Abstract
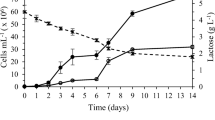
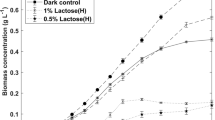
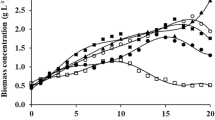
β-Galactosidase is a commercially important enzyme widely used in the food industry for the manufacturing of lactose-hydrolyzed products and synthesis of probiotic food ingredients. It could also be used to reduce the environmental impacts of the dairy industry relative to lactose disposal. This enzyme has been isolated from different sources, with varying properties and potential for diverse applications. Several microalgae have been screened for β-galactosidase activity, among which the chlorophyte Tetradesmus obliquus showed significant levels. The production of enzymes from microalgae could emerge as a valuable avenue for the utilization of its biomass. In addition, this particular microalga can grow mixotrophically on lactose as an organic carbon source, offering additional possibilities for the utilization of lactose. This study investigates the production of β-galactosidase from T. obliquus under different trophic conditions and known media composition, in order to assess their influence on its productivity and selectivity. Results show that the photoautotrophic cultures provide a highest selectivity, while mixotrophic conditions provide higher productivities due to faster growth and higher biomass yields. Further studies on mixotrophic cultures using lactose revealed no significant differences on β-galactosidase production when varying the concentrations of organic carbon and nitrogen nutrients. The age of culture has a strong influence on the enzyme production, suggesting a dependence on the growth phase. Maximal enzyme productivities obtained in mixotrophic conditions on lactose reach about 12.35 U L−1 day−1 after 7 days, which is a realistic duration for producing the enzyme at larger scales in bioreactors.





Similar content being viewed by others
References
Brasil BSAF, De Siqueira FG, Salum TFC, Zanette CM, Spier MR (2017) Microalgae and cyanobacteria as enzyme biofactories. Algal Res 25:76–89
Deschênes J-S, Boudreau A, Tremblay R (2015) Mixotrophic production of microalgae in pilot-scale photobioreactors: practicability and process considerations. Algal Res 10:80–86
Dickson RC, Markin J (1980) Physiological studies of β-galactosidase induction in Kluyveromyces lactis. J Bacteriol 142:777–785
Girard J-M, Deschênes J-S, Tremblay R, Gagnon J (2013) FT-IR/ATR univariate and multivariate calibration models for in situ monitoring of sugars in complex microalgal culture media. Bioresour Technol 144:664–668
Girard J-M, Roy M-L, Ben Hafsa M, Gagnon J, Faucheux N, Heitz M, Tremblay R, Deschênes J-S (2014) Mixotrophic cultivation of green microalgae Scenedesmus obliquus on cheese whey permeate for biodiesel production. Algal Res 5:241–248
González Siso MI (1996) The biotechnological utilization of cheese whey: a review. Bioresour Technol 57:1–11
Haider T, Husain Q (2007) Preparation of lactose free milk by using ammonium sulphate fractionated proteins from almonds. J Sci Food Agric 87:1278–1283
Jacob F, Monod J (1961) Genetic regulatory mechanisms in the synthesis of proteins. J Mol Biol 3:318–356
Kazemi S, Khayati G, Faezi-Ghasemi M (2016) β-Galactosidase production by Aspergillus niger ATCC 9142 using inexpensive substrates in solid-state fermentation: optimization by orthogonal arrays design. Iran Biomed J 20.5:287–294
Khabarova Y, Tornianen S, Tuomisto S, Järvelä I, Karhunen P, Isokoski M, Mattila K (2011) Lactase non-persistent genotype influences milk consumption and gastrointestinal symptoms in Northern Russians. BMC Gastroenterol 11:124
Kim JW, Rajagopal SN (2000) Isolation and characterization of β-galactosidase from Lactobacillus crispatus. Folia Microbiol 45:29–34
Law J, Lee S, Tseng A, Tsui KW, Yu N (2002) The role of glycerol and isopropyl thiogalactoside in Escherichia coli growth and lactose induction of β-galactosidase. J Exp Microbiol Immunol 2:97–102
Miksch G, Dobrowolski P (1995) Growth phase-dependent induction of stationary-phase promoters of Escherichia coli in different gram-negative bacteria. J Bacteriol 177:5374–5378
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor
Mlichova Z, Rosenberg M (2006) Current trends of β-galactosidase application in food technology. J Food Nutr Res 45:47–54
Nagy Z, Keresztessy Z, Szentirmai A, Biro S (2001) Carbon source regulation of β-galactosidase biosynthesis in Penicillium chrysogenum. J Basic Microbiol 41:351–362
Panesar PS, Kumari S, Panesar R (2010) Potential applications of immobilized β-galactosidase in food processing industries. Enzyme Res 2010:473137
Perini BLB, Souza HCM, Kelbert M, Apati GP, Pezzin APT, Schneider ALS (2013) Production of β-galactosidase from cheese whey using Kluyveromyces marxianus CBS 6556. Chem Eng Trans 32:991–996
Rosolen MD, Gennari A, Volpato G, De Souza CFV (2015) Lactose hydrolysis in milk and dairy whey using microbial β-galactosidases. Enzyme Res 2015:806240
Shaukat A, Levitt MD, Taylor BC, MacDonald R, Shamliyan TA, Kane RL, Wilt TJ (2010) Systematic review: effective management strategies for lactose intolerance. Ann Intern Med 152:797–803
Shukla RT (1975) Beta-galactosidase technology: a solution to the lactose problem. CRC Crit Rev Food Technol 5:325–356
Sienkiewicz T, Riedel CL (1990) Whey and whey utilization: possibilities for utilization in agriculture and foodstuffs. Verlag Th., Mann, Gelsenkirchen-Buer
Stein JR (ed) (1973) Handbook of phycological methods: culture methods and growth measurements. Cambridge University Press, Cambridge
Strickland JDH, Parsons TR (1968) A practical handbook of seawater analysis. Fisheries Research Board of Canada, Ottawa, pp 181–184
Tremblay G, Belzile C, Gosselin M, Poulin M, Roy S, Tremblay J (2009) Late summer phytoplankton distribution along a 3500 km transect in Canadian Arctic waters: strong numerical dominance by picoeukaryotes. Aquat Microb Ecol 54.1:55–70
Wynne MJ, Hallan JK (2015) Reinstatement of Tetradesmus GM Smith (Sphaeropleales, Chlorophyta). Feddes Repert 126:83–86
Zarate S, Lopez-Leiva MH (1990) Oligosaccharide formation during enzymatic lactose hydrolysis: a literature review. J Food Prot 53:262–268
Zhu CJ, Lee YK (1997) Determination of biomass dry weight of marine microalgae. J Appl Phycol 9:189–194
Funding
This study received a financial support from NSERC (National Science and Engineering Research Council of Canada), INAF (Institute of Nutrition And functional Foods - Pilot projects program), FRQNT (Fonds de Recherche du Québec sur la Nature et les Technologies), and MAPAQ (Ministère de l’Agriculture, des Pêcheries et de l’Alimentation du Québec - Innov’Action program).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bentahar, J., Doyen, A., Beaulieu, L. et al. Investigation of β-galactosidase production by microalga Tetradesmus obliquus in determined growth conditions. J Appl Phycol 31, 301–308 (2019). https://doi.org/10.1007/s10811-018-1550-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1550-y