Abstract
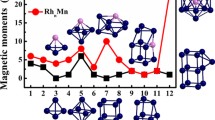
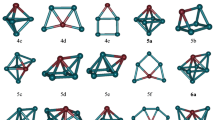
Using the density functional theory (DFT), the geometries, relative stabilities and magnetic properties of bimetallic RhnOs (n = 1–9) clusters have been investigated. The relative stability was analyzed by examining the binding energy, fragmentation energy, second-order differences of energies and HOMO–LUMO energy gaps. The obtained results indicate that RhOs, Rh3Os, Rh5Os and Rh7Os clusters are more stable than their neighboring clusters. In addition, the doping of the Os atom enhanced the stability of the Rh clusters. The chemical hardness and chemical potential show that RhOs cluster is less reactive, indicating that RhOs cluster is the most stable one among all the clusters. The magnetic properties calculations exhibited that total magnetic moments come mostly from the Rh atoms for RhnOs (n = 3–9) clusters, while the contribution of the Os atom is observed for RhOs and Rh2Os clusters. In addition, the d orbitals plays an important role in the magnetic moments of the RhnOs clusters.










Similar content being viewed by others
References
G. Schmid (1992). Chem. Rev. 92, 1709.
L. N. Lewis (1993). Chem. Rev. 93, 2693.
X. S. Xu, S. Y. Yin, R. Moro, and W. A. de Heer (2005). Phys. Rev. Lett. 95, 237209.
E. K. Parks, T. D. Klots, and S. J. Riley (1990). J. Chem. Phys. 92, 3813.
W. P. Halperin (1998). Rev. Mod. Phys. 58, 533.
A. Soltani and A. Boudjahem (2014). Comput. Theor. Chem. 1047, 6.
T. Yonezawa, K. Imamura, and N. Kimizuka (2001). Langmuir. 17, 4701.
J. Y. Zhang, Q. Fang, A. J. Kenyon, and I. W. Boyd (2003). Appl. Surf. Sci. 208–209, 364.
C. D. Dong and X. G. Gong (2008). Phys. Rev. B 78, 020409.
T. Teranishi and M. Miyake (1998). Chem. Mater. 10, 594.
K. R. Gopidas, J. M. Whitesell, and M. A. Fox (2003). Nano. Lett. 3, 1757.
K. B. Sidhpuria, H. A. Patel, P. A. Parikh, P. Bahadur, H. C. Bajaj, and R. V. Jasra (2009). Appl. Clay. Sci. 42, 386.
A. Sanchez, M. Fang, A. Ahmed, and R. A. Sanchez-Dolgado (2014). Appl. Catal. A-Gen. 477, 117.
C. H. Campos, E. Rosenberg, J. L. Fierro, B. F. Urbano, B. L. Rivas, C. C. Torres, and P. Reyes (2015). Appl. Catal. A-Gen. 489, 280.
A. Behr, Y. Brunsch, and A. Lux (2012). Tetrahedron. Lett. 53, 2680.
A. J. Bruss, M. A. Gelesky, G. Machado, and J. Dupont (2006). J. Mol. Catal. A: Chem. 252, 212.
Y. Izumi, K. Konishi, M. Tsukahara, D. M. Obaid, and K. I. Aika (2007). J. Phys. Chem. C 111, 10073.
D. Han, X. Li, H. Zhang, Z. Liu, G. Hu, and C. Li (2008). J. Mol. Catal. A: Chem. 283, 15.
T. J. Yoon, J. I. Kim, and J. K. Lee (2003). Inorg. Chim. Acta. 345, 228.
A. J. Cox, J. G. Louderback, and L. A. Bloomfield (1993). Phys. Rev. Lett. 71, 923.
R. D. Adams and X. Qu (1995). Organometallics 14, 4167.
C.-T. Au, C.-F. Ng, and M.-S. Liao (1999). J. Catal. 185, 12.
T. Zoberbier, et al. (2012). J. Am. Chem. Soc. 134, 3073.
T. W. Chamberlain, T. Zoberbier, J. Biskupek, A. Botos, U. Kaiser, and A. N. Khlobystov (2012). Chem. Sci. 3, 1919.
F. M. Mendes and M. Schmal (1997). Appl. Catal. A-Gen. 163, 153.
A. Trunschke, H. Ewald, D. Gutschick, H. Miessner, M. Skupin, B. Walther, and H. C. Bottcher (1989). J. Mol. Catal. 56, 95.
X. Yang, D. Chen, S. Liao, H. Song, Y. Li, Z. Fu, and Y. Su (2012). J. Catal. 291, 36.
S. Dennler, J. Morillo, and G. M. Pastor (2003). Surf. Sci. 532–535, 334.
J. H. Mokkath and G. M. Pastor (2012). Phys. Rev. B 85, 054407.
A. K. Srivastava and N. Misra (2014). Comput. Theor. Chem. 1047, 1.
J. Lv, X. Bai, J. F. Jia, X. H. Xu, and H. S. Wu (2012). Physica B. 407, 14.
J. Lv, F. Q. Zhang, X. H. Xu, and H. S. Wu (2009). Chem. Phys. 363, 65.
J. X. Yang, C. F. Wei, and J. J. Guo (2010). Physica. B 405, 4892.
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian, Inc. Wallingford CT, 2009.
Y. Zhao and D. G. Truhlar (2006). J. Chem. Phys. 125, 194101.
W. J. Stevens, H. Basch, and M. Krauss (1984). J. Chem. Phys 81, 6026.
N. S. Venkataramanan (2008). J. Mol. Struct. Theochem 856, 9.
J. Du, H. Wang, and G. Jiang (2007). J. Mol. Struct. Theochem 817, 47.
W. Bouderbala, A. Boudjahem, and A. Soltani (2014). Mol. Phys. 112, 1789.
F. A. Cotton, A. R. Chakravarty, D. A. Tocher, and T. A. Stephenson (1984). Inorg. Chim. Acta. 87, 115.
C. D. Tait, J. M. Garner, J. P. Collman, A. P. Sattelberger, and W. H. Woodruff (1989). J. Am. Chem. Soc. 111, 9072.
M. D. Morse (1986). Chem. Rev. 86, 1049.
Z. Wu, B. Han, Z. Dai, and P. Jin (2005). Chem. Phys. Lett. 403, 367.
J. Du, X. Sun, and H. Wang (2008). Int. J. Quant. Chem. 108, 1505.
K. Takahashi, S. Isobe, and S. Ohnuki (2013). Chem. Phys. Lett 555, 26.
K. A. Gingerich and D. L. Cocke (1972). J. Chem. Soc. Chem. Commun. 1, 536.
H. Wang, H. Haouari, R. Craig, Y. Liu, J. R. Lombardi, and D. M. Lindsay (1997). J. Chem. Phys. 106, 2101.
B. V. Reddy, S. K. Nayak, S. N. Khanna, B. K. Rao, and P. Jena (1999). Phys. Rev. B 59, 5214.
C. H. Chien, E. Blaisten-Barojas, and M. R. Pederson (1998). Phys. Rev. A 58, 2196.
Y. J. Xian, W. Cheng-Fu, and G. Jian-Jun (2010). Physica. B 405, 4892.
M. R. Beltrán, F. B. Zamudio, V. Chauhan, P. Sen, H. Wang, Y. J. Ko, and K. Bowen (2013). Eur. Phys. J. D 67, 63.
A. Soltani, A. Boudjahem, and M. Bettahar (2015). Int. J. Quantum. Chem.. doi:10.1002/qua.25038.
M. X. Chen and X. H. Yan (2008). J. Chem. Phys. 128, 174305.
R. G. Parr and W. Yang Density Functional Theory of Atoms and Molecules (Oxford, New York, 1989).
Acknowledgments
The authors would like to acknowledge Pr. Abdaoui Mohammed (Director of Applied Chemistry Laboratory).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is being retracted at the request of the authors. This work in the article was initiated under the direction of and with the support of two senior scientists (Prof. A.-G.Boudjahem and Dr. M. Chettibi) who were consulted about and are not in concurrence with publishing this paper. The conclusions of the paper are preliminary as the research is not finalized.
About this article
Cite this article
Soltani, A., Bouderbala, W. & Boudjahem, Ag. RETRACTED ARTICLE: First Principles Study of the Geometries, Relative Stabilities and Magnetic Properties of Bimetallic RhnOs (n = 1–9) Clusters. J Clust Sci 27, 715–731 (2016). https://doi.org/10.1007/s10876-016-0970-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-016-0970-6