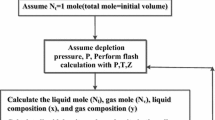
Abstract
This work demonstrates an effective procedure to correlate and predict viscosity of petroleum fluids using friction theory (FT) viscosity model coupled with perturbed-chain statistical associating fluid theory (PC-SAFT). The FT is used for viscosity prediction in reservoir fluids. The PC-SAFT is applied for calculation of equilibrium composition and density of vapor and liquid phases. The FT has a few characteristic parameters for each component for viscosity prediction. These parameters are not available for petroleum fractions. In this study, such a problem is addressed by finding a model to predict FT characteristic parameters for different petroleum fractions as a function of molecular weight and critical pressure. 20 real reservoir fluid samples are used to develop the model. Afterward, for 5 real reservoir oil samples in the evaluation step, the viscosity modeling results are compared against experimental data, and the methods of Tan et. al., Lohrenz et al., and Pedersen et al. for showing the accuracy of proposed model. It is concluded that with suitable characteristic parameters for the FT viscosity model and PC-SAFT, improvement in liquid viscosity estimation can be achieved. The average absolute deviation percent (AAD%) is 10.22% for FT + PC-SAFT (this work), 13.71% for Lohrenz et al. and 23.48 for Pedersen et al. In addition, since the free-volume (FV) theory like FT belongs to semi-empirical viscosity models, a comparison with the FV model (published in the work of Khoshnamvand and Assareh in In J Thermophys 39:1, 2018) is performed. The results demonstrate that the FT viscosity model with presented characteristic parameters in this study gives a comparable accuracy in viscosity prediction for the studied real reservoir fluids. Compared to FV, the FT is less dependent on the EOS calculation which is an advantage of FT.







Similar content being viewed by others
Abbreviations
- A res :
-
Residual Helmholtz free energy
- \({\tilde{a }}^{res}\) :
-
Reduced Residual Helmholtz free energy
- \({\tilde{a }}^{hc}\) :
-
Hard chain contribution to reduced Helmholtz free energy
- \({\tilde{a }}^{disp}\) :
-
Dispersion contribution to reduced Helmholtz free energy
- \({AD\%}_{FT}= 100.\left|\frac{{\upmu }^{exp}-{\upmu }^{FT}}{{\upmu }^{exp}}\right|\) :
-
Absolute deviation percent of calculated viscosity by FT model
- \({AD\%}_{LBC}= 100.\left|\frac{{\upmu }^{exp}-{\upmu }^{LBC}}{{\upmu }^{exp}}\right|\) :
-
Absolute deviation percent of calculated viscosity by LBC method
- a 1 :
-
Characteristic parameter of FT viscosity model
- a 2 :
-
Characteristic parameter of FT viscosity model
- b 1 :
-
Characteristic parameter of FT viscosity model
- b 2 :
-
Characteristic parameter of FT viscosity model
- c 2 :
-
Characteristic parameter of FT viscosity model
- a 10 :
-
Constant of FT viscosity model
- a 11 :
-
Constant of FT viscosity model
- a 20 :
-
Constant of FT viscosity model
- a 21 :
-
Constant of FT viscosity model
- b 10 :
-
Constant of FT viscosity model
- b 11 :
-
Constant of FT viscosity model
- b 20 :
-
Constant of FT viscosity model
- b 21 :
-
Constant of FT viscosity model
- c 20 :
-
Constant of FT viscosity model
- c 21 :
-
Constant of FT viscosity model
- P r :
-
Repulsive Pressure
- P a :
-
Attractive Pressure
- \({K}_{r}\) :
-
Temperature-dependent parameter of FT viscosity model
- \({K}_{a}\) :
-
Temperature-dependent parameter of FT viscosity model
- \({K}_{rr}\) :
-
Temperature-dependent parameter of FT viscosity model
- \({K}_{r,mx}\) :
-
Mixture temperature-dependent parameter of FT viscosity model
- \({K}_{a,mx}\) :
-
Mixture temperature-dependent parameter of FT viscosity model
- \({K}_{rr,mx}\) :
-
Mixture temperature-dependent parameter of FT viscosity model
- \({K}_{mx}\) :
-
Mixture temperature-dependent parameter of FT viscosity model
- EOS :
-
Equation of state
- \(OF\left(a\right)= \frac{1}{n}.\sum \left|\frac{{\upmu }^{exp}-{\upmu }^{calc}}{{\upmu }^{exp}}\right|\) :
-
Objective function for viscosity
- VS :
-
Volume shift
- k :
-
Boltzmann constant
- MW :
-
Molecular weight
- \(M\) :
-
Molecular weight
- MW_plus fraction:
-
Molecular weight of plus fraction
- m :
-
Segment length
- N :
-
Avogadro number
- N t :
-
Total mole in the cell
- N L :
-
Liquid mole number in the cell
- N G :
-
Gas mole number in the cell
- Pc :
-
Critical pressure
- Pbubble :
-
Bubble point pressure
- R:
-
Universal gas constant
- SP.GR:
-
Specific gravity
- S_plus fraction:
-
Specific gravity of plus fraction
- T:
-
Temperature [kelvin]
- \({T}_{r}\) :
-
Reduced temperature
- Tc :
-
Critical temperature
- Vc :
-
Critical volume
- V l :
-
Liquid volume
- V g :
-
Gas volume
- x:
-
Liquid composition
- xi :
-
Mole fraction of species I
- y :
-
Gas composition
- Zhc :
-
Hard chain contribution of Compressibility factor
- Zdisp :
-
Dispersion contribution of compressibility factor
- Z g :
-
Gas compressibility factor
- Z l :
-
Liquid compressibility factor
- η:
-
Viscosity
- \({\upeta }_{mx}\) :
-
Viscosity of mixture
- \({\upeta }_{0}\) :
-
Dilute gas viscosity
- \({\upeta }_{0,mx}\) :
-
Dilute gas viscosity of mixture
- \(\Delta\upeta\) :
-
Dense state viscosity
- \({\Delta\upeta }_{f,mx}\) :
-
Dense state viscosity of mixture
- η:
-
Packing fraction density
References
Y. Khoshnamvand, M. Assareh, Int. J. Thermophys 39, 1 (2018)
J.H. Dymond, M.A. Awan, Int. J. Thermophys 10, 941–951 (1989)
M.J. Assael, J.H. Dymond, M. Papadaki, P.M. Patterson, Fluid Ph. Equilibria 75, 245–255 (1992)
A. Allal, M. Moha-Ouchane, C. Boned, Phys. Chem. Liq. 39, 1–30 (2001)
S.E. Quiñones-Cisneros, C.K. Zéberg-Mikkelsen, E.H. Stenby, Fluid Ph. Equilibria 169, 249–276 (2000)
S.E. Quiñones-Cisneros, C.K. Zéberg-Mikkelsen, E.H. Stenby, Fluid Ph. Equilibria 178, 1–16 (2001)
S.E. Quiñones-Cisneros, U.K. Deiters, J. Phys. Chem 110, 12820–12834 (2006)
A.S. de Wijn, V. Vesovic, G. Jackson, J.M. Trusler, J. Chem. Phys. 128, 204–214 (2008)
R. Srivastava, D.K. Dwivedee, K.N. Khanna, J. Mol. Liq. 139, 29–34 (2008)
T. Goel, C.N. Patra, T. Mukherjee, C. Chakravarty, J. Chem. Phys. 129, 164–174 (2008)
G. Galliero, C. Boned, J. Fernández, J. Chem. Phys. 134, 64–74 (2011)
L.T. Novak, Int. J. Chem. React 9, 1–20 (2011)
G. Galliéro, C. Boned, A. Baylaucq, Ind. Eng. Chem. Res. 44, 6963 (2005)
G. Galliero, C. Boned, Phys. Rev 79, 102–120 (2009)
M.S. Zabaloy, J.M. Machado, E.A. Macedo, Int. J. Thermophys 22, 829–858 (2001)
M.S. Zabaloy, V.R. Vasquez, E.A. Macedo, J Supercrit 36, 106–117 (2005)
G. Soave, Chem. Eng. Sci. 27, 1197–1203 (1972)
D.-Y. Peng, D.B. Robinson, Ind. Eng. Chem. Fundam 15, 59–64 (1976)
W.G. Chapman, K.E. Gubbins, G. Jackson, M. Radosz, Ind. Eng. Chem. Res. 29, 1709 (1990)
W.G. Chapman, G. Jackson, K.E. Gubbins, Mol. Phys. 65, 1057–1079 (1988)
S.H. Huang, M. Radosz, Ind. Eng. Chem. Res. 29, 2284 (1990)
S.H. Huang, M. Radosz, Ind. Eng. Chem. Res. 30, 1994 (1991)
J. Gross, G. Sadowski, Ind. Eng. Chem. Res. 40, 1244 (2001)
C.K. Zéberg-Mikkelsen, S.E. Quiñones-Cisneros, E.H. Stenby, Ind. Eng. Chem. Res. 40, 3848 (2001)
K.A.G. Schmidt, S.E. Quiñones-Cisneros, B. Kvamme, Energy Fuels 19, 1303–1313 (2005)
C.K. Zéberg-Mikkelsen, S.E. Quiñones-Cisneros, E.H. Stenby, Pet Sci Technol 20, 27 (2002)
S.E. Quiñones-Cisneros, C.K. Zéberg-Mikkelsen, E.H. Stenby, Fluid Ph. Equilibria 212, 233 (2003)
S. Quiñones-Cisneros, C. Zéberg-Mikkelsen, A. Baylaucq, C. Boned, Int. J. Thermophys 25, 1353–1366 (2004)
C.K. Zéberg-Mikkelsen, S.E. Quiñones-Cisneros, E.H. Stenby, Fluid Ph. Equilibria 194, 1191 (2002)
A.J. Queimada, S.E. Quinones-Cisneros, I.M. Marrucho, J.A.P. Coutinho, E.H. Stenby, Int. J. Thermophys 24, 1221 (2003)
S.P. Tan, H. Adidharma, B.F. Towler, M. Radosz, Ind. Eng. Chem. Res. 44, 8409 (2005)
S.P. Tan, H. Adidharma, B.F. Towler, M. Radosz, Ind. Eng. Chem. Res. 45, 2116 (2006)
S.E. Quiñones-Cisneros, C.K. Zéberg-Mikkelsen, J. Fernández, J. García, AIChE J. 52, 1600–1610 (2006)
W.A. Burgess, D. Tapriyal, I.K. Gamwo, B.D. Morreale, M.A. McHugh, R.M. Enick, Ind. Eng. Chem. Res. 51, 16721 (2012)
R. Macías-Salinas, Ind. Eng. Chem. Res. 57, 1109 (2018)
M. Almasi, H. Nasim, J. Chem. Thermodyn 89, 1 (2015)
M. Almasi, J. Therm. Anal. Calorim. 124, 399 (2016)
M. Abutaqiya, J. Zhang, F.M. Vargas, Fuel 235, 113 (2019)
Y. Khemka, M.I.L. Abutaqiya, W.G. Chapman, F.M. Vargas, Ind. Eng. Chem. Res. 59, 21994 (2020)
R. Bagherinia, M. Assareh, F. Feyzi, Fluid Ph. Equilibria 425, 21–30 (2016)
Y.H. Dehaghani, M. Assareh, F. Feyzi, Fluid Ph. Equilibria 1, 14 (2017)
J. Lohrenz, B.G. Bray, C.R. Clark, J. Pet. Technol. 231, 1117–1127 (1964)
K.S. Pedersen, A. Fredenslund, P.L. Christensen, P. Thomassen, Chem. Eng. Sci. 39, 1011–1016 (1984)
M.R. Riazi, T.E. Daubert, Ind. Eng. Chem. Res. 26, 755–759 (1987)
B.I.K. Lee, M.G. Kesler, AIChE J. 21, 510–527 (1975)
C.H. Whitson, In Norwegian Institute of Technology (University of Trondheim, Department of Petroleum and Chemical Engineering, 1983)
X. Liang, W. Yan, K. Thomsen, G.M. Kontogeorgis, Fluid Ph. Equilibria 406, 147–155 (2015)
X. Liang, W. Yan, K. Thomsen, G.M. Kontogeorgis, Fluid Ph. Equilibria 375, 254–268 (2014)
I. Polishuk, Ind. Eng. Chem. Res. 53, 14127 (2014)
M. Assareh, C. Ghotbi, M. Tavakkoli, G. Bashiri, Fluid Ph. Equilibria 408, 273–283 (2016)
T.H. Chung, L.L. Lee, K.E. Starling, Ind. Eng. Chem. Fundam 23, 8–13 (1984)
T.H. Chung, M. Ajlan, L.L. Lee, K.E. Starling, Ind. Eng. Chem. Res. 27, 671–679 (1988)
C.R. Wilke, J. Chem. Phys. 18, 517–519 (1950)
L. Zipperer, F. Herning, Das Gas-und Wasserfach 4, 49–59 (1936)
S.E. Quiñones-Cisneros, C.K. Zéberg-Mikkelsen, E.H. Stenby, Fluid Phase Equilib. 169, 249 (2000)
Funding
No funding was received during this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing interests or personal relationships that could have appeared to influence the work reported in this paper. This scientific manuscript has been extracted from a thesis. Younes Khoshnamvand is the graduated student. He has done the data gathering and model programming. Mehdi Assareh is the supervisors of the master thesis. He has proposed the modeling approach and supervised/helped its implementation. The corresponding author (Mehdi Assareh) is responsible for ensuring that the descriptions are accurate and agreed by all authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khoshnamvand, Y., Assareh, M. Application of Friction Theory and PC-SAFT for Estimation of Viscosity in Live Reservoir Fluid Systems. Int J Thermophys 43, 76 (2022). https://doi.org/10.1007/s10765-022-03004-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-03004-1