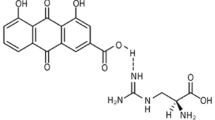
Abstract
Diabetes mellitus has been identified as a major risk factor for cardiovascular diseases. High glucose-induced endothelial dysfunction and apoptosis is an important pathological feature of diabetic vasculopathy. Neferine, an alkaloid ingredient in lotus seed embryo has many biological actions such as anticancer and antioxidant. But little is known about whether Neferine protects endothelial cells against high glucose-induced oxidative stress and apoptosis. The present study was conducted to investigate the preventive effects of Neferine on hyperglycemia-induced injury of human umbilical vein endothelial cells (HUVECs). Our study showed that Neferine pretreatment effectively suppressed high glucose-induced HUVECs apoptosis. Also, Neferine pretreatment inhibited the augment of reactive oxygen species (ROS) in high glucose-treated HUVECs. The changes of SOD and MDA level in high glucose-treated HUVECs were also prevented by Neferine. Further study showed that Neferine did not affect the phosphorylation of JNK and p38 in high glucose-treated HUVECs. Interestingly, Neferine markedly inhibited high glucose-induced activation of PI3K/Akt pathway in HUVECs. High glucose-induced activation of NF-κB signal was also obviously suppressed by Neferine pretreatment. Collectively, we found that Neferine inhibited high glucose-induced endothelial apoptosis via blocking ROS/Akt/NF-κB pathway, which provides the evidence for using Neferine to treat diabetic vasculopathy.






Similar content being viewed by others
References
A. Towfigh, M. Romanova, J.E. Weinreb, B. Munjas, M.J. Suttorp, A. Zhou, P.G. Shekelle, Self-monitoring of blood glucose levels in patients with type 2 diabetes mellitus not taking insulin: a meta-analysis. Am. J. Manag. Care 14, 468–475 (2008)
S. Del Prato, A.H. Barnett, H. Huisman, D. Neubacher, H.J. Woerle, K.A. Dugi, Effect of linagliptin monotherapy on glycaemic control and markers of beta-cell function in patients with inadequately controlled type 2 diabetes: a randomized controlled trial. Diabetes Obes. Metab. 13, 258–267 (2011)
S.B. Williams, A.B. Goldfine, F.K. Timimi, H.H. Ting, M.A. Roddy, D.C. Simonson, M.A. Creager, Acute hyperglycemia attenuates endothelium-dependent vasodilation in humans in vivo. Circulation 97, 1695–1701 (1998)
D.D. Belke, E.A. Swanson, W.H. Dillmann, Decreased sarcoplasmic reticulum activity and contractility in diabetic db/db mouse heart. Diabetes 53, 3201–3208 (2004)
F.M. Ho, W.W. Lin, B.C. Chen, C.M. Chao, C.R. Yang, L.Y. Lin, C.C. Lai, S.H. Liu, C.S. Liau, High glucose-induced apoptosis in human vascular endothelial cells is mediated through NF-kappaB and c-Jun NH2-terminal kinase pathway and prevented by PI3K/Akt/eNOS pathway. Cell. Signal. 18, 391–399 (2006)
M.L. Sheu, F.M. Ho, R.S. Yang, K.F. Chao, W.W. Lin, S.Y. Lin-Shiau, S.H. Liu, High glucose induces human endothelial cell apoptosis through a phosphoinositide 3-kinase-regulated cyclooxygenase-2 pathway. Arterioscler. Thromb. Vasc. Biol. 25, 539–545 (2005)
D. Detaille, B. Guigas, C. Chauvin, C. Batandier, E. Fontaine, N. Wiernsperger, X. Leverve, Metformin prevents high-glucose-induced endothelial cell death through a mitochondrial permeability transition-dependent process. Diabetes 54, 2179–2187 (2005)
X. Zhang, Z. Liu, B. Xu, Z. Sun, Y. Gong, C. Shao, Neferine, an alkaloid ingredient in lotus seed embryo, inhibits proliferation of human osteosarcoma cells by promoting p38 MAPK-mediated p21 stabilization. Eur. J. Pharmacol. 677, 47–54 (2012)
P. Poornima, R.S. Quency, V.V. Padma, Neferine induces reactive oxygen species mediated intrinsic pathway of apoptosis in HepG2 cells. Food Chem. 136, 659–667 (2013)
J.G. Cao, X.Q. Tang, S.H. Shi, Multidrug resistance reversal in human gastric carcinoma cells by neferine. World J. Gastroenterol. 10, 3062–3064 (2004)
Y. Pan, B. Cai, K. Wang, S. Wang, S. Zhou, X. Yu, B. Xu, L. Chen, Neferine enhances insulin sensitivity in insulin resistant rats. J. Ethnopharmacol. 124, 98–102 (2009)
L. Zhao, X. Wang, Q. Chang, J. Xu, Y. Huang, Q. Guo, S. Zhang, W. Wang, X. Chen, J. Wang, Neferine, a bisbenzylisoquinline alkaloid attenuates bleomycin-induced pulmonary fibrosis. Eur. J. Pharmacol. 627, 304–312 (2010)
J. Yu, W.S. Hu, Effects of neferine on platelet aggregation in rabbits. Yao Xue Xue Bao 32(1), 1–4 (1997)
C.Y. Wang, D.C. Guttridge, M.W. Mayo, A.S. Baldwin Jr, NF-kappaB induces expression of the Bcl-2 homologue A1/Bfl-1 to preferentially suppress chemotherapy-induced apoptosis. Mol. Cell. Biol. 19, 5923–5929 (1999)
K. Yamamoto, R. Morishita, S. Hayashi, H. Matsushita, H. Nakagami, A. Moriguchi, K. Matsumoto, T. Nakamura, Y. Kaneda, T. Ogihara, Contribution of Bcl-2, but not Bcl-xL and Bax, to antiapoptotic actions of hepatocyte growth factor in hypoxia-conditioned human endothelial cells. Hypertension 37, 1341–1348 (2001)
G. Lalitha, P. Poornima, A. Archanah, V.V. Padma, Protective effect of neferine against isoproterenol-induced cardiac toxicity. Cardiovasc. Toxicol. 13, 168–179 (2013)
T. Yu, S.S. Sheu, J.L. Robotham, Y. Yoon, Mitochondrial fission mediates high glucose-induced cell death through elevated production of reactive oxygen species. Cardiovasc. Res. 79, 341–351 (2008)
Y.J. Choi, H.S. Lim, J.S. Choi, S.Y. Shin, J.Y. Bae, S.W. Kang, I.J. Kang, Y.H. Kang, Blockade of chronic high glucose-induced endothelial apoptosis by Sasa borealis bamboo extract. Exp. Biol. Med. (Maywood) 233, 580–591 (2008)
P. Poornima, C.F. Weng, V.V. Padma, Neferine from Nelumbo nucifera induces autophagy through the inhibition of PI3K/Akt/mTOR pathway and ROS hyper generation in A549 cells. Food Chem. 141, 3598–3605 (2013)
Acknowledgments
This work was supported by Science and Technology Fund of Heilongjiang Education Bureau.
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guan, G., Han, H., Yang, Y. et al. Neferine prevented hyperglycemia-induced endothelial cell apoptosis through suppressing ROS/Akt/NF-κB signal. Endocrine 47, 764–771 (2014). https://doi.org/10.1007/s12020-014-0186-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-014-0186-1