Abstract
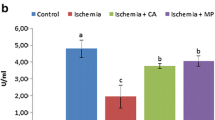
Acute arterial occlusions via different vascular pathologies are the main causes of spinal cord ischemia. We investigated neuroprotective effects of syringic acid on spinal cord ischemia injury in rats. Rats were divided into four groups: (I) sham-operated control rats, (II) spinal cord ischemia group, (III) spinal cord ischemia group performed syringic acid, and (IV) spinal cord ischemia group performed methylprednisolone intraperitoneally. Spinal cord ischemia was performed by the infrarenal aorta cross-clamping model. The spinal cord was removed after the procedure. The biochemical and histopathological changes were observed within the samples. Functional assessment was performed for neurological deficit scores. A significant decrease was seen in malondialdehyde levels in group III as compared to group II (P < 0.05). Besides these, nuclear respiratory factor-1 and superoxide dismutase activity of group III were significantly higher than group II (P < 0.05). In histopathological samples, when group III was compared with group II, there was a significant decrease in numbers of apoptotic neurons (P < 0.05). In immunohistochemical staining, BECN1 and caspase-3-immunopositive neurons were significantly decreased in group III compared with group II (P < 0.05). The neurological deficit scores of group III were significantly higher than group II at twenty-fourth hour of ischemia (P < 0.05). Our study revealed that syringic acid pretreatment in spinal cord ischemia/reperfusion reduced oxidative stress and neuronal degeneration as a neuroprotective agent. Ultrastructural studies are required for syringic acid to be developed as a promising therapeutic agent to be utilized for human spinal cord ischemia in the future.





Similar content being viewed by others
References
Weidauer, S., M. Nichtweiß, E. Hattingen, J. Berkefeld. 2014. Spinal cord ischemia: etiology, clinical syndromes and imaging features. Neuroradiology 16.
Aslan, A., M. Cemek, M.E. Buyukokuroglu, K. Altunbas, O. Bas, Y. Yurumez, and M. Cosar. 2009. Dantrolene can reduce secondary damage after spinal cord injury. European Spine Journal 18(10): 1442–1451.
Guven, M., T. Akman, A.B... Aras, A.U. Yener, M.H. Sehitoglu, Y. Yuksel, U.H. Golge, E. Karavelioglu, E. Bal, and M. Cosar. 2015. The neuroprotective effect of kefir on spinal cord ischemia/reperfusion injury in rats. Journal of the Korean Neurosurgical Society 57(3).
Carden, D.L., and D.N. Granger. 2000. Pathophysiology of ischaemia-reperfusion injury. Journal of Pathology 190: 255–266.
Cho, J.Y., J.H. Moon, K.Y. Seong, and K.H. Park. 1998. Antimicrobial activity of 4-hydroxybenzoic acid and trans 4-hydroxycinnamic acid isolated and identified from rice hull. Bioscience Biotechnology and Biochemistry 62: 2273–2276.
Guven, M., A.B... Aras, N. Topaloglu, A. Ozkan, H.M. Sen, Y. Kalkan, A. Okuyucu, A. Akbal, F. Gokmen, and M. Cosar. 2015. The protective effect of syringic acid on ischemia injury in rat brain. Turkish Journal of Medical Sciences 45: 233–240.
Simonyi, A., Q. Wang, R.L. Miller, M. Yusof, P.B. Shelat, A.Y. Sun, and G.Y. Sun. 2005. Polyphenols in cerebral ischemia: novel targets for neuroprotection. Molecular Neurobiology 31: 135–147.
Scalbert, A., and G. Williamson. 2000. Dietary intake and bioavailability of polyphenols. Journal of Nutrition 130: 2073–2085.
Hudson, E.A., P.A. Dinh, T. Kokubun, M.S. Simmonds, and A. Gescher. 2000. Characterization of potentially chemopreventive phenols in extracts of brown rice that inhibit the growth of human breast and colon cancer cells. Cancer Epidemiology, Biomarkers and Prevention 9: 1163–1170.
Kumar, S., P. Prahalathan, and B. Raja. 2012. Syringic acid ameliorates (L)-NAME-induced hypertension by reducing oxidative stress. Naunyn-Schmiedeberg’s Archives of Pharmacology 385: 1175–1184.
Morton, L.W., K.D. Croft, I.B. Puddey, and L. Byrne. 2000. Phenolic acids protect low density lipoproteins from peroxynitrite-mediated modification in vitro. Redox Report 5: 124–125.
Lafci, G., H.S. Gedik, K. Korkmaz, H. Erdem, O.F. Cicek, and O.A. Nacar. 2013. Efficacy of iloprost and montelukast combination on spinal cord ischemia/reperfusion injury in a rat model. Journal of Cardiothoracic Surgery 8: 64.
Basso, D.M., M.S. Beattie, and J.C. Bresnahan. 1995. A sensitive and reliable locomotor rating scale for open field-testing in rats. Journal of Neurotrauma 12: 1–21.
Gutsaeva, D.R., M.S. Carraway, H.B. Suliman, I.T. Demchenko, H. Shitara, H. Yonekawa, and C.A. Piantadosi. 2008. Transient hypoxia stimulates mitochondrial biogenesis in brain subcortex by a neuronal nitric oxide synthase-dependent mechanism. Journal of Neuroscience 28: 2015–2024.
Sun, Y., L.W. Oberley, and Y. Li. 1998. A simple method for clinical assay of superoxide dismutase. Clinical Chemistry 34: 497–500.
Buege, J.A., and S.D. Aust. 1978. Microsomal lipid peroxidation. Methods in Enzymology 52: 302–310.
Naoko, K., M. Kawaguchi, T. Horiuchi, S. Inoue, T. Sakamoto, M. Nakamura, N. Konishi, and H. Furuya. 2005. An evaluation of white matter injury after spinal cord ischemia in rats: a comparison with gray matter injury. Anesthesia and Analgesia 100: 847–854.
Ginsberg, M.D. 1995. Review: neuroprotection in brain ischemia: an update (part I). The Neuroscientist 1: 95–103.
Mauney, M.C., L.H. Blackbourne, S.E. Langenburg, S.A. Buchanan, I.L. Kron, and G.G. Tribble. 1995. Prevention of spinal cord injury after repair of the thoracic or thoracoabdominal aorta. Annals of Thoracic Surgery 59: 245–252.
Von Moltke, L.L., J.L. Weemhoff, E. Bedir, I.A. Khan, J.S. Harmatz, P. Goldman, and D.J. Greenblatt. 2004. Inhibition of human cytochromes P450 by components of ginkgo biloba. The Journal of Pharmacy and Pharmacology 56: 1039–1044.
Kang, S.S., J.Y. Lee, Y.K. Choi, S.S. Song, J.S. Kim, S.J. Jeon, Y.N. Han, K.H. Son, and B.H. Han. 2005. Neuroprotective effects of naturally occurring bioflavonoids. Bioorganic and Medicinal Chemistry Letters 15: 3588–3591.
Ozen, O.A., M. Cosar, O. Sahin, H. Fidan, O. Eser, and H. Mollaoglu. 2008. The protective effect of fish n-3 fatty acids on cerebral ischemia in rat prefrontal cortex. Neurological Science 29: 147–152.
Yin, W., A.P. Signore, M. Iwai, G. Cao, Y. Gao, and J. Chen. 2008. Rapidly increased neuronal mitochondrial biogenesis after hypoxic-ischemic brain injury. Stroke 39: 3057–3063.
Porter, A.G., and R.U. Janicke. 1999. Emerging roles of caspase-3 in apoptosis. Cell Death and Differentiation 6: 99–104.
Li, P., D. Nijhawan, I. Budihardjo, S.M. Srinivasula, M. Ahmad, and E.S. Alnemri. 1997. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91: 479–489.
Acknowledgments
The authors thank the experimental research center of Canakkale Onsekiz Mart University.
Conflict of Interest
The authors declare no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The authors received no financial support for the research and/or authorship of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tokmak, M., Yuksel, Y., Sehitoglu, M.H. et al. The Neuroprotective Effect of Syringic Acid on Spinal Cord Ischemia/Reperfusion Injury in Rats. Inflammation 38, 1969–1978 (2015). https://doi.org/10.1007/s10753-015-0177-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-015-0177-2