Abstract
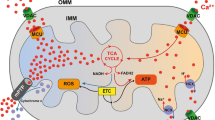
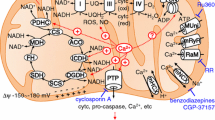
Calcium (Ca2+) plays diverse roles in all living organisms ranging from bacteria to humans. It is a structural element for bones, an essential mediator of excitation-contraction coupling, and a universal second messenger in the regulation of ion channel, enzyme and gene expression activities. In mitochondria, Ca2+ is crucial for the control of energy production and cellular responses to metabolic stress. Ca2+ uptake by the mitochondria occurs by the uniporter mechanism. The Mitochondrial Ca2+ Uniporter (MCU) protein has recently been identified as a core component responsible for mitochondrial Ca2+ uptake. MCU knockout (MCU KO) studies have identified a number of important roles played by this high capacity uptake pathway. Interestingly, this work has also shown that MCU-mediated Ca2+ uptake is not essential for vital cell functions such as muscle contraction, energy metabolism and neurotransmission. Although mitochondrial Ca2+ uptake was markedly reduced, MCU KO mitochondria still contained low but detectable levels of Ca2+. In view of the fundamental importance of Ca2+ for basic cell signalling, this finding suggests the existence of other currently unrecognized pathways for Ca2+ entry. We review the experimental evidence for the existence of alternative Ca2+ influx mechanisms and propose how these mechanisms may play an integral role in mitochondrial Ca2+ signalling.
Similar content being viewed by others
Reference list
Alavian KN, Beutner G, Lazrove E, Sacchetti S, Park HA, Licznerski P, Li H, Nabili P, Hockensmith K, Graham M, Porter GA Jr, Jonas EA (2014) An uncoupling channel within the c-subunit ring of the F1FO ATP synthase is the mitochondrial permeability transition pore. Proc. Natl. Acad. Sci.U.S.A 111:10580–10585
Baughman, J.M., Perocchi, F., Girgis, H.S., Plovanich, M., Belcher-Timme, C.A., Sancak,Y., Bao, X.R., Strittmatter, L., Goldberger, O., Bogorad, R.L., Koteliansky V., and Mootha, V.K. (2011): Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476(7360):341–345. doi:10.1038/nature10234
Bernardi P (1999) Mitochondrial transport of cations: channels, exchangers, and permeability transition. Physiol. Rev. 79:1127–1155
Beutner G, Sharma VK, Lin L, Ryu SY, Dirksen RT, Sheu SS (2005) Type 1 ryanodine receptor in cardiac mitochondria: transducer of excitation-metabolism coupling. Biochim. Biophys. Acta 1717:1–10
Bogeski I, Gulaboski R, Kappl R, Mirceski V, Stefova M, Petreska J, Hoth M (2011) Calcium binding and transport by coenzyme Q. J. Am. Chem. Soc. 133:9293–9303
Brookes PS, Parker N, Buckingham JA, Vidal-Puig A, Halestrap AP, Gunter TE, Nicholls DG, Bernardi P, Lemasters JJ, Brand MD (2008) UCPs--unlikely calcium porters. Nat. Cell Biol. 10:1235–1237
Cao C, Yudin Y, Bikard Y, Chen W, Liu T, Li H, Jendrossek D, Cohen A, Pavlov E, Rohacs T, Zakharian E (2013) Polyester modification of the mammalian TRPM8 channel protein: implications for structure and function. Cell Rep 4(2):302–315. doi:10.1016/j.celrep.2013.06.022
Chalmers S, Nicholls DG (2003) The relationship between free and total calcium concentrations in the matrix of liver and brain mitochondria. J. Biol. Chem. 278:19062–19070
Chaudhuri D, Sancak Y, Mootha VK, Clapham DE (2013) MCU encodes the pore conducting mitochondrial calcium currents. Elife. 2:e00704
Das S, Lengweiler UD, Seebach D, Reusch RN (1997) Proof for a nonproteinaceous calcium-selective channel in Escherichia coli by total synthesis from (R)-3-hydroxybutanoic acid and inorganic polyphosphate. Proc. Natl. Acad. Sci. U.S.A 94:9075–9079
De, S.D., Raffaello, A., Teardo, E., Szabo, I., and Rizzuto, R. (2011): A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature
De SD, Patron M, Rizzuto R (2015) Structure and function of the mitochondrial calcium uniporter complex. Biochim. Biophys. Acta 1853:2006–2011
Denton RM (2009) Regulation of mitochondrial dehydrogenases by calcium ions. Biochim. Biophys. Acta 1787:1309–1316
Dmitriev, O.Y., Jones, P.C., and Fillingame, R.H. (1999): Structure of the subunit c oligomer in the F1Fo ATP synthase: model derived from solution structure of the monomer and cross-linking in the native enzyme. Proc. Natl. Acad. Sci. U. S. A 96:7785–7790
Duchen MR (2000) Mitochondria and Ca(2+)in cell physiology and pathophysiology. Cell Calcium 28:339–348
Elustondo P, Zakharian E, Pavlov E (2012) Identification of the polyhydroxybutyrate granules in Mammalian cultured cells. Chem. Biodivers. 9:2597–2604
Elustondo PA, Angelova PR, Kawalec M, Michalak M, Kurcok P, Abramov AY, Pavlov EV (2013) Polyhydroxybutyrate targets mammalian mitochondria and increases permeability of plasmalemmal and mitochondrial membranes. PLoS. One 8:e75812
Elustondo PA, Nichols M, Negoda A, Thirumaran A, Zakharian E, Robertson GS, Pavlov EV (2016) Mitochondrial permeability transition pore induction is linked to formation of the complex of ATPase C-subunit, polyhydroxybutyrate and inorganic polyphosphate. Cell Death Dis. 2:160–170. doi:10.1038/cddiscovery.2016.70
Finkel T, Menazza S, Holmstrom KM, Parks RJ, Liu J, Sun J, Liu J, Pan X, Murphy E (2015) The ins and outs of mitochondrial calcium. Circ. Res. 116:1810–1819
Fritz, M.G., Walde, P., and Seebach, D. (1999): Oligoesters of (R)-3-hydroxybutanoic acid: transmembrane transport of Ca2+ across vesicle bilayers. Macromolecules 32:574–580
Greenawalt JW, Rossi CS, Lehninger AL (1964) Effect of active accumulation of calcium and phosphate ions on the structure of rat liver mitochondria. J. Cell Biol. 23:21–38
Griffiths EJ, Rutter GA (2009) Mitochondrial calcium as a key regulator of mitochondrial ATP production in mammalian cells. Biochim. Biophys. Acta 1787:1324–1333
Gunter TE, Gunter KK (2001) Uptake of calcium by mitochondria: transport and possible function. IUBMB. Life
Gunter TE, Sheu SS (2009) Characteristics and possible functions of mitochondrial Ca(2+) transport mechanisms. Biochim. Biophys. Acta 1787:1291–1308
Gunter, TE., Yule, DI., Gunter, KK., Eliseev, RA., and Salter, JD. (2004): Calcium and mitochondria. FEBS Lett 567:96–102
Hashimi H, McDonald L, Stribrna E, Lukes J (2013) Trypanosome Letm1 protein is essential for mitochondrial potassium homeostasis. J. Biol. Chem. 288:26914–26925
Hille, B. (1986): Ionic channels: molecular pores of excitable membranes. Harvey Lect, 82:47–69
Holmstrom KM, Pan X, Liu JC, Menazza S, Liu J, Nguyen TT, Pan H, Parks RJ, Anderson S, Noguchi A, Springer D, Murphy E, Finkel T (2015) Assessment of cardiac function in mice lacking the mitochondrial calcium uniporter. J. Mol. Cell Cardiol. 85:178–182
Hunter DR, Haworth RA (1979) The Ca2+-induced membrane transition in mitochondria. I. The protective mechanisms. Arch Biochem Biophys. 195(2):453–459
Jakob R, Beutner G, Sharma VK, Duan Y, Gross RA, Hurst S, Jhun BS, Uchi J, Sheu SS (2014) Molecular and functional identification of a mitochondrial ryanodine receptor in neurons. Neurosci. Lett. 575:7–12
Jendrossek D, Pfeiffer D (2014) New insights in the formation of polyhydroxyalkanoate granules (carbonosomes) and novel functions of poly(3-hydroxybutyrate). Environ. Microbiol. 16:2357–2373
Jiang D, Zhao L, Clapham DE (2009) Genome-wide RNAi screen identifies Letm1 as a mitochondrial Ca2+/H+ antiporter. Science 326:144–147
Jonas EA, Knox RJ, Kaczmarek LK (1997) Giga-ohm seals on intracellular membranes: a technique for studying intracellular ion channels in intact cells. Neuron 19:7–13
Jonas EA, Buchanan J, Kaczmarek LK (1999) Prolonged activation of mitochondrial conductances during synaptic transmission. Science 286:1347–1350
Jonas EA, Porter GA Jr, Beutner G, Mnatsakanyan N, Alavian KN (2015) Cell death disguised: the mitochondrial permeability transition pore as the c-subunit of the FF ATP synthase. Pharmacol Res 99:382–392. doi:10.1016/j.phrs.2015.04.013
Kane DA, Pavlov EV (2013) Calculation of ion currents across the inner membrane of functionally intact mitochondria. Channels (Austin.) 7:426–431
Kirichok Y, Krapivinsky G, Clapham DE (2004) The mitochondrial calcium uniporter is a highly selective ion channel. Nature 427:360–364
Kovacs-Bogdan E, Sancak Y, Kamer KJ, Plovanich M, Jambhekar A, Huber RJ, Myre MA, Blower MD, Mootha VK (2014) Reconstitution of the mitochondrial calcium uniporter in yeast. Proc. Natl. Acad. Sci. U.S.A 111:8985–8990
Marchi S, Pinton P (2014) The mitochondrial calcium uniporter complex: molecular components, structure and physiopathological implications. J. Physiol 592:829–839
McQuibban AG, Joza N, Megighian A, Scorzeto M, Zanini D, Reipert S, Richter C, Schweyen RJ, Nowikovsky K (2010) A Drosophila mutant of LETM1, a candidate gene for seizures in Wolf-Hirschhorn syndrome. Hum. Mol. Genet. 19:987–1000
Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191:144–148
Murgia M, Rizzuto R (2015) Molecular diversity and pleiotropic role of the mitochondrial calcium uniporter. Cell Calcium 58:11–17
Murphy E, Pan X, Nguyen T, Liu J, Holmstrom KM, Finkel T (2014) Unresolved questions from the analysis of mice lacking MCU expression. Biochem. Biophys. Res. Commun. 449:384–385
Negoda A, Xian M, Reusch RN (2007) Insight into the selectivity and gating functions of Streptomyces lividans KcsA. Proc. Natl. Acad. Sci. U.S.A, 104:4342–4346
Nicholls DG, Chalmers S (2004) The integration of mitochondrial calcium transport and storage. J. Bioenerg. Biomembr. 36:277–281
Nowikovsky K, Froschauer EM, Zsurka G, Samaj J, Reipert S, Kolisek M, Wiesenberger G, Schweyen RJ (2004) The LETM1/YOL027 gene family encodes a factor of the mitochondrial K+ homeostasis with a potential role in the Wolf-Hirschhorn syndrome. J. Biol. Chem. 279:30307–30315
O’Rourke B (2007) Mitochondrial ion channels. Annu. Rev. Physiol 69:19–49
Pan, S., Ryu, SY., and Sheu, SS. (2011): Distinctive characteristics and functions of multiple mitochondrial Ca2+ influx mechanisms. Sci.China Life Sci, 54:763–769
Pan X, Liu J, Nguyen T, Liu C, Sun J, Teng Y, Fergusson MM, Rovira II, Allen M, Springer DA, Aponte AM, Gucek M, Balaban RS, Murphy E, Finkel T (2013) The physiological role of mitochondrial calcium revealed by mice lacking the mitochondrial calcium uniporter. Nat. Cell Biol 15:1464–1472
Papageorgiou AC, Hermawan S, Singh CB, Jendrossek D (2008) Structural basis of poly(3-hydroxybutyrate) hydrolysis by PhaZ7 depolymerase from Paucimonas lemoignei. J. Mol. Biol. 382:1184–1194
Pardo B, Contreras L, Serrano A, Ramos M, Kobayashi K, Iijima M, Saheki T, Satrustegui J (2006) Essential role of aralar in the transduction of small Ca2+ signals to neuronal mitochondria. J. Biol. Chem. 281:1039–1047
Pavlov, E., Grimbly, C., Diao, C.T., and French, R.J. (2005a): A high-conductance mode of a poly-3-hydroxybutyrate/calcium/polyphosphate channel isolated from competent Escherichia coli cells. FEBS Lett, 579:5187–5192.
Pavlov E, Zakharian E, Bladen C, Diao CT, Grimbly C, Reusch RN, French RJ (2005b) A large, voltage-dependent channel, isolated from mitochondria by water-free chloroform extraction. Biophys. J. 88:2614–2625
Pendin D, Greotti E, Pozzan T (2014) The elusive importance of being a mitochondrial Ca(2+) uniporter. Cell Calcium 55:139–145
Perocchi F, Gohil VM, Girgis HS, Bao XR, McCombs JE, Palmer AE, Mootha VK (2010) MICU1 encodes a mitochondrial EF hand protein required for Ca(2+) uptake. Nature 467:291–296
Plovanich M, Bogorad RL, Sancak Y, Kamer KJ, Strittmatter L, Li AA, Girgis HS, Kuchimanchi S, De GJ, Speciner L, Taneja N, Oshea J, Koteliansky V, Mootha VK (2013) MICU2, a paralog of MICU1, resides within the mitochondrial uniporter complex to regulate calcium handling. PLoS. One 8:e55785
Reed KC, Bygrave FL (1974) The inhibition of mitochondrial calcium transport by lanthanides and ruthenium red. Biochem. J. 140:143–155
Reusch RN (1989) Poly-beta-hydroxybutyrate/calcium polyphosphate complexes in eukaryotic membranes. Proc. Soc. Exp. Biol. Med. 191:377–381
Reusch RN (1995) Low molecular weight complexed poly(3-hydroxybutyrate): a dynamic and versatile molecule in vivo. Can. J. Microbiol. 41(Suppl 1):50–54
Reusch RN (1999) Polyphosphate/poly-(R)-3-hydroxybutyrate) ion channels in cell membranes. Prog. Mol. Subcell. Biol 23:151–182
Reusch RN (2012) Physiological importance of poly-(R)-3-hydroxybutyrates. Chem. Biodivers. 9:2343–2366
Reusch RN, Sadoff HL (1988) Putative structure and functions of a poly-beta-hydroxybutyrate/calcium polyphosphate channel in bacterial plasma membranes. Proc. Natl. Acad. Sci. U.S.A 85:4176–4180
Reusch RN, Huang R, Bramble LL (1995) Poly-3-hydroxybutyrate/polyphosphate complexes form voltage-activated Ca2+ channels in the plasma membranes of Escherichia coli. Biophys. J. 69:754–766
Reusch, RN., Huang, R., and Kosk-Kosicka, D. (1997): Novel components and enzymatic activities of the human erythrocyte plasma membrane calcium pump. FEBS Lett, 412:592–596
Ryu, SY., Beutner, G., Dirksen, RT., Kinnally, KW., and Sheu, SS. (2010): Mitochondrial ryanodine receptors and other mitochondrial Ca2+ permeable channels. FEBS Lett, 584:1948–1955
Sancak Y, Markhard AL, Kitami T, Kovacs-Bogdan E, Kamer KJ, Udeshi ND, Carr SA, Chaudhuri D, Clapham DE, Li AA, Calvo SE, Goldberger O, Mootha VK (2013) EMRE is an essential component of the mitochondrial calcium uniporter complex. Science 342:1379–1382
Seebach D, Fritz MG (1999) Detection, synthesis, structure, and function of oligo(3-hydroxyalkanoates): contributions by synthetic organic chemists. Int. J. Biol. Macromol. 25:217–236
Seebach D, Brunner A, Burger HM, Schneider J, Reusch RN (1994) Isolation and 1H-NMR spectroscopic identification of poly(3-hydroxybutanoate) from prokaryotic and eukaryotic organisms. Determination of the absolute configuration (R) of the monomeric unit 3-hydroxybutanoic acid from Escherichia coli and spinach. Eur. J. Biochem. 224:317–328
Smithen M, Elustondo PA, Winkfein R, Zakharian E, Abramov AY, Pavlov E (2013) Role of polyhydroxybutyrate in mitochondrial calcium uptake. Cell Calcium 54:86–94
Solesio ME, Demirkhanyan L, Zakharian E, Pavlov EV (2016a) Contribution of inorganic polyphosphate towards regulation of mitochondrial free calcium. Biochim. Biophys. Acta 1860:1317–1325
Solesio, ME., Elustondo, PA., Zakharian, E., and Pavlov, EV. (2016b): Inorganic polyphosphate (polyP) as an activator and structural component of the mitochondrial permeability transition pore. Biochem.Soc.Trans, 44:7–12
Sparagna GC, Gunter KK, Sheu SS, Gunter TE (1995) Mitochondrial calcium uptake from physiological-type pulses of calcium. A description of the rapid uptake mode. J. Biol. Chem 270:27510–27515
Szabo, I. and Zoratti, M. (2014): Mitochondrial channels: ion fluxes and more. Physiol Rev, 94:519–608
Trenker M, Malli R, Fertschai I, Levak-Frank S, Graier WF (2007) Uncoupling proteins 2 and 3 are fundamental for mitochondrial Ca2+ uniport. Nat. Cell Biol. 9:445–452
Tsai MF, Jiang D, Zhao L, Clapham D, Miller C (2014) Functional reconstitution of the mitochondrial Ca2+/H+ antiporter Letm1. J. Gen. Physiol 143:67–73
Uribe S, Rangel P, Pardo JP (1992) Interactions of calcium with yeast mitochondria. Cell Calcium 13:211–217
Williams GS, Boyman L, Lederer WJ (2015) Mitochondrial calcium and the regulation of metabolism in the heart. J. Mol. Cell Cardiol. 78:35–45
Zakharian E, Reusch RN (2004) Functional evidence for a supramolecular structure for the Streptomyces lividans potassium channel KcsA. Biochem. Biophys. Res. Commun. 322:1059–1065
Zakharian E, Reusch RN (2007) Haemophilus influenzae outer membrane protein P5 is associated with inorganic polyphosphate and polyhydroxybutyrate. Biophys. J. 92:588–593
Zakharian E, Thyagarajan B, French RJ, Pavlov E, Rohacs T (2009) Inorganic polyphosphate modulates TRPM8 channels. PLoS. One 4:e5404
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elustondo, P.A., Nichols, M., Robertson, G.S. et al. Mitochondrial Ca2+ uptake pathways. J Bioenerg Biomembr 49, 113–119 (2017). https://doi.org/10.1007/s10863-016-9676-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-016-9676-6