 1500 to 6000 Da), which imparted sufficient repulsive steric forces to overcome the attractive van der Waals and magnetic forces. To predict appropriate PEG Mw resulting individual IONPs in water, the number density distribution of different PEG Mw coated particles was determined using MC simulation which employs interparticle potentials between the interacting particles as a function of their interparticle distance. The predictions gave very good agreement with our’s and others’ experimental data. Thus, the study helps to identify a suitable polymer Mw as a coating agent, resulting in individual nanoparticle dispersion.







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author, [C. Ravikumar], upon request.
Code Availability
An algorithm is provided in the article. The codes used for simulation are available from the corresponding author, [C. Ravikumar], upon request.
References
C. Cruje and D. Chithrani (2014). Rev Nanosci Nanotechnol. https://doi.org/10.1166/rnn.2014.1042.
J. M. Harris, Poly (ethylene glycol) chemistry: biotechnical and biomedical applications (Springer Science & Business Media, 1992).
D. Hutanu (2014). Mod Chem Appl. https://doi.org/10.4172/2329-6798.1000132.
A. P. Khandhar, P. Keselman, S. J. Kemp, R. M. Ferguson, P. W. Goodwill, S. M. Conolly, and K. M. Krishnan (2017). Nanoscale. https://doi.org/10.1039/C6NR08468K.
K. Knop, R. Hoogenboom, D. Fischer, and U. S. Schubert (2010). Angew Chem Int Ed. https://doi.org/10.1002/anie.200902672.
J. Sosa Acosta, C. Iriarte-Mesa, G. Ortega, and A. Díaz-García (2020). Top Curr Chem. https://doi.org/10.1007/s41061-019-0277-9.
J. Sosa Acosta, J. Silva, L. Fernandez Izquierdo, S. D. Castañón, M. Ortiz, J. C. Zuaznabar-Gardona, and A. Díaz-García (2018). Coll Surf A. https://doi.org/10.1016/j.colsurfa.2018.02.062.
S. Laurent, D. Forge, M. Port, A. Roch, C. Robic, L. Vander Elst, and R. N. Muller (2008). Chem Rev. https://doi.org/10.1021/cr068445e.
L. H. Reddy, J. L. Arias, J. Nicolas, and P. Couvreur (2012). Chem Rev. https://doi.org/10.1021/cr300068p.
M. Mahmoudi, S. Sant, B. Wang, S. Laurent, and T. Sen (2011). Adv Drug Delivery Rev. https://doi.org/10.1016/j.addr.2010.05.006.
S. Kango, S. Kalia, A. Celli, J. Njuguna, Y. Habibi, and R. Kumar (2013). Prog Polym Sci. https://doi.org/10.1016/j.progpolymsci.2013.02.003.
A. B. Abou Hammad, M. E. Abd El-Aziz, M. S. Hasanin, and S. Kamel (2019). Carbohydr Polym. https://doi.org/10.1016/j.carbpol.2019.03.038.
A. H. Basta, H. El-Saied, M. S. Hasanin, and M. M. El-Deftar (2018). Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2017.11.061.
M. S. Hasanin and S. A. Al Kiey (2020). Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2020.06.040.
F. T. Hsu, Z. H. Wei, Y. C. Y. Hsuan, W. Lin, Y. C. Su, C. H. Liao, and C. L. Hsieh (2018). Artif Cells Nanomed Biotechnol. https://doi.org/10.1080/21691401.2018.1499661.
E. K. U. Larsen, et al. (2009). ACS Nano. https://doi.org/10.1021/nn900330m.
E. K. U. Larsen, et al. (2012). Nanoscale. https://doi.org/10.1039/C2NR11554A.
C. A. Quinto, P. Mohindra, S. Tong, and G. Bao (2015). Nanoscale. https://doi.org/10.1039/c5nr02718g.
C. Yue-Jian, T. Juan, X. Fei, Z. Jia-Bi, G. Ning, Z. Yi-Hua, D. Ye, and G. Liang (2010). Drug Dev Ind Pharm. https://doi.org/10.3109/03639041003710151.
M. Ferrari (2005). Nat Rev Cancer. https://doi.org/10.1038/nrc1566.
D. Peer, J. M. Karp, S. Hong, O. C. Farokhzad, R. Margalit, and R. Langer (2007). Nat Nanotechnol. https://doi.org/10.1038/nnano.2007.387.
S. Salmaso and P. Caliceti (2013). J Drug Deliv. https://doi.org/10.1155/2013/374252.
S. Y. Fam, C. F. Chee, C. Y. Yong, K. L. Ho, A. R. Mariatulqabtiah, and W. S. Tan (2020). Nanomaterials. https://doi.org/10.3390/nano10040787.
E. Illés, M. Szekeres, I. Toth, K. Farkas, I. Földesi, A. Szabo, B. Iván, and E. Tombácz (2018). Nanomaterials. https://doi.org/10.3390/nano8100776.
K. G. Neoh and E. T. Kang (2011). Polym Chem. https://doi.org/10.1039/C0PY00266F.
W. Xue, Y. Liu, N. Zhang, Y. Yao, P. Ma, H. Wen, S. Huang, Y. Luo, and H. M. Fan (2018). Int J Nanomed. https://doi.org/10.2147/IJN.S165451.
C. C. Hanot, Y. S. Choi, T. B. Anani, and D. Soundarrajan (2016). Int. J. Mol. Sci. 17, 54.
L. H. Deng, et al. (2021). Int J Nanomed. https://doi.org/10.2147/IJN.S271461.
L. H. Dubois, B. R. Zegarski, and R. G. Nuzzo (1986). Langmuir. https://doi.org/10.1021/la00070a006.
A. Ali, H. Zafar, M. Zia, I. Haq, A. Phull, J. Sarfraz Ali, and A. Hussain (2016). Nanotechnol Sci Appl. https://doi.org/10.2147/NSA.S99986.
K. Davis, B. Qi, M. Witmer, C. L. Kitchens, B. A. Powell, and O. T. Mefford (2014). Langmuir. https://doi.org/10.1021/la502204g.
J. V. Jokerst, T. Lobovkina, R. N. Zare, and S. S. Gambhir (2011). Nanomedicine. https://doi.org/10.2217/nnm.11.19.
C. Ravikumar, S. Kumar, and R. Bandyopadhyaya (2012). Coll. Surf. A. https://doi.org/10.1016/j.colsurfa.2012.02.007.
S. Kumar, C. Ravikumar, and R. Bandyopadhyaya (2010). Langmuir. https://doi.org/10.1021/la1017196.
S. García-Jimeno and J. Estelrich (2013). Coll Surf A. https://doi.org/10.1016/j.colsurfa.2012.12.022.
Acknowledgements
We gratefully acknowledge the Department of Science and Technology, Science and Engineering Research Board, Extra Mural Research Funding Scheme (DST/SERB/EMR), India [No.: EMR/2016/003320] for providing financial support for the work. We thank the sophisticated analytical instrumental facility (SAIF) of the Indian Institute of Technology Bombay, India for providing TEM facilities. We also thank Aimil Ltd, India for their support in analyzing the particle size distribution and zeta potential measurements of samples using Malvern Zetasizer Nano ZS. We also thank Visvesvaraya National Institute of Technology for providing other characterization facilities.
Funding
Financial support was provided by the Department of Science and Technology, Science and Engineering Research Board, Extra Mural Research Funding Scheme (DST/SERB/EMR), India [No.: EMR/2016/003320].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singapati, A.Y., Muthuraja, V., Kuthe, A.M. et al. Influence of the Molecular Weight of Poly (Ethylene Glycol) on the Aqueous Dispersion State of Magnetic Nanoparticles: Experiments and Monte Carlo Simulation. J Clust Sci 34, 1975–1987 (2023). https://doi.org/10.1007/s10876-022-02360-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02360-0