Abstract
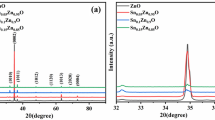
This paper reports the development of zinc oxide films electrodeposited at different potentials on tin-doped indium oxide substrates. The effect of deposition potential on ZnO microstructure, optical absorption, and photocatalytic activity for water splitting reaction were studied in detail. The films were potentiodynamically grown by applying different deposition potentials, such as − 0.7, − 0.8, and − 0.9 V at constant temperature (70 °C) for 30 min. The pH of precursor solution was maintained around 6 during the electrodeposition process. X-ray diffraction study revealed the hexagonal wurtzite crystal structure of the ZnO. The field emission scanning electron microscopy (FESEM) demonstrated a significant variation in the microstructure with changing deposition potential. UV–Visible spectroscopy demonstrated a significant change in the optical band gap values for the ZnO films deposited at different deposition potentials. The highest photocatalytic activity of water splitting was recorded for the films deposited at − 0.8 V under AM. 1.5 G solar light illumination.






Similar content being viewed by others
References
J. Zhu, D. Yang, Z. Yin, Q. Yan, H. Zhang, Small 10, 3480 (2014). https://doi.org/10.1002/smll.201303202
S. Liu, Z.R. Tang, Y. Sun, J.C. Colmenares, Y.J. Xu, Chem. Soc. Rev. 44, 5053 (2015). https://doi.org/10.1039/c4cs00408f
M. Ball, M. Weeda, Int. J. Hydrog. Energy 40, 7903 (2015). https://doi.org/10.1016/j.ijhydene.2015.04.032
H.B. Wu, B.Y. Xia, L. Yu, X.Y. Yu, X.W. Lou, Nat. Commun. 6, 6512 (2015). https://doi.org/10.1038/ncomms7512
S. Chen, S.S. Thind, A. Chen, Electrochem. Commun. 63, 10 (2016). https://doi.org/10.1016/j.elecom.2015.12.003
A. Eftekhari, V.J. Babu, S. Ramakrishna, Int. J. Hydrog. Energy (2017). https://doi.org/10.1016/j.ijhydene.2017.03.029
Y.K. Gaudy, S. Haussener, J. Mater. Chem. A 4, 3100 (2016). https://doi.org/10.1039/c5ta07328f
W. Ren, H. Zhang, C. Cheng, Electrochim. Acta 241, 316 (2017). https://doi.org/10.1016/j.electacta.2017.04.145
N. Iqbal, I. Khan, Z.H.A. Yamani, A. Qurashi, Sol. Energy 144, 604 (2017). https://doi.org/10.1016/j.solener.2017.01.057
P. Varadhan, H.C. Fu, D. Priante et al., Nano Lett. 17, 1520 (2017). https://doi.org/10.1021/acs.nanolett.6b04559
D. Cao, H. Xiao, J. Fang et al., Mater. Res. Express 4, 015019 (2017). https://doi.org/10.1088/2053-1591/aa56ee
L. Yang, M. Zhang, K. Zhu, J. Lv, G. He, Z. Sun, Appl. Surf. Sci. (2016). https://doi.org/10.1016/j.apsusc.2016.07.001
S. Banerjee, S.K. Mohapatra, M. Misra, J. Phys. Chem. C 115, 12643 (2011). https://doi.org/10.1021/jp106879p
N.D. Desai, S.S. Mali, R.M. Mane, V.B. Ghanwat, C.K. Hong, P.N. Bhosale, J. Mater. Sci.: Mater. Electron. 27, 11739 (2016). https://doi.org/10.1007/s10854-016-5312-9
Z. Liu, Q. Cai, C. Ma, J. Zhang, J. Liu, New J. Chem. (2017). https://doi.org/10.1039/c7nj01725a
P.R. Deshmukh, Y. Sohn, W.G. Shin, J. Alloys Compd. (2017). https://doi.org/10.1016/j.jallcom.2017.04.030
M.S. Islam, M.F. Hossain, S.M.A. Razzak, J. Photochem. Photobiol. A 326, 100 (2016). https://doi.org/10.1016/j.jphotochem.2016.04.002
T.D. Dongale, K.V. Khot, S.S. Mali et al., Mater. Sci. Semicond. Process. 40, 523 (2015). https://doi.org/10.1016/j.mssp.2015.07.004
K.V. Khot, S.S. Mali, R.M. Mane et al., J. Mater. Sci.: Mater. Electron. 26, 6897 (2015). https://doi.org/10.1007/s10854-015-3307-6
A. Mahmood, A. Naeem, InTech (2017). https://doi.org/10.5772/67857
F. Li, L. Yang, G. Xu et al., J. Alloys Compd. 577, 663 (2013). https://doi.org/10.1016/j.jallcom.2013.06.147
S. Agnihotri, G. Bajaj, S. Mukherji, S. Mukherji, Nanoscale 7, 7415 (2015). https://doi.org/10.1039/c4nr06913g
J. Jean, S. Chang, P.R. Brown et al., Adv. Mater. 25, 2790 (2013). https://doi.org/10.1002/adma.201204192
S.H. Chen, C.F. Yu, C.S. Chien, (2017) Microsc Res Tech. https://doi.org/10.1002/jemt.22848
S.K. Shaikh, S.I. Inamdar, V.V. Ganbavle, K.Y. Rajpure, J. Alloys Compd. 664, 242 (2016). https://doi.org/10.1016/j.jallcom.2015.12.226
V.K. Kaushik, C. Mukherjee, T. Ganguli, P.K. Sen, J. Alloys Compd. 689, 1028 (2016). https://doi.org/10.1016/j.jallcom.2016.08.022
R. Rayathulhan, B.K. Sodipo, AA Aziz, Ultrason. Sonochem. 35, 270 (2017). https://doi.org/10.1016/j.ultsonch.2016.10.002
G. Zhu, Y. Shen, K. Xu et al., J. Alloys Compd. 689, 192 (2016). https://doi.org/10.1016/j.jallcom.2016.07.182
S. Sampath, M. Shestakova, P. Maydannik et al., RSC Adv. 6, 25173 (2016). https://doi.org/10.1039/c6ra01655c
J. Laube, D. Nübling, H. Beh, S. Gutsch, D. Hiller, M. Zacharias, Thin Solid Films 603, 377 (2016). https://doi.org/10.1016/j.tsf.2016.02.060
K.G. Girija, K. Somasundaram, A. Topkar, R.K. Vatsa, J. Alloys Compd. 684, 15 (2016). https://doi.org/10.1016/j.jallcom.2016.05.125
N. Kıcır, T. Tüken, O. Erken, C. Gumus, Y. Ufuktepe, Appl. Surf. Sci. 377, 191 (2016). https://doi.org/10.1016/j.apsusc.2016.03.111
W. Riedel, Y. Tang, W. Ohm, J. Chen, M.C. Lux-Steiner, S. Gledhill, Thin Solid Films 574, 177 (2015). https://doi.org/10.1016/j.tsf.2014.12.006
J.K. Liang, H.L. Su, C.L. Kuo et al., Electrochim. Acta 125, 124 (2014). https://doi.org/10.1016/j.electacta.2014.01.029
F. Xu, Y. Lu, Y. Xie, Y. Liu, Mater. Des. 30, 1704 (2009). https://doi.org/10.1016/j.matdes.2008.07.024
L. Xu, Y. Guo, Q. Liao, J. Zhang, D. Xu, J. Phys. Chem. B 109, 13519 (2005)
R. Tena-Zaera, J. Elias, G. Wang, C. Lévy-Clément, J. Phys. Chem. C 111, 16706 (2007)
L. Vayssieres, K. Keis, S.E. Lindquist, A. Hagfeldt, J. Phys. Chem. B 105, 3350 (2001). https://doi.org/10.1021/jp010026s
A. Mahmood, F. Tezcan, G. Kardaş, Int. J. Hydrog. Energy (2017). https://doi.org/10.1016/j.ijhydene.2017.06.003
H. Li, Y. Fu, H. Liu et al., Inorg. Chem. Commun. 30, 182 (2013). https://doi.org/10.1016/j.inoche.2012.11.029
P. Paufler, Cryst. Res. Technol. 16, 982 (1981)
F. Xu, Y. Lu, Y. Xie, Y. Liu, J. Solid State Electrochem. 14, 63 (2009). https://doi.org/10.1007/s10008-009-0785-6
V. Kumar, N. Singh, R.M. Mehra, A. Kapoor, L.P. Purohit, H.C. Swart, Thin Solid Films 539, 161 (2013). https://doi.org/10.1016/j.tsf.2013.05.088
S. Xie, X. Lu, T. Zhai et al., J. Mater. Chem. 22, 14272 (2012)
Z. Han, L. Liao, Y. Wu, H. Pan, S. Shen, J. Chen, J. Hazard. Mater. 217, 100 (2012)
Y. Zheng, C. Chen, Y. Zhan et al., Inorg. Chem. 46, 6675 (2007)
A.B. Djurišić, Y.H. Leung, Small 2: 944 (2006)
P.C. Patel, S. Ghosh, P.C. Srivastava, Mater. Res. Bull. 81, 85 (2016). https://doi.org/10.1016/j.materresbull.2016.05.005
Y.F. Gao, M. Nagai, Y. Masuda, F. Sato, K. Koumoto, J. Cryst. Growth 286, 445 (2006). https://doi.org/10.1016/j.jcrysgro.2005.10.072
S. Srinivasan, Fuel Cells From Fundamentals To Applications (Springer, New York, 2006)
H. Gerischer, Solar Energy Conversion (Springer, Berlin, 1979), p. 115
M. Radecka, M. Rekas, A. Trenczek-Zajac, K. Zakrzewska, J. Power Sources 181, 46 (2008). https://doi.org/10.1016/j.jpowsour.2007.10.082
K. Gelderman, L. Lee, S.W. Donne, J. Chem. Educ. 84, 685 (2007)
K.S. Ahn, S. Shet, T. Deutsch et al., J. Power Sources 176, 387 (2008). https://doi.org/10.1016/j.jpowsour.2007.10.034
V. Ischenko, S. Polarz, D. Grote, V. Stavarache, K. Fink, M. Driess, Adv. Funct. Mater. 15, 1945 (2005)
Z. Chen, T.F. Jaramillo, T.G. Deutsch et al., J. Mater. Res. 25, 3 (2011). https://doi.org/10.1557/jmr.2010.0020
Acknowledgements
The authors acknowledge the financial support of the Scientific and Technological Research Council of Turkey under (TUBITAK- BİDEB) 2211- National Ph.D. Fellowship Programme and Scientific Research Project of Cukurova University (Project No: FDK-2014-3488).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dr. Gülfeza Kardaş is the first/primary corresponding author of the article.
Rights and permissions
About this article
Cite this article
Tezcan, F., Mahmood, A. & Kardaş, G. Enhanced photoelectrochemical activity of electrochemically deposited ZnO nanorods for water splitting reaction. J Mater Sci: Mater Electron 29, 9547–9554 (2018). https://doi.org/10.1007/s10854-018-8989-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-018-8989-0