Abstract
Purpose
To compare the efficacy of transscleral cyclophotocoagulation with MicroPulse® laser (Iridex, Silicon Valley, California, USA) with 3000 mW to Ahmed valve implantation in eyes with advanced stage of primary open-angle glaucoma.
Methods
In a prospective observational clinical study, 30 patients (30 eyes) with advanced open-angle glaucoma were randomized for either micropulse transscleral cyclophotocoagulation with 3000 mW or Ahmed valve implantation. Fifteen eyes were treated with transscleral cyclophotocoagulation with MicroPulse® laser with 3000 mW (group A) and 15 eyes with Ahmed valve implantation (group B). As inclusion criteria are included the diagnosis of advanced primary open-angle glaucoma, an intraocular pressure above 21 mmHg, cup-to-disk ratio 0.9–1.0, failure to meet the target IOP with either maximal tolerated local medical therapy (2–4 antiglaucoma agents) or systemic therapy (acetazolamide). The follow-up time of the study was 12 months. An absolute success was defined the achievement of IOP between 6 and 15 mmHg and at least 30% reduction of the IOP from baseline under reduced or the same number of antiglaucoma agents after the surgical procedure without following glaucoma surgeries and as qualified success the achievement of IOP between 6 and 18 mmHg and at least 20% reduction of the IOP from baseline regardless of the number of postoperative antiglaucoma agents. The efficacy was evaluated by estimating the absolute success rate and the qualified success rate using the Kaplan–Meier survival analysis.
Results
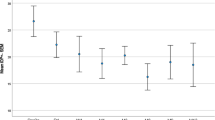
A reduction of the intraocular pressure > 30% was achieved in 33.3% of group A and in 73.3% of group B during the follow-up period of 12 months. A statistically significant decrease in the number of eye drops was observed in both groups (p < 0.01). 53.3% of the eyes of group A underwent additional anti-glaucoma procedures to achieve target intraocular pressure. The number of the local medications that were administered 12 months after the ocular surgery was 2 (± 1.3) in group A and 0.57 (± 0.9) in group B, compared to 3.2 (± 0.78) in group A and 3.33 (± 0.7) in group B administered prior to the surgery (p: 0.016). 8 eyes (53.33%) in group A were referred for additional treatment due to an acute postoperative rise in IOP (5 eyes: mTS-CPC, 1 eye: canaloplasty, 2 eyes: AGV implantation). No further anti-glaucoma procedures were necessary in group B.
Conclusion
The Ahmed valve implantation achieves a more efficient decrease of the intraocular pressure as well as of the number of antiglaucoma agents than the transscleral cyclophotocoagulation with MicroPulse® diode laser 3000 mW. Additionally, the Ahmed valve implantation showed better results in terms of absolute and qualified success rates in the treatment of advanced primary open-angle glaucoma.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article
Abbreviations
- AC:
-
Anterior chamber
- AGV:
-
Ahmed valve implantation
- BCVA:
-
Best corrected visual acuity
- GAT:
-
Goldmann applanation tonometry
- IOP:
-
Intraocular pressure
- MIGS:
-
Micro-invasive glaucoma surgery
- mTS-CPC:
-
MicroPulse® transscleral cyclophotocoagulation
- PEX:
-
Pseudoexfoliation
- POAG:
-
Primary open-angle glaucoma
References
Nassiri N, Kamali G, Rahnavardi M, Mohammadi B, Nassiri S, Rahmani L, Nassiri N (2010) Ahmed glaucoma valve and single-plate Molteno implants in treatment of refractory glaucoma: a comparative study. Am J Ophthalmol 149:893–902
Hu WD, Moster MR, Zheng CX, Sabherwal N, Pequignot E, Cvintal V, Ekici F, Waisbourd M (2016) Outcomes of sequential glaucoma drainage implants in refractory glaucoma. J Glaucoma 25:e340-345
Chen MF, Kim CH, Coleman AL (2019) Cyclodestructive procedures for refractory glaucoma. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD012223.pub2
Williams AL, Moster MR, Rahmatnejad K, Resende AF, Horan T, Reynolds M, Yung E, Abramowitz B, Kuchar S, Waisbourd M (2018) Clinical efficacy and safety profile of micropulse® transscleral cyclophotocoagulation in refractory glaucoma. J Glaucoma 27:445–449
Aquino MC, Barton K, Tan AM, Sng C, Li X, Loon SC, Chew PT (2015) MicroPulse® versus continuous wave transscleral diode cyclophotocoagulation in refractory glaucoma: a randomized exploratory study. Clin Exp Ophthalmol 43:40–46
Sanchez FG, Peirano-Bonomi JC, Grippo TM (2018) MicroPulse® transscleral cyclophotocoagulation: a hypothesis for the ideal parameters. Med Hypothesis Discov Innov Ophthalmol 7:94–100
Sarrafpour S, Saleh D, Ayoub S, Radcliffe NM (2019) MicroPulse® transscleral cyclophotocoagulation: a look at long-term effectiveness and outcomes. Ophthalmol Glaucoma 2:167–171
Yelenskiy A, Gillette TB, Arosemena A, Stern AG, Garris WJ, Young CT, Hoyt M, Worley N, Zurakowski D, Ayyala RS (2018) Patient outcomes following micropulse® transscleral cyclophotocoagulation: intermediate-term results. J Glaucoma 27:920–925
Varikuti VNV, Shah P, Rai O, Chaves AC, Miranda A, Lim BA, Dorairaj SK, Sieminski SF (2019) Outcomes of micropulse® transscleral cyclophotocoagulation in eyes with good central vision. J Glaucoma 28:901–905
Damji KF, Behki R, Wang L (2003) Target IOP Workshop participants. Canadian perspectives in glaucoma management: setting target intraocular pressure range. Can J Ophthalmol 38(3):189–97
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Eid TE, Katz LJ, Spaeth GL, Augsburger JJ (1997) Tube-shunt surgery versus neodymium:YAG cyclophotocoagulation in the management of neovascular glaucoma. Ophthalmology 104:1692–1700
Yildirim N, Yalvac IS, Sahin A, Ozer A, Bozca T (2009) A comparative study between diode laser cyclophotocoagulation and the Ahmed glaucoma valve implant in neovascular glaucoma: a long-term follow-up. J Glaucoma 18:192–196
Lima FE, Magacho L, Carvalho DM, Susanna R Jr, Avila MP (2004) A prospective, comparative study between endoscopic cyclophotocoagulation and the Ahmed drainage implant in refractory glaucoma. J Glaucoma 13:233–237
Choy BNK, Lai JSM, Yeung JCC, Chan JCH (2018) Randomized comparative trial of diode laser transscleral cyclophotocoagulation versus Ahmed glaucoma valve for neovascular glaucoma in Chinese - a pilot study. Clin Ophthalmol 12:2545–2552
Dubey S, Sharma V, Agrawal A, Chauhan L, Douglas G (2015) Safety and efficacy of Ahmed glaucoma valve implantation in refractory glaucomas in Northern Indian eyes. Saudi J Ophthalmol 29:103–8
Netland PA (2009) The Ahmed glaucoma valve in neovascular glaucoma (An AOS Thesis). Trans Am Ophthalmol Soc 107:325–342
Min SH, Chung YS, Kim HK, Sohn YH (2005) neovascular glaucoma: diode laser trans-scleral cyclophotocoaculation versus ahmed glaucoma valve implant. J Korean Ophthalmol Soc 46:1151–1157
Noureddin BN, Wilson-Holt N, Lavin M (1992) Advanced uncontrolled glaucoma, Nd: YAG cyclophotocoagulation or tube surgery. Ophthalmology 99:430–437
Christakis PG, Kalenak JW, Tsai JC, Zurakowski D, Kammer JA, Harasymowycz PJ, Mura JJ, Cantor LB, Ahmed IIK (2016) the ahmed versus baerveldt study: five-year treatment outcomes. Ophthalmology 123:2093–102
Barton K, Feuer WJ, Budenz DL, Schiffman J, Costa VP, Godfrey DG, Buys YM (2014) Three-year treatment outcomes in the Ahmed Baerveldt comparison study. Ophthalmology 121:1547–1557
Wang YW, Wang PB, Zeng C, Xia XB (2015) Comparison of the Ahmed glaucoma valve with the Baerveldt glaucoma implant: a meta-analysis. BMC Ophthalmol 15:132
Dietlein TS, Neugebauer A, Fricke J, Lappas A, Rosentreter A (2016) Postoperative follow-up of glaucoma drainage devices. Klin Monast Augenheilkd 233:606–612
Acknowledgements
The authors received no outside financial support for this study, including pharmacological or industry support.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SF, KK, IV, GP, SAP, EZA and MK. The first draft of the manuscript was written by SF and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in this observatory study involving human participants were in accordance with the ethical standards of the institutional research committee of St-Johannes-Hospital in Dortmund, Germany and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent for participation was obtained from all individual participants included in the study.
Consent to publish
Informed consent for publication was obtained from all individual participants included in the study.
Clinical trial registration
This is an observatory study and no clinical trial registration is necessary.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fili, S., Kontopoulou, K., Vastardis, I. et al. Transscleral cyclophotocoagulation with MicroPulse® laser versus Ahmed valve implantation in patients with advanced primary open-angle glaucoma. Int Ophthalmol 41, 1271–1282 (2021). https://doi.org/10.1007/s10792-020-01682-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-020-01682-0