Abstract
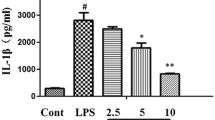
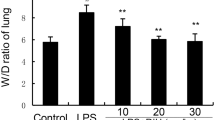
Acute lung injury and acute respiratory distress syndrome (ALI/ARDS) have high mortality rates. Though corticosteroids are commonly used for the treatment of these conditions, their efficacy has not been conclusively demonstrated and their use can induce various adverse reactions. Hence, the application of corticosteroids as therapeutic modalities for ALI/ARDS is limited. Meanwhile, the aporphine alkaloid oxocrebanine isolated from Stephania pierrei tubers has demonstrated anti-inflammatory efficacy in murine/human macrophage cell lines stimulated by lipopolysaccharide (LPS). Accordingly, the primary objectives of the present study are to investigate the anti-inflammatory effects of oxocrebanine on LPS-induced murine alveolar epithelial (MLE-12) cells and its efficacy against LPS-induced murine ALI. Results show that oxocrebanine downregulates the abundance of interleukin (IL)-1beta, IL-6, and inducible nitric oxide synthase, as well as the phosphorylation of nuclear factor-kappaB (NF-κB), stress-activated protein kinase (SAPK)/c-Jun N-terminal kinase (JNK), p38, protein kinase B (Akt), and glycogen synthase kinase-3beta signalling proteins in LPS-induced MLE-12 cells. Moreover, in a murine ALI model, oxocrebanine lowers lung injury scores and lung wet/dry weight ratios while reducing inflammatory cell infiltration. It also suppresses LPS-induced tumour necrosis factor-alpha and IL-6 in the bronchoalveolar lavage fluid and plasma. Moreover, oxocrebanine downregulates NF-κB, SAPK/JNK, p38, and Akt phosphorylation in the lung tissues of LPS-treated mice. Taken together, the foregoing results show that oxocrebanine provides significant protection against LPS-induced ALI in mice primarily by suppressing various inflammatory signalling pathways in alveolar epithelial cells and lung tissues. Hence, oxocrebanine might prove effective as an anti-inflammatory agent for the treatment of lung inflammation.






Similar content being viewed by others
Data availability
All the data presented in this study are included in the article.
References
Ahmadian E, Hosseiniyan Khatibi SM, Razi Soofiyani S et al (2021) Covid-19 and kidney injury: pathophysiology and molecular mechanisms. Rev Med Virol 31:e2176. https://doi.org/10.1002/rmv.2176
Backhouse N, Delporte C, Givernau M, Cassels BK, Valenzuela A, Speisky H (1994) Anti-inflammatory and antipyretic effects of boldine. Agents Actions 42:114–117. https://doi.org/10.1007/BF01983475
Bailey SA, Zidell RH, Perry RW (2004) Relationships between organ weight and body/brain weight in the rat: What is the best analytical endpoint? Toxicol Pathol 32:448–466. https://doi.org/10.1080/01926230490465874
Chaichompoo W, Rojsitthisak P, Pabuprapap W et al (2021) Stephapierrines A-H, new tetrahydroprotoberberine and aporphine alkaloids from the tubers of Stephania pierrei Diels and their anti-cholinesterase activities. RSC Adv 11:21153–21169. https://doi.org/10.1039/d1ra03276c
Chen GS, Huang KF, Huang CC, Wang JY (2015) Thaliporphine derivative improves acute lung injury after traumatic brain injury. BioMed Res Int 2015:729831. https://doi.org/10.1155/2015/729831
Chulrik W, Jansakun C, Chaichompoo W et al (2022) Oxocrebanine from Stephania pierrei exerts macrophage anti-inflammatory effects by downregulating the NF-κB, MAPK, and PI3K/Akt signalling pathways. Inflammopharmacology 30:1369–1382. https://doi.org/10.1007/s10787-022-01021-y
Dawood DRM, Salum GM, El-Meguid MA (2022) The impact of COVID-19 on liver injury. Am J Med Sci 363:94–103. https://doi.org/10.1016/j.amjms.2021.11.001
Ding Q, Liu G, Zeng Y et al (2017) Glycogen synthase kinase-3β inhibitor reduces LPS-induced acute lung injury in mice. Mol Med Rep 16:6715–6721. https://doi.org/10.3892/mmr.2017.7469
Feng J, Shi J, Li S, Jie Z (2016) p38MAPK inhibitor ameliorates lipopolysaccharide-induced mild acute respiratory distress syndrome through regulating the balance of Treg cells and Th17 cells. Chest 149:A158. https://doi.org/10.1016/j.chest.2016.02.164
Feng T, Zhou L, Gai S et al (2019) Acacia catechu (Lf) Willd and Scutellaria baicalensis Georgi extracts suppress LPS-induced pro-inflammatory responses through NF-кB, MAPK, and PI3K-Akt signaling pathways in alveolar epithelial type II cells. Phytother Res 33:3251–3260. https://doi.org/10.1002/ptr.6499
Freundt K, Herzmann C, Biedziak D, Scheffzük C, Gaede KI, Stamme C (2022) Surfactant protein a enhances the degradation of LPS-Induced TLR4 in primary alveolar macrophages involving Rab7, β-Arrestin2, and Mtorc1. Infect Immun 90:e0025021. https://doi.org/10.1128/IAI.00250-21
Grahnemo L, Jochems C, Andersson A et al (2015) Possible role of lymphocytes in glucocorticoid-induced increase in trabecular bone mineral density. J Endocrinol 224:97–108. https://doi.org/10.1530/JOE-14-0508
He YQ, Zhou CC, Yu LY et al (2021) Natural product derived phytochemicals in managing acute lung injury by multiple mechanisms. Pharmacol Res 163:105224. https://doi.org/10.1016/j.phrs.2020.105224
Huang C, Wang Y, Li X et al (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395:497–506. https://doi.org/10.1016/S0140-6736(20)30183-5
Intayoung P, Limtrakul P, Yodkeeree S (2016) Antiinflammatory activities of crebanine by inhibition of NF-κB and AP-1 activation through suppressing MAPKs and Akt signaling in LPS-induced RAW 264.7 macrophages. Biol Pharm Bull 39:54–61. https://doi.org/10.1248/bpb.b15-00479
Intusaitrakul C (2010) Trakul Wan Thai. In: Intusaitrakul C (ed) Stephania pierrei Diels, vol 2. Duangkamol Publishing, Bangkok, pp 272–274
Kornecki A, Singh RN (2019) Acute respiratory distress syndrome. In: Wilmott RW, Bush A, Deterding RR, Ratjen F, Sly P, Zar H, Li A (eds) Kendig’s disorders of the respiratory tract in children, 9th edn. Elsevier, Amsterdam, pp 606–614
Kumar V (2020) Pulmonary innate immune response determines the outcome of inflammation during pneumonia and sepsis-associated acute lung injury. Front Immunol 11:1722. https://doi.org/10.3389/fimmu.2020.01722
Landolf KM, Lemieux SM, Rose C et al (2022) Corticosteroid use in ARDS and its application to evolving therapeutics for coronavirus disease 2019 (COVID-19): a systematic review. Pharmacotherapy 42:71–90. https://doi.org/10.1002/phar.2637
Li Y, Huang J, Foley N et al (2016) B7H3 ameliorates LPS-induced acute lung injury via attenuation of neutrophil migration and infiltration. Sci Rep 6:31284. https://doi.org/10.1038/srep31284
Li D, Ren W, Jiang Z, Zhu L (2018) Regulation of the NLRP3 inflammasome and macrophage pyroptosis by the p38 MAPK signaling pathway in a mouse model of acute lung injury. Mol Med Rep 18:4399–4409. https://doi.org/10.3892/mmr.2018.9427
Li W, Veeraraghavan VP, Ma W (2020) Effects of boldine on antioxidants and allied inflammatory markers in mouse models of asthma. J Environ Pathol Tox 39:225–234. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2020034039
Liu M, Chen Y, Wang S et al (2020) α-Ketoglutarate modulates macrophage polarization through regulation of PPARγ transcription and mTORC1/p70S6K pathway to ameliorate ALI/ARDS. Shock 53:103–113. https://doi.org/10.1097/SHK.0000000000001333
Matthay MA, Zemans RL, Zimmerman GA et al (2019) Acute respiratory distress syndrome. Nat Rev Dis Primers 5:18. https://doi.org/10.1038/s41572-019-0069-0
Matute-Bello G, Frevert CW, Martin TR (2008) Animal models of acute lung injury. Am J Physiol-Lung C 295:L379–L399. https://doi.org/10.1152/ajplung.00010.2008
Park KH, Chung EY, Choi YN, Jang HY, Kim JS, Kim GB (2020) Oral administration of Ulmus davidiana extract suppresses interleukin-1β expression in LPS-induced immune responses and lung injury. Genes Genom 42:87–95. https://doi.org/10.1007/s13258-019-00883-x
Pijl H, Meinders AE (1996) Bodyweight change as an adverse effect of drug treatment: mechanisms and management. Drug Saf 14:329–342. https://doi.org/10.2165/00002018-199614050-00005
Rashid M, Khan S, Datta D et al (2021) Efficacy and safety of corticosteroids in acute respiratory distress syndrome: an overview of meta-analyses. Int J Clin Pract 75:e14645. https://doi.org/10.1111/ijcp.14645
Riviello ED, Kiviri W, Twagirumugabe T et al (2016) Hospital incidence and outcomes of the acute respiratory distress syndrome using the Kigali modification of the Berlin definition. Am J Resp Crit Care 193:52–59. https://doi.org/10.1164/rccm.201503-0584OC
Shi X, An X, Yang L et al (2021) Reticulocalbin 3 deficiency in alveolar epithelium attenuated LPS-induced ALI via NF-κB signaling. Am J Physiol-Lung C 320:L627–L639. https://doi.org/10.1152/ajplung.00526.2020
Singh AK, Majumdar S, Singh R, Misra A (2020) Role of corticosteroid in the management of COVID-19: a systemic review and a Clinician’s perspective. Diabetes Metab Syndr 14:971–978. https://doi.org/10.1016/j.dsx.2020.06.054
Tao H, Xu Y, Zhang S (2023) The role of macrophages and alveolar epithelial cells in the development of ARDS. Inflammation 46:47–55. https://doi.org/10.1007/s10753-022-01726-w
Wang J, Yang X, Li Y, Huang JA, Jiang J, Su N (2021) Specific cytokines in the inflammatory cytokine storm of patients with COVID-19-associated acute respiratory distress syndrome and extrapulmonary multiple-organ dysfunction. Virol J 18:117. https://doi.org/10.1186/s12985-021-01588-y
Wijeratne EMK, Hatanaka Y, Kikuchi T, Tezuka Y, Gunatilaka AAL (1996) A dioxoaporphine and alkaloids of two annonaceous plants of Sri lanka. Phytochemistry 42:1703–1706. https://doi.org/10.1016/0031-9422(96)00181-1
Wu H, Yang Y, Guo S et al (2017) Nuciferine ameliorates inflammatory responses by inhibiting the TLR4-mediated pathway in lipopolysaccharide-induced acute lung injury. Front Pharmacol 8:939. https://doi.org/10.3389/fphar.2017.00939
Yasir M, Goyal A, Sonthalia S (2018) Corticosteroid adverse effects. StatPearls Publishing, FL
Yodkeeree S, Pompimon W, Limtrakul Dejkriengkraikul P (2014) Crebanine, an aporphine alkaloid, sensitizes TNF-α-induced apoptosis and suppressed invasion of human lung adenocarcinoma cells A549 by blocking NF-κB-regulated gene products. Tumor Biol 35:8615–8624. https://doi.org/10.1007/s13277-014-1998-6
Zhou X, Gao XP, Fan J et al (2005) LPS activation of Toll-like receptor 4 signals CD11b/CD18 expression in neutrophils. Am J PhysioL-Lung C 288:L655–L662. https://doi.org/10.1152/ajplung.00327.2004
Zhu Z, Sun G (2018) Silymarin mitigates lung impairments in a rat model of acute respiratory distress syndrome. Inflammopharmacology 26:747–754. https://doi.org/10.1007/s10787-017-0407-3
Acknowledgements
This research was supported by the Royal Patronage of Her Royal Highness Princess Maha Chakri Sirindhorn (grant no. WUBG-018/2565). W. Chulrik acknowledges the support of the Walailak University Graduate Research Fund (grant no. CGS-RF-2021/05) and the Walailak University Ph.D. Excellence Scholarship (grant no. 04/2020). The Center of Excellence for Innovation in Chemistry, Ministry of Higher Education, Science, Research, and Innovation provided partial funding for this study to A. Suksamrarn and W. Chaichompoo. We would like to thank Editage (www.editage.com) for English language editing.
Funding
This research was financed by the Royal Patronage of Her Royal Highness Princess Maha Chakri Sirindhorn (grant no. WUBG-018/2565). W. Chulrik thanks the Walailak University Graduate Research Fund (grant no. CGS-RF-2021/05) and a Walailak University Ph.D. Excellence Scholarship (grant no. 04/2020). The Center of Excellence for Innovation in Chemistry, Ministry of Higher Education, Science, Research, and Innovation provided partial funding for this study to A. Suksamrarn and W. Chaichompoo.
Author information
Authors and Affiliations
Contributions
Conceptualisation: WC and WC; methodology: WC and WC; validation: AT and CP; formal analysis: WC and CJ; investigation: WC, CJ, WC, NS, and RK; resources: KR and AS; writing—original draft preparation; WC and WC; writing—review and editing: WC, KR, AS, and WC; supervision and funding acquisition: WC and AS. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The experimental protocol was approved by the Animal Ethics Committee of the Laboratory Animal Unit Research Institute for Health Science of Walailak University (Nakhon Si Thammarat, Thailand) under approval no. WU-AICUC-64015.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chulrik, W., Jansakun, C., Chaichompoo, W. et al. Protective effects of Stephania pierrei tuber-derived oxocrebanine against LPS-induced acute lung injury in mice. Inflammopharmacol 31, 2023–2035 (2023). https://doi.org/10.1007/s10787-023-01231-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01231-y