Abstract
Purpose
The cystic fibrosis transmembrane conductance regulator (CFTR) is the most common causative gene attributed to congenital obstructive azoospermia (OA). The aim of this study was to conduct an epidemiological survey of congenital OA patients, to screen for CFTR mutations, and to follow their pregnancy outcomes in assisted reproductive technology (ART).
Methods
This cohort study enrolled congenital OA patients undergoing ART and whole-exome sequencing from January 2018 to September 2023. Semen parameters, sex hormones, and seminal plasma biochemistry were evaluated. CFTR mutations identified in OA patients were analyzed. In addition, the laboratory outcomes, clinical outcomes, and neonatal outcomes were compared between OA patients carrying two CFTR mutations and the others after surgical sperm extraction-intracytoplasmic sperm injection (ICSI) treatment.
Results
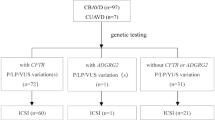
A total of 76 patients with congenital OA were enrolled. CFTR mutations were identified in 35 (46.1%) congenital OA patients. A total of 60 CFTR mutation sites of 27 types were identified, and 10 of them were novel. The average frequency was 1.71 (60/35) per person. The most common mutation was c.1210-11T > G (25%, 15/60). After ICSI treatment, there were no statistically significant differences in laboratory outcomes, clinical outcomes, and neonatal outcomes between OA patients carrying two CFTR mutations (n = 25) and other OA patients (n = 51).
Conclusion
Apart from the IVS9-5T mutation, the genetic mutation pattern of CFTR in Chinese OA patients is heterogeneous, which is significantly different from that of Caucasians. Although carrying two CFTR mutations or not had no effect on the pregnancy outcomes in OA patients after ICSI, genetic counseling is still recommended for such patients.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Jarow JP, Espeland MA, Lipshultz LI. Evaluation of the azoospermic patient. J Urol. 1989;142:62–5.
Wosnitzer M, Goldstein M, Hardy MP. Review of azoospermia. Spermatogenesis. 2014;4: e28218.
Lotti F, Maggi M. Ultrasound of the male genital tract in relation to male reproductive health. Hum Reprod Update. 2015;21:56–83.
Yu J, Chen Z, Ni Y, Li Z. CFTR mutations in men with congenital bilateral absence of the vas deferens (CBAVD): a systemic review and meta-analysis. Human Reprod (Oxford, England). 2012;27:25–35.
Jentsch TJ, Stein V, Weinreich F, Zdebik AA. Molecular structure and physiological function of chloride channels. Physiol Rev. 2002;82:503–68.
Fedder J, Crüger D, Oestergaard B, Petersen GB. Etiology of azoospermia in 100 consecutive nonvasectomized men. Fertil Steril. 2004;82:1463–5.
de Souza DAS, Faucz FR, Pereira-Ferrari L, Sotomaior VS, Raskin S. Congenital bilateral absence of the vas deferens as an atypical form of cystic fibrosis: reproductive implications and genetic counseling. Andrology. 2018;6:127–35.
Wang H, An M, Liu Y, Hu K, Jin Y, Xu S, et al. Genetic diagnosis and sperm retrieval outcomes for Chinese patients with congenital bilateral absence of vas deferens. Andrology. 2020;8:1064–9.
Feng J, Zhang Y, Yang X, Zhang Y. Heterogeneous spectrum of CFTR gene mutations in Chinese patients with CAVD and the dilemma of genetic blocking strategy. Reproduction. 2022;164:R47-r56.
Cheng H, Yang S, Meng Q, Zheng B, Gu Y, Wang L, et al. Genetic analysis and intracytoplasmic sperm injection outcomes of Chinese patients with congenital bilateral absence of vas deferens. J Assist Reprod Genet. 2022;39:719–28.
Minhas S, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, et al. European Association of Urology Guidelines on male sexual and reproductive health: 2021 update on male infertility. Eur Urol. 2021;80:603–20.
Li J, Chen J, Tian S, Jiao T, Wang J, Wei Y, et al. Effects of different sperm sources on the clinical outcomes of in vitro oocyte maturation cycles combined with intracytoplasmic sperm injection. Front Endocrinol (Lausanne). 2023;14:1115210.
Wang M, Yang QY, Zhou JP, Tan HP, Hu J, Jin L, et al. Novel compound heterozygous mutations in DNAH1 cause primary infertility in Han Chinese males with multiple morphological abnormalities of the sperm flagella. Asian J Androl. 2023;25:512–9.
Wang M, Jiang J, Xi Q, Li D, Ren X, Li Z, et al. Repeated cryopreservation process impairs embryo implantation potential but does not affect neonatal outcomes. Reprod Biomed Online. 2021;42:75–82.
Wang M, Yang Q, Ren X, Hu J, Li Z, Long R, et al. Investigating the impact of asymptomatic or mild SARS-CoV-2 infection on female fertility and in vitro fertilization outcomes: a retrospective cohort study. EClinicalMedicine. 2021;38: 101013.
Wang M, Yang Q, Zhu L, Jin L. Investigating impacts of CoronaVac vaccination in males on in vitro fertilization: a propensity score matched cohort study. World J Mens Health. 2022;40:570–9.
Choi JY, Muallem D, Kiselyov K, Lee MG, Thomas PJ, Muallem S. Aberrant CFTR-dependent HCO3- transport in mutations associated with cystic fibrosis. Nature. 2001;410:94–7.
Zondek LH, Zondek T. Normal and abnormal development of the epididymis of the fetus and infant. Eur J Pediatr. 1980;134:39–44.
Zielenski J, Tsui LC. Cystic fibrosis: genotypic and phenotypic variations. Annu Rev Genet. 1995;29:777–807.
Wang Y, Wrennall JA, Cai Z, Li H, Sheppard DN. Understanding how cystic fibrosis mutations disrupt CFTR function: from single molecules to animal models. Int J Biochem Cell Biol. 2014;52:47–57.
Bombieri C, Claustres M, De Boeck K, Derichs N, Dodge J, Girodon E, et al. Recommendations for the classification of diseases as CFTR-related disorders. J Cyst Fibros. 2011;10(Suppl 2):S86-102.
Luo S, Feng J, Zhang Y, Yang X, Ma G, Hu T, et al. Mutation analysis of the cystic fibrosis transmembrane conductance regulator gene in Chinese congenital absence of vas deferens patients. Gene. 2021;765: 145045.
Feng J, Wu X, Zhang Y, Yang X, Ma G, Chen S, et al. A novel mutation (-195C>A) in the promoter region of CFTR gene is associated with Chinese congenital bilateral absence of vas deferens (CBAVD). Gene. 2019;719: 144007.
Cai H, Qing X, Niringiyumukiza JD, Zhan X, Mo D, Zhou Y, et al. CFTR variants and renal abnormalities in males with congenital unilateral absence of the vas deferens (CUAVD): a systematic review and meta-analysis of observational studies. Genet Med. 2019;21:826–36.
Mieusset R, Bieth E, Daudin M, Isus F, Delaunay B, Bujan L, et al. Male partners of infertile couples with congenital unilateral absence of the vas deferens are mainly non-azoospermic. Andrology. 2020;8:645–53.
Li H, Wen Q, Li H, Zhao L, Zhang X, Wang J, et al. Mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) in Chinese patients with congenital bilateral absence of vas deferens. J Cyst Fibros. 2012;11:316–23.
Yang X, Sun Q, Yuan P, Liang H, Wu X, Lai L, et al. Novel mutations and polymorphisms in the CFTR gene associated with three subtypes of congenital absence of vas deferens. Fertil Steril. 2015;104:1268-75.e1-2.
Bai S, Du Q, Liu X, Tong Y, Wu B. The detection and significance of cystic fibrosis transmembrane conductance regulator gene promoter mutations in Chinese patients with congenital bilateral absence of the vas deferens. Gene. 2018;672:64–71.
Yuan P, Liang ZK, Liang H, Zheng LY, Li D, Li J, et al. Expanding the phenotypic and genetic spectrum of Chinese patients with congenital absence of vas deferens bearing CFTR and ADGRG2 alleles. Andrology. 2019;7:329–40.
Sharma N, Acharya N, Singh SK, Singh M, Sharma U, Prasad R. Heterogenous spectrum of CFTR gene mutations in Indian patients with congenital absence of vas deferens. Hum Reprod (Oxford, England). 2009;24:1229–36.
Sharma H, Mavuduru RS, Singh SK, Prasad R. Heterogeneous spectrum of mutations in CFTR gene from Indian patients with congenital absence of the vas deferens and their association with cystic fibrosis genetic modifiers. Mol Hum Reprod. 2014;20:827–35.
Kerem B, Rommens JM, Buchanan JA, Markiewicz D, Cox TK, Chakravarti A, et al. Identification of the cystic fibrosis gene: genetic analysis. Science. 1989;245:1073–80.
Casals T, Bassas L, Egozcue S, Ramos MD, Giménez J, Segura A, et al. Heterogeneity for mutations in the CFTR gene and clinical correlations in patients with congenital absence of the vas deferens. Hum Reprod (Oxford, England). 2000;15:1476–83.
Field PD, Martin NJ. CFTR mutation screening in an assisted reproductive clinic. Aust N Z J Obstet Gynaecol. 2011;51:536–9.
Girardet A, Ishmukhametova A, Willems M, Coubes C, Hamamah S, Anahory T, et al. Preimplantation genetic diagnosis for cystic fibrosis: the Montpellier center’s 10-year experience. Clin Genet. 2015;87:124–32.
Xu WM, Shi QX, Chen WY, Zhou CX, Ni Y, Rowlands DK, et al. Cystic fibrosis transmembrane conductance regulator is vital to sperm fertilizing capacity and male fertility. Proc Natl Acad Sci U S A. 2007;104:9816–21.
McBride JA, Kohn TP, Mazur DJ, Lipshultz LI, Coward RM. Sperm retrieval and intracytoplasmic sperm injection outcomes in men with cystic fibrosis disease versus congenital bilateral absence of the vas deferens. Asian J Androl. 2021;23:140–5.
Lu S, Cui Y, Li X, Zhang H, Liu J, Kong B, et al. Association of cystic fibrosis transmembrane-conductance regulator gene mutation with negative outcome of intracytoplasmic sperm injection pregnancy in cases of congenital bilateral absence of vas deferens. Fertil Steril. 2014;101:1255–60.
Meniru GI, Gorgy A, Podsiadly BT, Craft IL. Results of percutaneous epididymal sperm aspiration and intracytoplasmic sperm injection in two major groups of patients with obstructive azoospermia. Hum Reprod (Oxford, England). 1997;12:2443–6.
Kamal A, Fahmy I, Mansour R, Serour G, Aboulghar M, Ramos L, et al. Does the outcome of ICSI in cases of obstructive azoospermia depend on the origin of the retrieved spermatozoa or the cause of obstruction? A comparative analysis. Fertil Steril. 2010;94:2135–40.
Glina S, Fragoso JB, Martins FG, Soares JB, Galuppo AG, Wonchockier R. Percutaneous epididymal sperm aspiration (PESA) in men with obstructive azoospermia. Int Braz J Urol. 2003;29:141–5. discussion 5-6.
Funding
This study was supported by the National Key Research and Development Project (2021YFC2700603 and 2022YFC2702503) and the Key Research of Huazhong University of Science and Technology, Tongji Hospital (2022A20).
Author information
Authors and Affiliations
Contributions
Conceptualization: LZ and LJ. Sample collection: RL, RM, XW, and LG. Experiment conduction: MW, JZ. Formal analysis: MW, JZ. Funding acquisition: LZ and LJ. Writing—original draft: MW. Writing—review and editing: LZ, YC, and LJ.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Ethical Committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology with written informed consent provided by the participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Zhou, J., Long, R. et al. An overview of CFTR mutation profiles and assisted reproductive technology outcomes in Chinese patients with congenital obstructive azoospermia. J Assist Reprod Genet 41, 505–513 (2024). https://doi.org/10.1007/s10815-023-03004-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-023-03004-6