Abstract
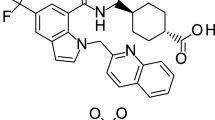
Prostaglandin E2 (PGE2) is a terminal prostaglandin in the cyclooxygenase (COX) pathway. Inhibition of PGE2 production may relieve inflammatory symptoms such as fever, arthritis, and inflammatory pain. We report here the profile of a novel selective PGE2 synthesis inhibitor, compound A [N-[(1S,3S)-3-carbamoylcyclohexyl]-1-(6-methyl-3-phenylquinolin-2-yl)piperidine-4-carboxamide], in animal models of pyrexia and inflammation. The compound selectively suppressed the synthesis of PGE2 in human alveolar adenocarcinoma cell line A549 cells and rat macrophages. In the lipopolysaccharide-induced pyrexia model, this compound selectively reduced PGE2 production in cerebrospinal fluid and showed an anti-pyretic effect. In the adjuvant-induced arthritis model, compound A therapeutically decreased foot swelling in the established arthritis. Our data demonstrates that selective suppression of PGE2 synthesis shows anti-pyretic and anti-inflammatory effects, suggesting that selective PGE2 synthesis inhibitors can be applied as an alternative treatment to nonsteroidal, anti-inflammatory drugs (NSAIDs) or COX-2-selective inhibitors.






Similar content being viewed by others
References
Langman, M.J., J. Weil, P. Wainwright, D.H. Lawson, M.D. Rawlins, R.F. Logan, M. Murphy, M.P. Vessey, and D.G. Colin-Jones. 1994. Risks of bleeding peptic ulcer associated with individual non-steroidal anti-inflammatory drugs. Lancet 343(8905): 1075–1078.
Masferrer, J.L., B.S. Zweifel, P.T. Manning, S.D. Hauser, K.M. Leahy, W.G. Smith, P.C. Isakson, and K. Seibert. 1994. Selective inhibition of inducible cyclooxygenase 2 in vivo is antiinflammatory and nonulcerogenic. Proc Natl Acad Sci U S A 91(8): 3228–3232.
Seibert, K., Y. Zhang, K. Leahy, S. Hauser, J. Masferrer, W. Perkins, L. Lee, and P. Isakson. 1994. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc Natl Acad Sci U S A 91(25): 12013–12017.
Bombardier, C., L. Laine, A. Reicin, D. Shapiro, R. Burgos-Vargas, B. Davis, R. Day, M.B. Ferraz, C.J. Hawkey, M.C. Hochberg, VIGOR Study Group, et al. 2000. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. N Engl J Med 343(21): 1520–1528. 2 p following 1528.
Goldstein, J.L., G.M. Eisen, N. Agrawal, W.F. Stenson, J.D. Kent, and K.M. Verburg. 2004. Reduced incidence of upper gastrointestinal ulcer complications with the COX-2 selective inhibitor, valdecoxib. Aliment Pharmacol Ther 20(5): 527–538.
Singh, G., J.G. Fort, J.L. Goldstein, R.A. Levy, P.S. Hanrahan, A.E. Bello, L. Andrade-Ortega, C. Wallemark, N.M. Agrawal, G.M. Eisen, et al. 2006. Celecoxib versus naproxen and diclofenac in osteoarthritis patients: SUCCESS-I Study. Am J Med 119(3): 255–266.
Bresalier, R.S., R.S. Sandler, H. Quan, J.A. Bolognese, B. Oxenius, K. Horgan, C. Lines, R. Riddell, D. Morton, A. Lanas, et al. 2005. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 352(11): 1092–1102.
Couzin, J. 2004. Drug safety. Withdrawal of Vioxx casts a shadow over COX-2 inhibitors. Science 306(5695): 384–385.
Nussmeier, N.A., A.A. Whelton, M.T. Brown, R.M. Langford, A. Hoeft, J.L. Parlow, S.W. Boyce, and K.M. Verburg. 2005. Complications of the COX-2 inhibitors parecoxib and valdecoxib after cardiac surgery. N Engl J Med 352(11): 1081–1091.
Cheng, Y., S.C. Austin, B. Rocca, B.H. Koller, T.M. Coffman, T. Grosser, J.A. Lawson, and G.A. FitzGerald. 2002. Role of prostacyclin in the cardiovascular response to thromboxane A2. Science 296(5567): 539–541.
Cheng, Y., M. Wang, Y. Yu, J. Lawson, C.D. Funk, and G.A. Fitzgerald. 2006. Cyclooxygenases, microsomal prostaglandin E synthase-1, and cardiovascular function. J Clin Invest 116(5): 1391–1399.
Kamei, D., K. Yamakawa, Y. Takegoshi, M. Mikami-Nakanishi, Y. Nakatani, S. Oh-Ishi, H. Yasui, Y. Azuma, N. Hirasawa, K. Ohuchi, et al. 2004. Reduced pain hypersensitivity and inflammation in mice lacking microsomal prostaglandin e synthase-1. J Biol Chem 279(32): 33684–33695.
Trebino, C.E., J.L. Stock, C.P. Gibbons, B.M. Naiman, T.S. Wachtmann, J.P. Umland, K. Pandher, J.M. Lapointe, S. Saha, M.L. Roach, et al. 2003. Impaired inflammatory and pain responses in mice lacking an inducible prostaglandin E synthase. Proc Natl Acad Sci U S A 100(15): 9044–9049.
Xu, D., S.E. Rowland, P. Clark, A. Giroux, B. Cote, S. Guiral, M. Salem, Y. Ducharme, R.W. Friesen, N. Methot, et al. 2008. MF63 [2-(6-chloro-1H-phenanthro[9,10-d]imidazol-2-yl)-isophthalonitrile], a selective microsomal prostaglandin E synthase-1 inhibitor, relieves pyresis and pain in preclinical models of inflammation. J Pharmacol Exp Ther 326(3): 754–763.
Chan, C.C., S. Boyce, C. Brideau, S. Charleson, W. Cromlish, D. Ethier, J. Evans, A.W. Ford-Hutchinson, M.J. Forrest, J.Y. Gauthier, et al. 1999. Rofecoxib [Vioxx, MK-0966; 4-(4'-methylsulfonylphenyl)-3-phenyl-2-(5H)-furanone]: a potent and orally active cyclooxygenase-2 inhibitor. Pharmacological and biochemical profiles. J Pharmacol Exp Ther 290(2): 551–560.
Penning, T.D., J.J. Talley, S.R. Bertenshaw, J.S. Carter, P.W. Collins, S. Docter, M.J. Graneto, L.F. Lee, J.W. Malecha, J.M. Miyashiro, et al. 1997. Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benze nesulfonamide (SC-58635, celecoxib). J Med Chem 40(9): 1347–1365.
Talley, J.J., D.L. Brown, J.S. Carter, M.J. Graneto, C.M. Koboldt, J.L. Masferrer, W.E. Perkins, R.S. Rogers, A.F. Shaffer, Y.Y. Zhang, et al. 2000. 4-[5-Methyl-3-phenylisoxazol-4-yl]- benzenesulfonamide, valdecoxib: a potent and selective inhibitor of COX-2. J Med Chem 43(5): 775–777.
Ushiyama, S., T. Yamada, Y. Murakami, S. Kumakura, S. Inoue, K. Suzuki, A. Nakao, A. Kawara, and T. Kimura. 2008. Preclinical pharmacology profile of CS-706, a novel cyclooxygenase-2 selective inhibitor, with potent antinociceptive and anti-inflammatory effects. Eur J Pharmacol 578(1): 76–86.
Leclerc, P., S.C. Pawelzik, H. Idborg, L. Spahiu, C. Larsson, P. Stenberg, M. Korotkova, and P.J. Jakobsson. 2013. Characterization of a new mPGES-1 inhibitor in rat models of inflammation. Prostaglandins Other Lipid Mediat 102–103: 1–12.
Kawakami K, Kiho T, Tengeiji A, Shimizu H, Kuroyanagi J, Kitamura T, Sugita R, Kubota K, Sugimoto K (2013) Novel quinoline derivative. WO 2013146970 A1
Yamaki, K., T. Yonezawa, and K. Ohuchi. 2000. Signal transduction cascade in staurosporine-induced prostaglandin E(2) production by rat peritoneal macrophages. J Pharmacol Exp Ther 293(1): 206–213.
Winder, C.V., L.A. Lembke, and M.D. Stephens. 1969. Comparative bioassay of drugs in adjuvant-induced arthritis in rats: flufenamic acid, mefenamic acid, and phenylbutazone. Arthritis Rheum 12(5): 472–482.
Trebino, C.E., J.D. Eskra, T.S. Wachtmann, J.R. Perez, T.J. Carty, and L.P. Audoly. 2005. Redirection of eicosanoid metabolism in mPGES-1-deficient macrophages. J Biol Chem 280(17): 16579–16585.
Monrad, S.U., F. Kojima, M. Kapoor, E.L. Kuan, S. Sarkar, G.J. Randolph, and L.J. Crofford. 2011. Genetic deletion of mPGES-1 abolishes PGE2 production in murine dendritic cells and alters the cytokine profile, but does not affect maturation or migration. Prostaglandins Leukot Essent Fat Acids 84(3–4): 113–121.
Kapoor, M., F. Kojima, M. Qian, L. Yang, and L.J. Crofford. 2006. Shunting of prostanoid biosynthesis in microsomal prostaglandin E synthase-1 null embryo fibroblasts: regulatory effects on inducible nitric oxide synthase expression and nitrite synthesis. FASEB J 20(13): 2387–2389.
Boulet, L., M. Ouellet, K.P. Bateman, D. Ethier, M.D. Percival, D. Riendeau, J.A. Mancini, and N. Methot. 2004. Deletion of microsomal prostaglandin E2 (PGE2) synthase-1 reduces inducible and basal PGE2 production and alters the gastric prostanoid profile. J Biol Chem 279(22): 23229–23237.
Leclerc, P., H. Idborg, L. Spahiu, C. Larsson, N. Nekhotiaeva, J. Wannberg, P. Stenberg, M. Korotkova, and P.J. Jakobsson. 2013. Characterization of a human and murine mPGES-1 inhibitor and comparison to mPGES-1 genetic deletion in mouse models of inflammation. Prostaglandins Other Lipid Mediat 107: 26–34.
Bruno, A., L. Di Francesco, I. Coletta, G. Mangano, M.A. Alisi, L. Polenzani, C. Milanese, P. Anzellotti, E. Ricciotti, M. Dovizio, et al. 2010. Effects of AF3442 [N-(9-ethyl-9H-carbazol-3-yl)-2-(trifluoromethyl)benzamide], a novel inhibitor of human microsomal prostaglandin E synthase-1, on prostanoid biosynthesis in human monocytes in vitro. Biochem Pharmacol 79(7): 974–981.
Wang, M., W.L. Song, Y. Cheng, and G.A. Fitzgerald. 2008. Microsomal prostaglandin E synthase-1 inhibition in cardiovascular inflammatory disease. J Intern Med 263(5): 500–505.
Engblom, D., S. Saha, L. Engstrom, M. Westman, L.P. Audoly, P.J. Jakobsson, and A. Blomqvist. 2003. Microsomal prostaglandin E synthase-1 is the central switch during immune-induced pyresis. Nat Neurosci 6(11): 1137–1138.
Saha, S., L. Engstrom, L. Mackerlova, P.J. Jakobsson, and A. Blomqvist. 2005. Impaired febrile responses to immune challenge in mice deficient in microsomal prostaglandin E synthase-1. Am J Physiol Regul Integr Comp Physiol 288(5): R1100–R1107.
Paulson, S.K., J.Y. Zhang, A.P. Breau, J.D. Hribar, N.W. Liu, S.M. Jessen, Y.M. Lawal, J.N. Cogburn, C.J. Gresk, C.S. Markos, et al. 2000. Pharmacokinetics, tissue distribution, metabolism, and excretion of celecoxib in rats. Drug Metab Dispos 28(5): 514–521.
Pulichino, A.M., S. Rowland, T. Wu, P. Clark, D. Xu, M.C. Mathieu, D. Riendeau, and L.P. Audoly. 2006. Prostacyclin antagonism reduces pain and inflammation in rodent models of hyperalgesia and chronic arthritis. J Pharmacol Exp Ther 319(3): 1043–1050.
Acknowledgments
We are grateful to Miwako Takeuchi for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sugita, R., Kuwabara, H., Sugimoto, K. et al. A Novel Selective Prostaglandin E2 Synthesis Inhibitor Relieves Pyrexia and Chronic Inflammation in Rats. Inflammation 39, 907–915 (2016). https://doi.org/10.1007/s10753-016-0323-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-016-0323-5