Abstract
Purpose
To investigate vascular endothelial growth factor (VEGF) and proliferating cell nuclear antigen (PCNA) immunoreactivities, as well as apoptosis and oxidative stress levels in Streptozotocin (STZ)-induced diabetic rats, and determine how neferine affected these parameters.
Methods
Thirty-five male Sprague Dawley rats were divided into five groups of seven. Fasting blood glucose was measured 72 h after diabetes mellitus (DM) induction in 21 rats using 60 mg/kg STZ dissolved in 0.4 ml (0.1 M) sodium-citrate buffer (pH:4.5), with values > 250 mg/dl considered diabetic. Group 1 received no treatment. Group 3 (healthy rats) received daily intraperitoneal (IP) 4 mg/kg neferine. Following DM induction: Group 2 (sham) received daily IP 0.25 ml/kg 0.9% normal saline; Group 4 received single IP 0.01 mL (2.5 mg/kg) bevacizumab, followed by daily IP 0.25 mL/kg 0.9% normal saline; and Group 5 received daily IP 4 mg/kg neferine. Total antioxidant capacity (TAC) and total oxidative stress (TOS) levels in serum and ocular tissue homogenates were evaluated using ELISA. TUNEL method was used for determining apoptosis and immuno-histochemical staining for PCNA and VEGF immunoreactivities.
Results
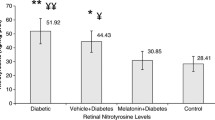
Group 5 had significantly higher TAC and lower TOS in serum and ocular tissue homogenates than Group 4 (p < 0.05). Despite significantly lower VEGF levels and apoptosis (p < 0.05), there was no significant change in PCNA immunoreactivity in Group 5 (p > 0.05).
Conclusions
DM was associated with lower TAC, higher TOS and apoptotic cells, as well as VEGF and PCNA immunoreactivities in the retina. Neferine altered parameters other than PCNA in the opposite direction, demonstrating reductive effects on DM.







Similar content being viewed by others
Data Availability
Data are available from the corresponding author on reasonable request.
References
Whiting DR, Guariguata L, Weil C, Shaw J (2011) IDF diabetes atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract 94:311–321. https://doi.org/10.1016/j.diabres.2011.10.029
Bourne RR, Stevens GA, White RA, Smith JL, Flaxman SR, Price H, Jonas JB, Keeffe J, Leasher J, Naidoo K, Pesudovs K, Resnikoff S, Taylor HR (2013) Vision Loss Expert Group. Causes of vision loss worldwide, 1990–2010: a systematic analysis. Lancet Glob Health 1:e339-349. https://doi.org/10.1016/S2214-109X(13)70113-X
Furukawa H (1965) On the alkaloids of Nelumbo nucifera Gaertn. IX. Alkaloids of loti embryo. (2) Structure of neferine, a new biscoclaurine alkaloid. Yakugaku Zasshi 85:335–338 (Japanese PMID: 14319438)
Chandra S, Rawat DS (2015) Medicinal plants of the family Caryophyllaceae: a review of ethno-medicinal uses and pharmacological properties. Integr Med Res 4:123–131. https://doi.org/10.1016/j.imr.2015.06.004
Kalai Selvi S, Vinoth A, Varadharajan T, Weng CF, Vijaya Padma V (2017) Neferine augments therapeutic efficacy of cisplatin through ROS- mediated non-canonical autophagy in human lung adenocarcinoma (A549 cells). Food Chem Toxicol 103:28–40. https://doi.org/10.1016/j.fct.2017.02.020
Baskaran R, Priya LB, Kalaiselvi P, Poornima P, Huang CY, Padma VV (2017) Neferine from Nelumbo nucifera modulates oxidative stress and cytokines production during hypoxia in human peripheral blood mononuclear cells. Biomed Pharmacother 93:730–736. https://doi.org/10.1016/j.biopha.2017.07.003
Mukherjee PK, Saha K, Giri SN, Pal M, Saha BP (1995) Antifungal screening of Nelumbo nucifera (Nymphaeaceae) rhizome extract. Indian J Microbiol 33:327–330
Sharma BR, Gautam LN, Adhikari D, Karki R (2017) A comprehensive review on chemical profiling of Nelumbo Nucifera: potential for drug development. Phytother Res 31:3–26. https://doi.org/10.1002/ptr.5732
Yu J, Hu WS (1997) Effects of neferine on platelet aggregation in rabbits. Yao Xue Xue Bao 32:1–4 (Chinese PMID: 11243209)
Zhou YJ, Xiang JZ, Yuan H, Liu H, Tang Q, Hao HZ, Yin Z, Wang J, Ming ZY (2013) Neferine exerts its antithrombotic effect by inhibiting platelet aggregation and promoting dissociation of platelet aggregates. Thromb Res 132:202–210. https://doi.org/10.1016/j.thromres.2013.05.018
Zhang X, Liu Z, Xu B, Sun Z, Gong Y, Shao C (2012) Neferine, an alkaloid ingredient in lotus seed embryo, inhibits proliferation of human osteosarcoma cells by promoting p38 MAPK-mediated p21 stabilization. Eur J Pharmacol 677:47–54. https://doi.org/10.1016/j.ejphar.2011.12.035
Kadioglu O, Law BYK, Mok SWF, Xu SW, Efferth T, Wong VKW (2017) Mode of action analyses of Neferine, a Bisbenzylisoquinoline Alkaloid of Lotus (Nelumbo nucifera) against multidrug-resistant tumor cells. Front Pharmacol 8:238. https://doi.org/10.3389/fphar.2017.00238
Zhang Q, Li Y, Miao C, Wang Y, Xu Y, Dong R, Zhang Z, Griffin BB, Yuan C, Yan S, Yang X, Liu Z, Kong B (2018) Anti-angiogenesis effect of Neferine via regulating autophagy and polarization of tumor-associated macrophages in high-grade serous ovarian carcinoma. Cancer Lett 432:144–155. https://doi.org/10.1016/j.canlet.2018.05.049
Bharathi Priya L, Baskaran R, Huang CY, Vijaya Padma V (2018) Neferine modulates IGF-1R/Nrf2 signaling in doxorubicin treated H9c2 cardiomyoblasts. J Cell Biochem 119:1441–1452. https://doi.org/10.1002/jcb.26305
Huang Y, Bai Y, Zhao L, Hu T, Hu B, Wang J, Xiang J (2007) Pharmacokinetics and metabolism of neferine in rats after a single oral administration. Biopharm Drug Dispos 28:361–372. https://doi.org/10.1002/bdd.556
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275 (PMID: 14907713)
Baynes JW, Thorpe SR (1999) Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes 48:1–9. https://doi.org/10.2337/diabetes.48.1.1
Akkuş I, Kalak S, Vural H, Caglayan O, Menekşe E, Can G, Durmuş B (1996) Leukocyte lipid peroxidation, superoxide dismutase, glutathione peroxidase and serum and leukocyte vitamin C levels of patients with type II diabetes mellitus. Clin Chim Acta 244:221–227. https://doi.org/10.1016/0009-8981(96)83566-2
Li C, Miao X, Li F, Wang S, Liu Q, Wang Y, Sun J (2017) Oxidative stress-related mechanisms and Antioxidant therapy in diabetic retinopathy. Oxid Med Cell Longev 2017:9702820. https://doi.org/10.1155/2017/9702820
Liu X, Song X, Lu J, Chen X, Liang E, Liu X, Zhang M, Zhang Y, Du Z, Zhao Y (2016) Neferine inhibits proliferation and collagen synthesis induced by high glucose in cardiac fibroblasts and reduces cardiac fibrosis in diabetic mice. Oncotarget 7:61703–61715. https://doi.org/10.18632/oncotarget.11225
Pan Y, Cai B, Wang K, Wang S, Zhou S, Yu X, Xu B, Chen L (2009) Neferine enhances insulin sensitivity in insulin resistant rats. J Ethnopharmacol 124:98–102. https://doi.org/10.1016/j.jep.2009.04.008
Li G, Xu H, Zhu S, Xu W, Qin S, Liu S, Tu G, Peng H, Qiu S, Yu S, Zhu Q, Fan B, Zheng C, Li G, Liang S (2013) Effects of neferine on CCL5 and CCR5 expression in SCG of type 2 diabetic rats. Brain Res Bull 90:79–87. https://doi.org/10.1016/j.brainresbull.2012.10.002
Beyazyıldız E, Cankaya AB, Ergan E, Anayol MA, Ozdamar Y, Sezer S, Tırhış MH, Yılmazbaş P, Oztürk F (2013) Changes of total antioxidant capacity and total oxidant status of aqueous humor in diabetes patients and correlations with diabetic retinopathy. Int J Ophthalmol 6:531–536. https://doi.org/10.3980/j.issn.2222-3959.2013.04.23
Jung HA, Karki S, Kim JH, Choi JS (2015) BACE1 and cholinesterase inhibitory activities of Nelumbo nucifera embryos. Arch Pharm Res 38:1178–1187. https://doi.org/10.1007/s12272-014-0492-4
Guan G, Han H, Yang Y, Jin Y, Wang X, Liu X (2014) Neferine prevented hyperglycemia-induced endothelial cell apoptosis through suppressing ROS/Akt/NF-κB signal. Endocrine 47:764–771. https://doi.org/10.1007/s12020-014-0186-1
Marthandam Asokan S, Mariappan R, Muthusamy S, Velmurugan BK (2018) Pharmacological benefits of neferine-A comprehensive review. Life Sci 199:60–70. https://doi.org/10.1016/j.lfs.2018.02.032
Pierce EA, Avery RL, Foley ED, Aiello LP, Smith LE (1995) Vascular endothelial growth factor/vascular permeability factor expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci U S A 92:905–909. https://doi.org/10.1073/pnas.92.3.905
Hammes HP, Lin J, Bretzel RG, Brownlee M, Breier G (1998) Upregulation of the vascular endothelial growth factor/vascular endothelial growth factor receptor system in experimental background diabetic retinopathy of the rat. Diabetes 47:401–406. https://doi.org/10.2337/diabetes.47.3.401
Ferrara N, Carver-Moore K, Chen H, Dowd M, Lu L, O’Shea KS, Powell-Braxton L, Hillan KJ, Moore MW (1996) Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380:439–442. https://doi.org/10.1038/380439a0
Mandava N, Blackburn P, Paul DB, Wilson MW, Read SB, Alspaugh E, Tritz R, Barber JR, Robbins JM, Kruse CA (2002) Ribozyme to proliferating cell nuclear antigen to treat proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci 43:3338–3348
Funding
This research was funded by the Firat University Scientific Research Projects Unit under Project Number TF1958.
Author information
Authors and Affiliations
Contributions
The authors all contributed to the study’s conception and design. MC, HHG, and OA prepared the materials, collected data, and analyzed the results. MC wrote the first draft of the manuscript, and all authors provided feedback on previous drafts. The final manuscript was read and approved by all of the authors. MC = Mehmet CEVIK, HHG = Hamidu Hamisi GOBEKA, OA = Orhan AYDEMİR.
Corresponding author
Ethics declarations
Conflict of Interest
The author(s) declare(s) no conflict of interest.
Financial Interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials mentioned in this article.
Ethics Approval
The Animal Research and Ethics Committee at Firat University approved this animal study with the approval number and date 169-18.09.2019
Consent to Participate and Consent for Publication
My colleagues and I conducted the research and co-authored the manuscript. We have all approved the manuscript for submission and publication in your journal, and I am the corresponding author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cevik, M., Gobeka, H.H. & Aydemir, O. Effects of neferine on retinal tissue in experimental diabetic rat model. Int Ophthalmol 43, 249–260 (2023). https://doi.org/10.1007/s10792-022-02424-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-022-02424-0