Abstract
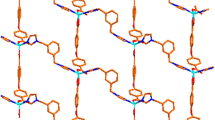
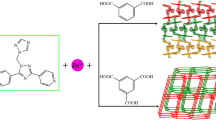
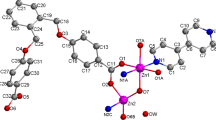
Two new Zn(II) coordination polymers based on bis(imidazol), namely, [Zn(hba)2(bib)]n(1) and [Zn(NAA)(bib)1.5]n·nNO3−·3nH2O (2) (Hhba = 3-hydroxybenzoic acid, HNAA = 1-naphthyl acetic acid, bib = 1,4-bis(imidazol-1-yl)-butane) have been successfully synthesized under hydrothermal conditions. Their structures have been determined by single crystal X-ray diffraction analyses, elemental analyses, IR, TG and fluorescence spectrums. Complex 1 shows a one-dimensional zigzag chain structure. Complex 2 exhibits two-dimensional network structure. The intermolecular hydrogen bonding and π–π stacking interactions extend the complexes 1 and 2 into supramolecular architectures and play an important role in stabilizing complexes 1 and 2. Furthermore, complex 1 exhibits intense blue luminescence and appears to be a good candidate for novel hybrid inorganic–organic photoactive materials, while no photoluminescent response was observed for complex 2. In addition, we analyzed the Natural Bond Orbitals (NBO) of 1 and 2 using the PBE0/LANL2DZ method in the Gaussian 09 Program. The calculations showed the obvious covalent interaction between the coordinated atoms and Zn(II) ion.
Graphic Abstract
Two new Zn(II) coordination polymers based on bis(imidazol), namely, [Zn(hba)2(bib)]n(1) and [Zn(NAA)(bib)1.5]n·nNO3−·3nH2O (2) have been successfully synthesized under hydrothermal conditions. Their structures have been determined by single crystal X-ray diffraction analyses, elemental analyses, IR, TG and fluorescence spectrum. Complex 1 shows a one-dimensional zigzag chain structure. Complex 2 exhibits two-dimensional network structure. The intermolecular hydrogen bonding and π–π stacking interactions extend the complex 1 and 2 into supramolecular architectures and play an important role in stabilizing complex 1 and 2. In addition, we analyzed Natural Bond Orbital (NBO) of 1 and 2 in using the PBE0/LANL2DZ method built in Gaussian 09 Program. The calculation results showed the obvious covalent interaction between the coordinated atoms and Zn(II) ion.












Similar content being viewed by others
References
Ma L, Wu CD, Wanderley MM, Lin W (2010) Angew Chem Int Ed 49:8244
Zhang ZQ, Huang RD, Xu YQ, Hu CW (2008) Chem J Chin Univ 29:1528
Liu GZ, Xin LY, Wang LY (2011) CrystEngComm 13:3013
Li GF, Wang YN, Wang QW, Li XM, Ji JY, Pan YR (2015) Chin J Inorg Chem 31:183
Sorace L, Benellib C, Gatteschi D (2011) Chem Soc Rev 40:3092
Jiang DY, Sui W, Li XM, Liu B, Wang QW, Pan YR (2016) Chin J Struct Chem 35:505
Dechambenoit P, Long JR (2011) Chem Soc Rev 40:3249
Hong MC, Zhao YJ, Su WP, Cao R, Fujita M, Zhou ZY, Chan ASC (2000) J Am Chem Soc 122:4819
Abrahams BF, Batten SR, Grannas MJ, Hamit H, Hoskins BF, Robson R (1999) Angew Chem Int Ed 38:1475
Bu XH, Chen W, Lu SL, Zhang RH, Liao DZ, Bu WM, Shionoya M, Brisse F, Ribas J (2001) Angew Chem Int Ed 40:3201
Qi Y, Che YX, Zheng JM (2008) CrystEngComm 10:1137
Aakeröy CB, Champness NR, Janiak C (2010) CrystEngComm 12:22
Kasai K, Aoyagi M, Fujita M (2000) J Am Chem Soc 122:2140
Du M, Bu XH, Guo YM, Ribas J, Diaz C (2002) Chem Commun 21:2550
Li XM, Pan YR, Ji JY, Niu YL, Niu QW (2014) J Inorg Organomet Polym 24:836
Pan YR, Sun M, Li XM (2015) Chin J Struct Chem 34:576
Tang YZ, Zhou M, Huang J, Tan YH, Wu JS, Wen HR (2013) Inorg Chem 52:1679
Sheldrick GM (1997) SHELXS-97, programs for X-ray crystal structure solution. University of Göttingen, Göttingen
Sheldrick GM (1997) SHELXL-97, programs for X-ray crystal structure refinement. University of Göttingen, Göttingen
Devereux M, Shea DO, Kellett A, McCann M, Walsh M, Egan D, Deegan C, Kedziora E, Rosair G, Müller-Bunz H (2007) Inorg Biochem 101:881
Kreno LE, Leong K, Farha OK, Allendorf M, Duyne RPV, Hupp JT (2012) Chem Rev 112:1105
Lin JD, Long XF, Lin P, Du SW (2010) Cryst Growth Des 10:146
Mohamed GG, El-Gamel NEA (2004) Spectrochim Acta A 60:3141
Li GL, Liu GZ, Huang LL, Li L, Zhang X (2014) J Inorg Organomet Polym 24:617
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Gaussian Inc., Wallingford
Parr RG, Yang W (1989) Density functional theory of atoms and molecules. Oxford University Press, Oxford
Ernzerhof M, Scuseria GE (1999) J Chem Phys 110:5029
Adamo C, Barone VJ (1999) Chem Phys 110:6158
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Perdew JP, Burke K, Ernzerhof M (1997) Phys Rev Lett 78:1396
Dunning TH, Hay PJ Jr (1976) In: Schaefer HF III (ed) Modern theoretical chemistry. Plenum, New York, p 1
Wang L, Zhao J, Ni L, Yao J (2012) J Inorg Gen Chem 638:224
Li ZP, Xing YH, Zhang YH (2009) Acta Phys Chim Sin 25:741
Acknowledgements
This work was supported by the Science and Technology Development Project of Jilin Provincial Science & Technology Department (Grant No. 201205080) and the Science and Technology Research Projects of the Education Department of Jilin Province (Grant No. 2013.384). Program supports from State Key Laboratory of Theoretical and Computational Chemistry of Tonghua Normal University are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, XM., Wang, ZT., Valtchev, V. et al. Syntheses, Crystal Structures and NBO Calculation of Two New Zinc(II) Coordination Polymers. J Chem Crystallogr 50, 155–163 (2020). https://doi.org/10.1007/s10870-019-00807-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-019-00807-3