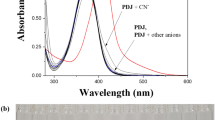
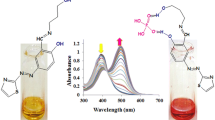
Abstract
A new and simple colorimetric receptor 1, based on the combination of 2-amino-4-methylphenol moiety and julolidine moiety, has been designed and synthesized. 1 showed a selective colorimetric sensing ability toward F− ion by changing color from yellow to orange, and could be utilized to monitor F− without any inhibition by competitive anions such as CH3COO− and CN−. Based on Job plot, ESI-mass spectrometry and the 1H NMR titration, the binding mode of 1 for F− was proposed to be 1:1. Moreover, the sensing mechanism for F− was theoretically supported by DFT and TD-DFT calculations.
Graphical Abstract











Similar content being viewed by others
References
Park, J.J., Kim, Y.-H., Kim, C., Kang, J.: Naked eye detection of fluoride and pyrophosphate with an anion receptor utilizing anthracene and nitrophenyl group as signaling group. Tetrahedron Lett. 52, 2759–2763 (2011)
Kim, Y.-J., Kwak, H., Lee, S.J., Lee, J.S., Kwon, H.J., Nam, S.H., Lee, K., Kim, C.: Urea/thiourea-based colorimetric chemosensors for the biologically important ions: efficient and simple sensors. Tetrahedron 62, 9635–9640 (2006)
Noh, J.Y., Hwang, I.H., Kim, H., Song, E.J., Kim, K.B., Kim, C.: Salicylimine-based colorimetric and fluorescent chemosensor for selective detection of cyanide in aqueous buffer. Bull. Korean Chem. Soc. 34, 1985–1989 (2013)
Miyaji, H., Sato, W., Sessler, J.: Naked-eye detection of anions in dichloromethane: colorimetric anion sensors based on calix. Angew. Chem. Int. Ed. Engl. 39, 1777–1780 (2000)
Kim, S.K., Sessler, J.L.: Ion pair receptors. Chem. Soc. Rev. 39, 3784–3809 (2010)
Aaseth, J., Shimshi, M., Gabrilove, J.L., Birketvedt, G.S.: Fluoride: a toxic or therapeutic agent in the treatment of osteoporosis. J. Trace Elem. Exp. Med. 17, 83–92 (2004)
Chen, J.-S., Zhou, P.-W., Zhao, L., Chu, T.-S.: A DFT/TDDFT study of the excited state intramolecular proton transfer based sensing mechanism for the aqueous fluoride chemosensor BTTPB. RSC Adv. 4, 254–259 (2014)
Chen, J.-S., Zhou, P.-W., Li, G.-Y., Chu, T.-S., He, G.-Z.: Fluoride anion sensing mechanism of 2-ureido-4[1H]-pyrimidinone quadruple hydrogen-bonded supramolecular assembly: photoinduced electron transfer and partial configuration change. J. Phys. Chem. B. 117, 5212–5221 (2013)
Mattiwala, N.M., Kamal, R., Sahoo, S.K.: Schiff base bis(5-nitrosalycilaldehyde)ethylenediamine as colorimetric sensor for fluoride. Res. Chem. Intermed. 41, 391–400 (2015)
Kim, Y., Gabbaï, F.P.: Cationic boranes for the complexation of fluoride ions in water below the 4 ppm maximum contaminant level. J. Am. Chem. Soc. 131, 3363–3369 (2009)
Broomsgrove, A.E.J., Addy, D.A., Di Paolo, A., Morgan, I.R., Bresner, C., Chislett, V., Fallis, I.A., Thompson, A.L., Vidovic, D., Aldridge, S.: Evaluation of electronics, electrostatics and hydrogen bond cooperativity in the binding of cyanide and fluoride by Lewis acidic ferrocenylboranes. Inorg. Chem. 49, 157–173 (2010)
Madhu, S., Ravikanth, M.: Boron-dipyrromethene based reversible and reusable selective chemosensor for fluoride detection. Inorg. Chem. 53, 1646–1653 (2014)
Tayade, K., Sahoo, S.K., Singh, A., Singh, N., Mahulikar, P., Attarde, S., Kuwar, A.: Architecture of dipodal ratiometric motif showing discrete nanomolar response towards fluoride ion. Sens. Actuators B Chem. 202, 1333–1337 (2014)
Wang, L., Fang, G., Cao, D.: A reversible and reusable selective chemosensor for fluoride detection using a phenolic OH-containing BODIPY dye by both colorimetric “naked-eye” and fluorometric modes. J. Fluoresc. 24, 1757–1766 (2014)
Bamesberger, A., Schwartz, C., Song, Q., Han, W., Wang, Z., Cao, H.: Rational design of a rapid fluorescent approach for detection of inorganic fluoride in MeCN-H2O: a new fluorescence switch based on N-aryl-1,8-naphthalimide. New J. Chem. 38, 884–888 (2014)
Song, E.J., Kim, H., Hwang, I.H., Kim, K.B., Kim, A.R., Noh, I., Kim, C.: A single fluorescent chemosensor for multiple target ions: Recognition of Zn2+ in 100% aqueous solution and F− in organic solvent. Sens. Actuators B Chem. 195, 36–43 (2014)
Anand, T., Sivaraman, G., Iniya, M., Siva, A., Chellappa, D.: Aminobenzohydrazide based colorimetric and “turn-on” fluorescence chemosensor for selective recognition of fluoride. Anal. Chim. Acta. 876, 1–8 (2015)
Peng, X., Wu, Y., Fan, J., Tian, M., Han, K.: Colorimetric and ratiometric fluorescence sensing of fluoride: tuning selectivity in proton transfer. J. Org. Chem. 70, 10524–10531 (2005)
Na, Y.J., Choi, Y.W., Yun, J.Y., Park, K.M., Chang, P.S., Kim, C.: Dual-channel detection of Cu2+ and F− with a simple Schiff-based colorimetric and fluorescent sensor. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 136, 1649–1657 (2015)
Sreenivasa Rao, K., Balaji, T., Prasada Rao, T., Babu, Y., Naidu, G.R.K.: Determination of iron, cobalt, nickel, manganese, zinc, copper, cadmium and lead in human hair by inductively coupled plasma-atomic emission spectrometry. Spectrochim. Acta Part B At. Spectrosc. 57, 1333–1338 (2002)
Sturgeon, R.E., Berman, S.S., Desaulniers, A., Russell, D.S.: Determination of iron, manganese, and zinc in seawater by graphite furnace atomic absorption spectrometry. Anal. Chem. 51, 2364–2369 (1979)
Gulaboski, R., Mireski, V., Scholz, F.: An electrochemical method for determination of the standard Gibbs energy of anion transfer between water and n-octanol. Electrochem. commun. 4, 277–283 (2002)
Yoshino, J., Kano, N., Kawashima, T.: Fluorescence properties of simple N-substituted aldimines with a B–N interaction and their fluorescence quenching by a cyanide ion. J. Org. Chem. 74, 7496–7503 (2009)
Divya, K.P., Sreejith, S., Balakrishna, B., Jayamurthy, P., Anees, P., Ajayaghosh, A.: A Zn2+-specific fluorescent molecular probe for the selective detection of endogenous cyanide in biorelevant samples. Chem. Commun. (Camb) 46, 6069–6071 (2010)
Yu, H., Fu, M., Xiao, Y.: Switching off FRET by analyte-induced decomposition of squaraine energy acceptor: A concept to transform “turn off” chemodosimeter into ratiometric sensors. Phys. Chem. Chem. Phys. 12, 7386–7391 (2010)
Lv, X., Liu, J., Liu, Y., Zhao, Y., Chen, M., Wang, P., Guo, W.: Rhodafluor-based chromo and fluorogenic probe for cyanide anion. Sens. Actuators B Chem. 158, 405–410 (2011)
Noh, J.Y., Park, G.J., Na, Y.J., Jo, H.Y., Lee, S.A., Kim, C.: A colorimetric “naked-eye” Cu(II) chemosensor and pH indicator in 100 % aqueous solution. Dalton Trans. 43, 5652–5656 (2014)
Vilar, R.: Anion-templated synthesis. Angew. Chem. Int. Ed. 42, 1460–1477 (2003)
Hijji, Y.M., Barare, B., Kennedy, A.P., Butcher, R.: Synthesis and photophysical characterization of a Schiff base as anion sensor. Sens. Actuators B Chem. 136, 297–302 (2009)
Helal, A., Thao, N.T.T., Lee, S.W., Kim, H.-S.: Thiazole-based chemosensor II: synthesis and fluorescence sensing of fluoride ions based on inhibition of ESIPT. J. Incl. Phenom. Macrocycl. Chem. 66, 87–94 (2010)
Noh, J.Y., Kim, S., Hwang, I.H., Lee, G.Y., Kang, J., Kim, S.H., Min, J., Park, S., Kim, C., Kim, J.: Solvent-dependent selective fluorescence assay of aluminum and gallium ions using julolidine-based probe. Dye. Pigment. 99, 1016–1021 (2013)
Maity, D., Manna, A.K., Karthigeyan, D., Kundu, T.K., Pati, S.K., Govindaraju, T.: Visible-near-infrared and fluorescent copper sensors based on julolidine conjugates: selective detection and fluorescence imaging in living cells. Chem. Eur. J. 17, 11152–11161 (2011)
Kumari, N., Jha, S., Bhattacharya, S.: Colorimetric probes based on anthraimidazolediones for selective sensing of fluoride and cyanide ion via intramolecular charge transfer. J. Org. Chem. 76, 8215–8222 (2011)
Udhayakumari, D., Velmathi, S.: Azo linked thiourea based effective dual sensor and its real samples application in aqueous medium. Sens. Actuators B Chem. 209, 462–469 (2015)
Benesi, H.A., Hildebrand, J.H.: A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 71, 2703–2707 (1949)
Tsui, Y.K., Devaraj, S., Yen, Y.P.: Azo dyes featuring with nitrobenzoxadiazole (NBD) unit: a new selective chromogenic and fluorogenic sensor for cyanide ion. Sens. Actuators B Chem. 161, 510–519 (2012)
Shenderovich, I.G., Limbach, H.-H., Smirnov, S.N., Tolstoy, P.M., Denisov, G.S., Golubev, N.S.: H/D isotope effects on the low-temperature NMR parameters and hydrogen bond geometries of (FH)2F− and (FH)3F− dissolved in CDF3/CDF2Cl. Phys. Chem. Chem. Phys. 4, 5488–5497 (2002)
Lee, J.J., Park, G.J., Choi, Y.W., You, G.R., Kim, Y.S., Lee, S.Y., Kim, C.: Detection of multiple analytes ( CN− and F− ) based on a simple pyrazine-derived chemosensor in aqueous solution : experimental and theoretical approaches. Sens. Actuators B. Chem. 207, 123–132 (2015)
Jo, T.G., Na, Y.J., Lee, J.J., Lee, M.M., Lee, S.Y., Kim, C.: A diaminomaleonitrile based selective colorimetric chemosensor for copper(II) and fluoride ions. New J. Chem. 39, 2580–2587 (2015)
Becke, A.D.: Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993)
Lee, C., Yang, W., Parr, R.G.: Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988)
Hay, P.J., Wadt, W.R.: Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 82, 270–283 (1985)
Hay, P.J., Wadt, W.R.: Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi. J. Chem. Phys. 82, 284–298 (1985)
Barone, V., Cossi, M.: Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J. Phys. Chem. A. 102, 1995–2001 (1998)
Cossi, M., Barone, V.: Time-dependent density functional theory for molecules in liquid solutions. J. Chem. Phys. 115, 4708–4717 (2001)
O’Boyle, N.M., Tenderholt, A.L., Langner, K.M.: Cclib: a library for package-independent computational chemistry algorithms. J. Comput. Chem. 29, 839–845 (2008)
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Montgomery, J.A. Jr., Vreven, T., Kudin, K.N., Burant, J.C., Millam, J.M., Iyengar, S.S., Tomasi, J., Barone, V., Mennucci, B., Cossi, M., Scalmani, G., Rega, N., Petersson, G.A., Nakatsuji, H., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Klene, M., Li, X. Knox, J.E., Hratchian, H.P., Cross, J.B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A.J., Cammi, R., Pomelli, C., Ochterski, J.W., Ayala, P.Y., Morokuma, K., Voth, G.A., Salvador, P., Dannenberg, J.J., Zakrzewski, V.G., Dapprich, S., Daniels, A.D., Strain, M.C., Farkas, O., Malick, D.K., Rabuck, A.D., Raghavachari, K., Foresman, J.B., Ortiz, J.V., Cui, Q., Baboul, A.G., Clifford, S., Cioslowski, J., Stefanov, B.B., Liu, G., Liashenko, A., Piskorz, P., Komaromi, I., Martin, R.L., Fox, D.J., Keith, T., Al-Laham, M.A., Peng, C.Y., Nanayakkara, A., Challacombe, M., Gill, P.M.W., Johnson, B., Chen, W., Wong, M.W., Gonzalez, C., Pople, J.A.: Gaussian 03, Revision D.01, Gaussian, Inc., Wallingford CT (2004)
Acknowledgments
Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2014R1A2A1A11051794 and NRF-2015R1A2A2A09001301) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ryu, H.H., Lee, Y.J., Kim, S.E. et al. A colorimetric F− chemosensor with high selectivity: experimental and theoretical studies. J Incl Phenom Macrocycl Chem 86, 111–119 (2016). https://doi.org/10.1007/s10847-016-0646-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-016-0646-8