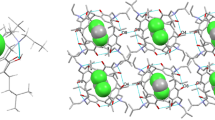
Abstract
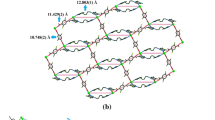
Co-crystallization of C-ethyl-calix[4]resorcinarene (1) and 4,4′-bipyridine (4,4′-bipy) from ethanol in the presence of ferrocene (FcH) yielded a multi-component complex FcH@1·2(4,4′-bipy) (3) that consists of a one-dimensional wave-like polymer, 1·2(4,4′-bipy), in which the cavity of 1 is deepened supramolecularly to enclose ferrocene as a guest. Heating mixture of C-methyl-calix[4]resorcinarene (2), trans-1,4-bis-(pyridyl)ethylene (bpe) and FcH in ethanol led to isolation of a similar host–guest complex FcH@2·2(bpe)·0.5C6H14 (4) in which the FcH guests have been assembled within a wave-like framework (2)·2(bpe).
Graphical Abstract
Ferrocene acts as the guest molecule for the self-assembly of the 1:2:1 C-ethyl-calix[4]resorcinarene/4,4′-bipyridine/ferrocene and the 1:2:1 C-methyl-calix[4]resorcinarene/trans-1,4-bis(pyridyl)ethylene/ferrocene supramolecular co-crystals.



Similar content being viewed by others
References
MacGillivray, L.R., Atwood, J.L.: A chiral spherical molecular assembly held together by 60 hydrogen bonds. Nature 389, 469–472 (1997)
MacGillivray, L.R., Atwood, J.L.: Structural classification and general principles for the design of spherical molecular hosts. Angew. Chem. Int. Ed. Engl. 38, 1018–1033 (1999)
Atwood, J.L., Barbour, L.J., Jerga, A.: Hydrogen-bonded molecular capsules are stable in polar media. Chem. Commun. 22, 2376–2377 (2001)
Cave, G.W.V., Ferrarelli, M.C., Atwood, J.L.: Nano-dimensions for the pyrogallol[4]arenecavity. Chem. Commun 22, 2787–2789 (2005)
Ma, B.Q., Zhang, Y.G., Coppens, P.: Structural variation and supramolecular isomerism in the C-methylcalix[4]resorcinarene/bipyridine system. Cryst. Growth Des. 2, 7–13 (2002)
Brown, P.O., Enright, G.D., Ripmeester, J.A.: Cationic guests in extended anionic C-methylcalix[4]resorcinarene-inorganic frameworks: exercising conformational control over C-methylcalix[4]resorcinarene. J. Supramol. Chem. 2, 497–500 (2002)
MacGillivray, L.R., Holman, K.T., Atwood, J.L.: Hydrogen bonds assist the organization of up to 11 guests within self-assembling cavities of nanometer dimensions. J. Supramol. Chem. 1, 125–130 (2001)
MacGillivray, L.R., Atwood, J.L.: Rational design of multicomponent calix[4]arenes and control of their alignment in the solid state. J. Am. Chem. Soc. 119, 6931–6932 (1997)
Nakamura, A., Sato, T., Kuroda, R.: Novel layered structures of the crystals based on C-methylcalix[4]resorcinarene formed at high temperature. CrystEngComm 5, 318–325 (2003)
Ma, B.Q., Coppens, P.: Transformation of a C-methylcalix[4]resorcinarene-based host–guest complex from a wave-like to a novel triangular brick-wall architecture. Chem. Commun. 4, 504–505 (2003)
Ma, B.Q., Coppens, P.: Three-fold interpenetrating three-dimensional networks based on C-methylcalix[4]resorcinarene incorporating benzophenone guest molecules. Chem. Commun. 3, 412–413 (2003)
MacGillivray, L.R., Reid, J.L., Ripmeester, J.A.: Conformational isomerism leads to supramolecular isomerism and nanoscale inclusion in 2D extended framework solids based on C-methylcalix[4]resorcinarene. Chem. Commun 11, 1034–1035 (2001)
Zhang, Y.G., Kim, C.D., Coppens, P.: Does C-methylcalix[4]resorcinarene always adopt the crown shape conformation? a resorcinarene/bipyridine/decamethylruthenocene supramolecular clathrate with a novel framework structure. Chem. Commun. 23, 2299–2300 (2000)
MacGillivray, L.R., Diamente, P.R., Reid, J.L., Ripmeester, J.A.: Encapsulation of two aromatics by a carcerand-like capsule of nanometre-scale dimensions. Chem. Commun. 5, 359–360 (2000)
Ma, B.Q., Coppens, P.: Variable conformation of benzophenone in a series of resorcinarene-based supramolecular frameworks. Cryst. Growth Des. 4, 1377–1385 (2001)
MacGillivray, L.R., Papaefstathiou, G.S., Reid, J.L., Ripmeester, J.A.: A rod-shaped guest leads to architectural isomerism in a multicomponent crystalline framework based on a resorcin[4]arene. Cryst. Growth Des. 1, 373–375 (2001)
Ma, B.Q., Zhang, Y.G., Coppens, P.: Multiple structures in supramolecular solids: Benzophenone embedded in three different C-methylcalix[4]resorcinarene/bipyridine frameworks. Cryst. Growth Des. 1, 271–275 (2001)
Ma, B.Q., Zhang, Y.G., Coppens, P.: The chair conformation of C-methylcalix[4]-resorcinarene in a novel, stepped, supramolecular framework. CrystEngComm 3, 78–80 (2001)
MacGillivray, L.R., Reid, J.L., Ripmeester, J.A.: Solvent provides a trap for the guest-induced formation of 1D host frameworks based upon supramolecular, deep-cavity resorcin[4]arenes. CrystEngComm 1, 1–4 (1999)
Colquhoun, H.M., Stoddart, J.F., Williams, D.J.: Second-sphere coordination—a novel rǒle for molecular receptors. Angew. Chem. Int. Ed. Engl. 25, 487–507 (1986)
Klingert, B., Rihs, G.: Molecular encapsulation of transition-metal complexes in cyclodextrins. 1. Synthesis and x-ray crystal structure of [(η5-C5H5)Fe(η6-C6H6)]PF6.2α-CD·8H2O. Organometallics 9, 1135–1141 (1990)
Klingert, B., Rihs, G.: Molecular encapsulation of transition metal complexes in cyclodextrins. Part 2. Synthesis and crystal structures of 2:1 adducts between α-cyclodextrin and metallocenium hexafluorophosphates [η5-C5H5)2 M]PF6 (M=Fe Co, Rh). J. Incl. Phenom. 10, 255–265 (1991)
Klingert, B., Rihs, G.: Molecular encapsulation of transition metal complexes in cyclodextrins. Part 3. Structural consequences of varying the guest geometry in channel-type inclusion compounds. J. Chem. Soc. Dalton Trans. 6, 2749–2760 (1991)
Odagaki, Y., Hirotsu, K., Higuchi, T., Harada, A., Takahashi, S.: X-Ray structure of the α-cyclodextrin–ferrocene (2 : 1) inclusion compound. J. Chem. Soc., Perkin Trans. 1, 1230–1231 (1990)
MacGillivray, L.R., Spinney, H.A., Reid, J.L., Ripmeester, J.A.: Entrapment of ferrocenes within supramolecular, deep-cavity resorcin[4]arenes. Chem. Commun. 6, 517–518 (2000)
Friščič, T., MacGillivray, L.R.: Double inclusion of ferrocene within a doubly interpenetrated three-dimensional framework based on a resorcin[4]arene. J. Organomet. Chem. 666, 43–48 (2003)
Hardie, M.J.: Solid state confinement of ferrocene by calixarenes. Supramol. Chem. 14, 7–10 (2002)
Cram, D.J., Karbach, S., Kim, H.E., Knobler, C.B., Maverick, E.F., Ericson, J.L., Helgeson, R.C.: Host-guest complexation. 46. Cavitands as open molecular vessels form solvates. J. Am. Chem. Soc. 110, 2229–2237 (1988)
SMART and SAINT + for Windows NT Version 6.02a. Bruker Analytical X-ray Instruments Inc., Madison, Wisconsin, USA (1998)
Sheldrick, G.M.: SADABS. University of Göttingen, Germany (1996)
Sheldrick, G.M.: SHELXTL Software Reference Manual. Version 5.1. Bruker AXS Inc., Madison, USA (1997)
Sheldrick, G.M.: A short history of SHELX. Acta Crystallogr. A 64, 112–122 (2008)
Acknowledgments
This project was supported by Natural Science Foundation of China (90922008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, YJ., Liu, JL., Yang, HM. et al. Inclusion of ferrocene within the deepened cavities formed from the hydrogen bonding self-assembly of calix[4]resorcinarenes with bis-pyridines. J Incl Phenom Macrocycl Chem 85, 105–110 (2016). https://doi.org/10.1007/s10847-016-0609-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-016-0609-0