Abstract
Knowledge of the genetic variability of different species for conservation purposes and quality seedling production after longer storage time is important. The aim of the research paper was to examine the effect of longer seed storage periods (19–30 year) on seed viability, seed vigour and initial growth of seedlings from five provenances of Pterolobium stellatum (Forssk). Seeds collected from Dabena, Gambo, Agaro, Bishofitu, and Merti localities and stored for 19, 19, 21, 25, and 30 years, respectively used for the study. Different seed dormancy-breaking treatments such as; soaking in cold water for 48 h (CW-48); soaking in hot water for 10 min (HW-10); 20 min (HW-20) and mechanical scarification; i.e., nicking (NK) used for the germination of the seeds. A randomized complete block design was used for the experiment and the study undertaken in a greenhouse. For each treatment, four replications and for each replication, 50 seeds were used. One-way analysis of variance was used for germination index, early seedling growth and seed vigour index data. The results showed that for all the provenance seeds stored for different periods, NK treatment resulted relatively in a higher mean germination percentage and germination indices as compared to the other pre-treatments. The seed vigour index result showed significant differences among the different provenances (P < 0.001). Similarly, there were significant differences among the early growth of seedlings germinated from different provenance seeds (P < 0.001). The seeds collected from Merti provenance and stored for 30 years and Dabena provenance, stored for 19 years, have relatively resulted in higher mean germination index, seed vigour and seedling growth relative to the other studied provenance seeds. We observed that provenance had a more significant influence on germination, seed vigour and seedling growth than the seed storage period for the P. stellatum. Finally it is recommended that despite there is inter population variation, the seeds of P. stellatum can be stored for a longer time and application of nicking as a dormancy breaking treatment could enhance seed germination of the species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pterolobium stellatum (Forssk.) Brenan, which belongs to the Fabaceae family, is a perennial tall scandent woody species. It is an evergreen dicotyledonous flowering plant and semierect multistemmed climbing woody thorny shrub that can reach 2–15 m high (Janson and Cardon 2005; Ward and Spellenberg 1988). The species is native to Africa, and its distribution extends from South Africa to Eastern Africa (Janson and Cardon 2005; Teketay 1998). Traditionally, the species is used as a medicinal plant to treat tuberculosis, pneumonia, and other related respiratory diseases (Kigen et al. 2016; Getahun 1976). Leaves and bark are used for ink production (Janson and Cardon 2005). It is also used to treat infertility, epilepsy, and neuroglia (Kigen et al. 2016; Ragunthan and Abay 2009) and to treat sexually transmitted diseases (Njoroge and Bussmann 2006). Some chemical analyses of the plant parts showed that the species possess cytotoxic and anti-diarrhoea properties (Andualem et al. 2014). The plant is also used for animal fodder, live fences, firewood, charcoal, ornamental tree, and soil conservation purposes to have a healthy environment (Alemayehu et al. 2015; Zegeye et al. 2005).
The storage of seeds for a long time is one of the strategies of ex-situ conservation. Keeping seed viability in a conducive environment is an excellent alternative for maintaining or minimizing the loss of physiological quality of seeds during storage until sowing as an ex-situ conservation strategy (Solberg et al. 2020a, b; Silva et al 2019; Vijay 2015; Pradhan and Badola 2008; Ellis et al. 1991). For the conservation of plant genetic resources through ex-situ conservation, knowledge of the characteristics of the seed is important (Pradhan and Badola 2012). Different factors, such as temperature, nature of the seeds, moisture, insects, etc., could affect the viability of the seeds in a storage room, and pre-storage conditions also affect seed longevity (Mdlalose et al. 2021; Solberg et al. 2020a, b; De Vitis et al. 2020; Rao et al. 2017; Pradhan and Badola 2012; Delouche et al. 1973). Seed moisture, vigour, germination percentage, rate of germination, etc., are some of the most important parameters for assessing the seed quality of a species after storage of the seed (Mdlalose et al. 2021). Seed dormancy problems could also affect the germination of the seeds of different species. Seed coat-imposed dormancy is an ecological mechanism that allows the seed to germinate only when conditions are favourable for supporting seedling growth (Argel and Paton 1999). In the case of legume species, the impermeable seed coat is considered to be the main dormancy problem for the germination of seeds (Grubb and Coomes 1997; Albrecht 1993). Therefore, to break seed dormancy problems using pre-sowing treatments, rapid and uniform germination and high germination capacity can be obtained (Huang and Gutterman 2000; Schutz and Rave 1999; Teketay and Tigabu 1996; Teketay 1996).
Different pre-sowing treatments, such as cold stratification, mechanical disruption, or acid and hot water treatments are widely used to break seed dormancy problems, and these techniques can improve the germination of seeds within a short period (Tadros et al. 2011). Research shows that the seeds of P. stellatum have a prolonged dormancy problem, which is associated with the hard seed coat cover of the species (Jemaneh 2019; Teketay 1998). Some authors have indicated that if the seed of P. stellatum is stored properly and placed in airtight containers, cool, dry areas and insect-free places, it can stay for a longer period without losing its viability (Teketay 1998; Bein et al. 1996). However, knowledge of the seed longevity of the species of the different provenances of P. stellatum and its effect on the germination, seed vigour and seedling growth are lacking. Such knowledge is necessary for conservation purposes for farmers and seed enterprises. The objectives of this study were (1) to assess the impacts of different seed dormancy-breaking treatments on the germination of the seeds of different provenances of P. stellatum that were stored for 19–30 years (2) to evaluate the viability of the seeds of different provenances of P. stellatum after different periods of storage (3) to examine the seed vigour of the different provenances seeds (4) to assess the quality of the seedlings from the different provenances that were stored for different periods. At the beginning of the research, it was hypothesized that (1) different seed dormancy-breaking treatments have different impacts on the germination of the seeds of P. stellatum that were stored for different periods and (2) the storage period and provenances do not have an influence on the viability of the seeds, on seed vigour and seedling quality of P. stellatum.
Materials and methods
Seed sources and the storage period
The seeds of P. stellatum were collected in Ethiopia from Merti, Bishoftu, Agaro, Dabena, and Gambo localities in 1990, 1996, 2000, 2002, and 2002, respectively. It means that the storage ages of the collected seeds from Dabena, Gambo, Agaro, Bishofitu, and Merti localities were 19, 19, 21, 25, and 30 years, respectively. The mean altitude (meters above sea level) of the area where the provenance seeds of Merti, Bishoftu, Agaro, Dabena and Gambo collected were about 1550 m, 1886 m, 1744 m, 1823 m and 2059 m, correspondingly. A map of the localities where the different seeds of P. stellatum were collected is presented in Fig. 1. The documented pieces of information showed that the collected seeds from Agaro, Dabena, Bishoftu, Gambo, and Merti localities were air-dried under shade and stored in a seed store that has a temperature of 5 °C. While storing the seeds have a moisture percentage of 5.6%, 5.4%, 6.3%, 5.3%, and 5%, respectively. According to the information gathered in the seed laboratory, the mean germination percentages of the seeds of the different provenances using nicking as a dormancy-breaking treatment at the time of the seed collection period for Agaro, Dabena, Bishoftu, Gambo, and Merti were 51%, 64%, 65%, 58%, and 65%, respectively.
Germination experiment and seed dormancy-breaking treatments
Randomized complete block design (RCBD) was used for the experiment and the study was undertaken in a greenhouse. Each treatment had four replications (tray boxes), and for each replication, 50 seeds were used. It means a total of 200 seeds were used for a single treatment. The different dormancy-breaking treatments applied for seed germination were (a) soaking in hot water for 10 min (HW-10), (b) soaking in hot water for 20 min (HW-20), (c) soaking in cold water for 48 h (CW-48), (d) mechanical scarification, i.e., nicking (NK), and e) control (CN), i.e., without any pre-sowing treatment. These dormancy breaking treatments are selected since the seed of the species has a hard seed coat cover, and the treatments are mostly recommended.
The germination experiment was performed using a tray box filled with sand (i.e., the sand has a thickness of approximately 10 cm) in a greenhouse. The sand that was used as a germination medium was cleaned using tape water through washing before the experiment. The seeds were sown in the sand-filled tray and then carefully covered by the sand. Watering of sown seeds was undertaken once a day in the morning tills the end of the experiment. The germination study was attended between 17.03.2021 and 12.04.2021, i.e., for 26 days. After germination, the seedlings were kept in the tray to grow until the end of the experiment and used to evaluate the seed vigour (Fig. 2).
Data collection and analysis
Data on the germination of the emerged seedlings were collected daily. At the end of the experiment, i.e., 13/04/2021, each of the individual germinated and growing seedlings of the different provenances and different treatments were carefully uprooted. The root length and shoot length of uprooted individual seedlings were measured using a ruler, independently. Viability test of un-germinated seeds was not undertaken since the experiment was performed using sand, that serves as a media of germination, and for this reason it was difficult to find the buried and un-germinated seeds.
The collected germination and seedling shoot length and root length data were encoded in an MS Excel sheet. The germination data was arc-sine transformed before the analysis. The normality of the distribution of the data was evaluated. One-way analysis of variance (ANOVA) was used for the handling of the data. When there were significant differences, Fisher's least significance difference (LSD) test was used for the mean separation at a significance level of P = 0.05. SigmaPlot 13 (Systat Software, Inc., San Jose, CA, USA) program was used for the statistical analysis.
The mean final germination percentage (GP) using methodology of Scott et al. (1984), mean germination time (MGT) (Orchard 1977) and mean germination index (GI) (Bench et al. 1991) were analysed using the Eq. 1, 2, and 3, correspondingly. While the seed vigour index was calculated using the formula suggested by Abdul-Baki (1973) as presented in Eq. 4.
The final germination percentage (GP) was calculated using Formula (1):
where TG = total number of seeds germinated.
TS = total number of sown seeds.
The mean germination time (MGT) of seeds was calculated using Formula (2):
where N = number of newly germinated seeds.
T = time from the beginning of the experiment.
The germination index (GI) was calculated using Formula (3):
where N = number of newly germinated seeds,
T = time from the beginning of the experiment.
The seed vigour Index (VI) was calculated using the formula (4)
where SL = Seedling length (root length + shoot length).
GP = Germination percentage.
Results
Effect of pre-sowing treatments on seed germination
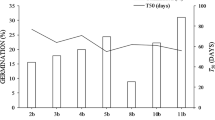
The effects of the different seed dormancy-breaking treatments on the mean germination percentage of P. stellatum seeds stored for different periods is presented in Table 1. The results revealed that, despite their variation in their storage periods, for all the studied provenances seeds, the NK treatment resulted in a higher mean germination percentage relative to the other dormancy-breaking treatments applied to the seeds of P. stellatum (Table 1). Similarly, the mean germination index result, which combines the germination percentage and mean germination time of the seeds of the different provenances of P. stellatum, shows that seeds treated with NK also resulted in a higher mean germination index relative to the other seed dormancy-breaking treatments applied to the seeds of the species (Table 2). The seeds collected from Bishoftu provenance and stored for 25 years sown after NK treatment resulted in the highest mean germination percentage relative to the other provenances (Table 1). The seeds from Dabena provenance that is treated with CW-48, HW-20, HW-10 and CN have relatively higher mean germination percentages compared to the mean germination percentage of the seeds of other provenances (Table 1). However, the Gambo provenance which has the same period of storage as the Dabena provenance with all seed dormancy-breaking treatments has resulted in a relatively lower mean germination percentage as compared to the other provenance seeds (Table 1).
Despite, the NK treatment resulted in a relatively higher mean germination percentage (Table 1) and germination index (Table 2) for all the provenance, the one-way ANOVA result revealed no significant differences among the different seed dormancy breaking treatments (HW-10, HW-20, CW-48, NK, and CN) within the Agaro provenance (P = 0.121, Table 1), within Gambo provenance (P = 0.443, Table 1), and Merti provenance (P = 0.27, Table 1). On the other hand, the statistical analysis results showed a significant difference in the mean germination percentage between the seed dormancy-breaking treatments applied within the Dabena provenance (P = 0.05, Table 1) and within Bishoftu provenance (P = 0.001, Table 1). Figure 3 shows the one-way ANOVA result on the mean germination percentage of the different provenance seeds with different pre-sowing treatments. Accordingly, the one-way ANOVA analysis result revealed no significant differences in the mean germination percentage of the seeds among the Agaro, Dabena, Bishoftu, Gambo, and Merti provenances that were treated with CW-48 (P = 0.636), HW-10 (P = 0.186), CN seeds (P = 0.295) (Fig. 3). However, the one-way ANOVA results revealed significant differences in the mean germination percentage of the seeds among the provenances of Agaro, Dabena, Bishoftu, Gambo and Merti that were treated with HW-20 (P = 0.006) and NK (P = 0.01, Fig. 3).
Seed viability and seed vigour
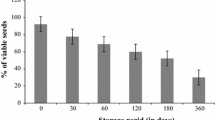
The seed laboratory test result on the mean germination percentage of the different provenances of P. stellatum seeds in different years, with NK treatment, is presented in Fig. 4. At the time of seed collection, the mean germination percentage of the different provenance seeds of P. stellatum with NK treatment for Dabena, Agaro, Bishoftu, Merti and Gambo provenances was 64%, 51%, 65%, 65%, and 58%, respectively (Fig. 5). In the year 2021, the mean germination percentages of the seeds with NK treatment for the Dabena, Agaro, Bishoftu, Merti and Gambo provenances were 40%, 31%, 44%, 29%, and 29%, respectively. This result further indicated that the mean annual viability losses of the seeds of P. stellatum collected from Dabena, Agaro, Bishoftu, Merti, and Gambo provenances were 1.1%, 0.91%, 1.9%, 1.2% and 1.2%, respectively.
Mean germination percentage of the different provenance seeds of Pterolobium stellatum using mechanical scarification; i.e., nicking, as a pre-sowing treatment in the seed laboratory (germination results between 1990 and 2020) and in the final germination with the present experiment in the year 2021
The seed vigour index analysis result for the Agaro, Dabena, Bishoftu, Gambo, and Merti provenances is presented in Fig. 5. The analysis result showed that the mean seed vigour index for the Agaro, Dabena, Bishoftu, Gambo, and Merti provenances were 613, 852, 685, 536, and 714, respectively. The statistical one-way ANOVA result revealed significant differences among the different seed provenances stored for different periods (Fig. 5). The significant differences in the seed vigour indices were between the provenances of Dabena and Gambo; Dabena and Agaro; Dabena and Bishoftu; Gambo and Merti and Bishoftu and Gambo seeds (P<0.05, Fig. 5). However, the statistical analysis results showed no significant differences in the seed vigour index between the Dabena and Merti; Merti and Agaro; Merti and Bishoftu; Bishoftu and Agaro; and Agaro and Gambo provenances (P˃0.05, Fig. 5).
Early growth of seedlings from the different provenances
The analysis result showed that the mean root lengths of the seedlings germinated and grown from Gambo, Dabena, Agaro, Bishoftu and Merti provenances where their seeds stored for 19, 19, 21, 25 and 30 years were 8.6 cm, 9.3 cm, 8.8 cm, 7.5 cm and 10.2 cm, correspondingly. The one-way ANOVA analysis result showed significant differences in the mean root lengths of the germinated seedlings from the seeds of the different provenances (P = < 0.001, Fig. 6). Accordingly, the significant differences in the mean seedling root lengths were between the provenance seeds of Merti and Agaro; Merti and Gambo and Merti and Bishoftu (Fig. 6). However, the statistical one-way ANOVA result showed no significant differences in the mean root lengths of the seedlings that were germinated and grown from Agaro, Gambo and Dabena provenances (Fig. 6, P > 0.05). The result on the mean shoot lengths of the seedlings germinated and grown from the provenance seeds of Gambo, Dabena, Agaro, Bishoftu and Merti provenances were 16.2 cm, 17.4 cm 17.5 cm, 17.8 cm and 18.4 cm, respectively. The one-way ANOVA analysis results on the mean shoot lengths of the seedlings grown from the different provenance seeds are presented in Fig. 7. The analysis result showed significant differences in the mean shoot lengths of seedlings grown from the different provenance seeds (P < 0.001), and the significant differences were between Merti and Gambo; and Merti and Dabena provenances (Fig. 7). However, the statistical analysis result revealed no significant differences in the mean shoot lengths of the seedlings grown from the provenances seeds between Merti and Agaro; Merti and Bishoftu and Dabena and Bishoftu (Fig. 7, P > 0.05).
Discussion
The present study finding showed that for all the provenance of P. stellatum seeds that were stored for different periods, NK as a dormancy-breaking treatment, resulted, in relatively a higher mean germination percentage and mean germination index relative to the other seed dormancy-breaking treatments applied for the study. This result corresponds with the findings of Teketay et al. (1998) and Jemaneh (2019), who also in their findings indicated that nicking is the best treatment to break the seed dormancy problem of freshly collected P. stellatum seeds. Rodrigues-Junior et al. (2020) and Burrows et al. (2019) indicated that most of the Fabaceae family has a problem of physical dormancy, which is related to blockage of water uptake during the dormant state. Similar to many other Fabaceae species, the seeds of P. stellatum have a dormancy problem, which can be solved using NK. The species (P. stellatum) belongs to the Fabaceae family and different findings also recommended that scarification (mechanical treatment) as the recommended pre-sowing preparation for the dormancy breaking of this family species (Parera and Ruiz 2003; Seng and Cheong 2020; Schnadelbach et al. 2016; Iroko et al. 2021; Perez-Garcia 2009; Fulbright and Flenniken 1987). Some species have a problem with seed germination since part of the seed testa slows the embryo water intake, which slows seed germination (Burrows et al. 2019). Generally, the present results showed that the seeds of P. stellatum had a hard seed coat cover that inhibits water entrance to the cotyledon, which affects seed germination, and this problem can be solved with NK treatment.
Knowledge of inter-population variation can improve the understanding of the mechanisms regulating seed germination (Bhatt 2021). The present result shows that despite the Gambo provenance and Dabena provenance seeds were stored for equal periods (19 years), their mean germination percentage and mean germination index of the Gambo provenance seeds with all the dormancy breaking treatments were relatively lower than those of the Dabena provenance seeds. This differences could be associated with the inter-population variation of the two provenances. The seed vigour index result also proved that the seeds of Dabana provenance were more vigorous than the Gambo provenance despite both having equal storage time. These results further could show that provenance affects the seed germination and seed vigour of P. stellatum which shows a variation on the provenances of the species. The differences in the mean germination of the P. stellatum seeds collected from the Dabena provenance relative to Gambo provenance could be associated with their differences in the imbibition patterns of the seeds of the provenances. Differences in the texture of the seed, temperature, seed coat thickness, seed coat colour affects the imbibition pattern of a seed. Burrows et al. (2009) indicated that variability in the germination rate of three Tasmanian provenances of Acacia melanoxylon was associated with a difference in their imbibition patterns. Similarly, Burrows et al. (2019) found a variation in their imbibition in seeds of 48 Acacia species, which might be the cause of the differences in the germination of the different provenances. Fredrick et al. (2017) also revealed significant differences in different provenances of Faidherbia albida germination percentage that was associated with the variation of the seeds in imbibition.
Similarly, despite the storage period of the seeds of the Gambo provenance (19 years) was lower than that of the Merti (30 years), Bishoftu (25 years), and Agaro (21 years) provenances, its mean germination percentage and mean germination index with all the different seed treatments was lower than that of the other studied provenances. This may show that provenance may have a more significant effect on the germination of the P. stellatum species instead of the storage period. The results also showed that the mean annual seed viability loss for the seeds collected from Dabena, Agaro, Bishoftu, Merti and Gambo provenances was on average 1.1%, 0.9%, 1.9%, 1.2%, and 1.2%, respectively. This could indicate that the mean storage time of the seeds has an impact on the mean germination capacity of the seeds of different provenances of P. stellatum. Research shows that seed storage of Fabaceae species for a longer time alters the germination capacity of different species (Jayasuriya et al. 2013; Colombe et al. 2019). The study showed that the viability of the seeds of the different provenances declined in its germination about 1.3% per year. The species seed can be stored for a longer time. Merritt et al. (2014) examined 172 Australian wild species and found many of the legume (Fabaceae) species to be stored for a longer time. Bhatt et al. (2021) found a variation in the germination percentage of three Astragalus sieberi D.C. (Fabaceae) populations.
Seed vigour refers to seed properties with the potential for rapid, uniform emergence and development of healthy and vigorous quality seedlings (Mangena 2021). Abdul-Baki and Anderson (1973) also indicated that the seed lot that shows the higher seed vigour index is considered to be more vigorous. The present study results revealed that the seeds collected from Dabena provenance have a higher vigour index compared to all other studied provenances, which shows that the seed lot is more vigorous. This might be associated with the lower storage period of the seeds of the provenance. De Vitis et al. (2020) indicated that increasing seed age can reduce germination vigour as the seed metabolic system begins to break down, resulting in seeds being slow or even unable to germinate, poor seedling development, and lower establishment for aged seeds that germinate. However, the Gambo provenance which has equal storage age with the Dabena provenance has resulted in the lowest seed vigour index even lower than those provenances where their seeds stored longer than the seeds of other studied provenances. Some provenances could result in a higher seed vigour index despite their long time of seed storage. This result could be associated with inter population variation of the provenance seeds.
The seed storage period is an important factor that affects seed viability and seedling growth of different plant species. For both the root and shoot growths, seedlings grown from Merti provenances which were stored for 30 years were relatively greater than the other studied provenance where their seeds were stored for less period. Despite the Gambo and Dabena provenance seeds were stored for an equal period (19 years) the results showed that seedlings grown from Dabena provenances achieved higher root length and shoot length relative to the Gambo provenance seedlings. The one-way ANOVA result also showed significant differences in the mean root lengths and shoot lengths of the seedlings grown from different provenance seeds that were stored for different periods. All these results could show that in the case of P. stellatum provenance has more effect on the seedling growth than storage time which might be associated with genetic factors that influence the seed quality. Different research also showed that the effect of the seed storage period on seedling growth may vary depending on the provenance of the seeds, as different environmental and genetic factors may influence the seed quality and dormancy (Molla and Desta 2022; Wawrzyniak et al. 2020).
Conclusion
Overall, the present study shows that (1) the seeds of P. stellatum have a physical dormancy problem that can be solved using the nicking as a presowing treatment and (2) It is observed that provenance had a significant influence on germination, seed vigour and seedling growth than seed storage period for the P. stellatum which shows the genetic variability of the species (3) the mean annual viability loss of the seeds of P. stellatum collected from different provenances was 1.3%. Finally, based on the present study it is recommended that the P. stellatum species has inter-population variation and for the purpose of ex-situ conservation their viability period as the storage time increases varies.
References
Abdul-Baki AA, Anderson JD (1973) Vigour determination in soybean seeds by multiple criteria. Crop Sci 13:630–633. https://doi.org/10.2135/cropsci1973.0011183X001300060013x
Albrecht J (1993) Tree seed handbook of Kenya. Kenya Forest Research Institute, Nairobi
Alemayehu G, Asfaw Z, Kelbessa E (2015) Ethnobotonical study of medicinal plants used by local communities of Minjar Shenkora District, north Showa zone of Amhara region Ethiopia. J Med Plants Stud 3:93–105
Andualem G, Umar S, Getinet F, Tekewe A, Alemayehu H, Kebede N (2014) Antimicrobial and phytochemical screening of methanol extracts of three medicinal plants in Ethiopia. Adv Biol Res 8:101–106. https://doi.org/10.5829/idosi.abr.2014.8.3.8383
Argel PJ, Paton CJ (1999) Overcoming legume hard seediness. In: Loch DS, Ferguson JE (eds) Forage seed production: tropical and subtropical species, vol 2. CAB International, Wallingford, pp 247–265
Bein E, Habte B, Jaber A, Birnie A, Tengnäs B (1996) Useful trees and shrubs in Eritrea: identification, propagation and management for agricultural and pastoral communities; technical Handbook No 12. Regional Soil Conservation Unit, Nairobi
Bench AR, Fenner M, Edwards P (1991) Changes in germinability, ABA content and ABA embryonic sensitivity in developing seeds of Sorghum bicolor (L.) Moench induced by water stress during grain filling. New Phytol 118:339–347. https://doi.org/10.1111/j.1469-8137.1991.tb00986.x
Bhatt A, Carón MM, Souza-Filho PRM, Gallacher DJ (2021) Maternal source affects seed germination of a rare Arabian desert species (Astragalus sieberi). Botany 99:293–301. https://doi.org/10.1139/cjb-2020-0144
Burrows GE, Virgona JM, Heady RD (2009) Effect of boiling water, seed coat structure and provenance on the germination of Acacia melanoxylon seeds. Aust J Bot 57:139–147. https://doi.org/10.1071/BT08194
Burrows G, Alden R, Robinson W (2019) Markedly different patterns of imbibition in seeds of 48 Acacia species. Seed Sci Res 29:270–282. https://doi.org/10.1017/S0960258519000242
Colombe D, Aggrey BN, Jane K, Achille EA (2019) Effect of storage temperature and dormancy breaking pre-treatments on germination and early seedling growth of Garcinia kola (Heckel): a threatened medicinal fruit tree in Benin. For Sci Technol 15:173–179. https://doi.org/10.1080/21580103.2019.1646673
De Vitis MD, Hay FR, Dickie JB, Trivedi C, Choi J, Fiegener R (2020) Seed storage: maintaining seed viability and vigor for restoration use. Restor Ecol 28:249–255. https://doi.org/10.1111/rec.13174
Delouche JC, Matthes RK, Dougherty GM, Boyd AH (1973) Storage of seed in sub-tropical and tropical regions. Seed Sci Technol 1:671–700
Ellis RH, Hong TD, Roberts EH (1991) Seed moisture content, storage, viability and vigour. Seed Sci Res 1:275–277. https://doi.org/10.1017/S0960258500001008
Fredrick C, Muthuri C, Ngamau K, Sinclair F (2017) Provenance and pre-treatment effect on seed germination of six provenances of Faidherbia albida (Delile) A. Chev. Agroforest Syst 91:1007–1017. https://doi.org/10.1007/s10457-016-9974-3
Fulbright TE, Flenniken KS (1987) Temperature and scarification effect on germination of prostrate bundleflower seeds. J Range Manag 40:170–173
Getahun A (1976) Some common medicinal and poisonous plants used in ethiopian folk medicine. Addis Ababa University, Ethiopia
Grubb PJ, Coomes DA (1997) Seed mass and nutrient content in nutrient-starved tropical rain forests in Venezuela. Seed Sci Res 7:269–280. https://doi.org/10.1017/S0960258500003627
Huang ZY, Gutterman Y (2000) Comparison of germination strategies of Artemisia ordosica with its two congeners from deserts of China and Israel. J Integr Plant Biol 42:71–80
Iroko OA, Rufal SO, Wahab WT (2021) Effect of seed size and different pretreatment methods on germination of Albizia zygia (DC.) J. F. Macbr J Appl Sci Environ Manag 25:815–818. https://doi.org/10.4314/jasem.v25i5.19
Janson PCM, Cardon D (2005) Plant resource of tropical Africa 3: dyes and tannins. Backhuys Publishers, Wageningen
Jayasuriya GKMG, Wijetunga ASTB, Baskin JM, Baskin CC (2013) Seed dormancy and storage behaviour in tropical Fabaceae: a study of 100 species from Sri Lanka. Seed Sci Res 23:257–269. https://doi.org/10.1017/S0960258513000214
Jemaneh K (2019) In vitro propagation of Pterolobium stellatum from shoot tip explants. M.Sc. thesis, Addis Abeba University, Addis Abeba
Kigen G, Maritim A, Some F, Kibosia J, Rono H, Chepkwony S, Kipkore W, Wanjoh B (2016) Ethnopharmacological survey medicinal plants used in Tindiret, Nandi country, Kenya. Afr J Tradit Complement Altern Med 13:156–168. https://doi.org/10.4314/ajtcam.v13i3.19
Mangena P (2021) Analysis of correlation between seed vigour, germination and multiple shoot induction in soybean (Glycine max L. Merr.). Heliyon 7:e07913. https://doi.org/10.1016/j.heliyon.2021.e07913
Mdlalose S, Workneh T, Sibanda S, Laing M (2021) Evaluation of a seed storage facility in relation to preserving seed moisture, vigour and germination. Afr J Food Agric Nutr Dev 21:18368–18390. https://doi.org/10.18697/ajfand.102.20140
Merritt DJ, Martyn AJ, Ainsley P, Young RE, Seed LU, Thorpe M, Hay FL, Commander LE, Shackelford N, Offord CA, Dixon KW, Probert RJ (2014) A continental-scale study of seed lifespan in experimental storage examining seed, plant, and environmental traits associated with longevity. Biodivers Conserv 23:1081. https://doi.org/10.1007/s10531-014-0641-6
Mola HG, Desta WD (2022) Effect of seed storage period on seed quality and seedling growth of basil (Ocimum basilicum L.)varieties. Int J Adv Res Biol Sci. 9:42–51. https://doi.org/10.22192/ijarbs.2022.09.10.005
Njoroge GN, Bussmann RW (2006) Traditional management of ear, nose and throat (ENT) diseases in central Kenya. J Ethnobiol Ethnomed 2:54. https://doi.org/10.1186/1746-4269-2-54
Orchard T (1977) Estimating the parameters of plant seedling emergence. Seed Sci Technol 5:61–69
Parera CA, Ruiz M (2003) Adesmia subterranea Clos germination physiology and presowing treatments. J Range Manag 56:273–276. https://doi.org/10.2307/4003818
Perez-Garcia F (2009) Germination characteristics and intrapopulation variation in carob (Ceratonia siliqua L.) seeds. Span J Agric Res 7:398–406. https://doi.org/10.5424/sjar/2009072-431
Pradhan BK, Badola HK (2008) Seed germination response of populations of Swertia chirayita [(Roxb. ex Fleming) H. Karst] following periodical storage. Seed Technol 30:63–69
Pradhan BK, Badola HK (2012) Effect of Storage conditions and storage periods on seed germination in eleven populations of Swertia chirayita: a critically endangered medicinal herb in Himalaya. Sci World J. https://doi.org/10.1100/2012/128105
Ragunthan M, Abay SM (2009) Ethno medicinal Survey of folk drugs used in Bahirdar zuria district, northwestern Ethiopia. Indian J Tradit Knowl 8:281–284
Rao NK, Dulloo ME, Engels JMM (2017) A review of factors that influence the production of quality seed for long-term conservation in genebanks. Genet Resour Crop Evol 64:1061–1074. https://doi.org/10.1007/s10722-016-0425-9
Rodrigues-Junior AG, Santos MTA, Hass J, Paschoal BSM, De-Paula OC (2020) What kind of seed dormancy occurs in the legume genus Cassia? Sci Rep 10:12194. https://doi.org/10.1038/s41598-020-69215-4
Schnadelbach A, Veiga-Barbosa L, Ruiz C, Pita JM, Pérez-García F (2016) Dormancy breaking and germination of Adenocarpus desertorum, Astragalus gines-lopezii and Hippocrepis grosii (Fabaceae) seeds three threatened endemic Spanish species. Seed Sci Technol 44:1–14. https://doi.org/10.15258/sst.2016.44.1.04
Schutz W, Rave G (1999) The effect of cold stratification and light on the seed germination of temperature sedges (Carex) from various habitats and implications for regenerative strategies. Plant Ecol 144:215–233. https://doi.org/10.1023/A:1009892004730
Scott S, Jones R, Williams W (1984) Review of data analysis methods for seed germination. Crop Sci 24:1192–2119. https://doi.org/10.2135/cropsci1984.0011183X002-400060043x
Seng M, Cheong EJ (2020) Comparative study of various pretreatment on seed germination of Dalbergia cochinchinensis. For Sci Technol 16:68–74. https://doi.org/10.1080/21580103.2020.1758801
Silva D, Stuepp CA, Wendling I, Helm C, Angelo AC (2019) Influence of seed storage conditions on quality of Torresea acreana seedlings. Cerne 25:60–67. https://doi.org/10.1590/01047760201925012611
Solberg SØ, Yndgaard F, Andreasen C, von Bothmer R, Loskutov IG, Asdal Å (2020a) Long-term storage and longevity of orthodox seeds: a systematic review. Front Plant Sci 11:1007. https://doi.org/10.3389/fpls.2020.01007
Solberg SØ, Brodal G, von Bothmer R, Meen E, Yndgaard F, Andreasen C, Asdal Å (2020b) Seed germination after 30 years storage in permafrost. Plants 9:579. https://doi.org/10.3390/plants9050579
Tadros MJ, Samarah NH, Alqudah AM (2011) Effect of different pre-sowing seed treatments on the germination of Leucaena leucocephala (Lam.) and Acacia farnesiana (L.). New for 42:397–407. https://doi.org/10.1007/s11056-011-9260-1
Teketay D (1996) The effect of different pre-sowing seed treatments, temperature and light on the germination of five Senna species from Ethiopia. New for 11:155–171. https://doi.org/10.1007/BF00033411
Teketay D (1998) Germination of Acacia origena, A. pilispina & Pterolobium stellatum in response to different pre-sowing seed treatments, temperature and light. J Arid Environ 38:551–560. https://doi.org/10.1006/jare.1997.0332
Teketay D, Tigabu M (1996) The effect of pre-sowing seed treatments, temperature and light on the germination of Tamarindus indica L., a multipurpose tree. J Trop for 12:73–79
Vijay K, Lokesh GY, Basave G, Patil SB, Ganiger BS, Rakesh CM (2015) Accelerated ageing test to study the relative storage potential of hybrid sunflower-RSFH-130 (Helianthus annuus). Afr J Agric Res 10:3502–3506. https://doi.org/10.5897/AJAR2015.9822
Ward DE, Spellenberg R (1988) Chromosome counts of angiosperms from new Mexico and adjacent areas. Phytologia 64:390–398. https://doi.org/10.5962/bhl.part.26120
Wawrzyniak MK, Michalak M, Chmielarz P (2020) Effect of different conditions of storage on seed viability and seedling growth of six European wild fruit woody plants. Ann for Sci 77:58. https://doi.org/10.1007/s13595-020-00963-z
Zegeye H, Teketay D, Kelbessa E (2005) Diversity, regeneration status and socio-economic importance of the vegetation in the islands of lake Ziway, south-central Ethiopia. Flora 201:483–498. https://doi.org/10.1016/j.flora.2005.10.006
Acknowledgements
Many persons supported us in the realization of this study, and the authors would like to thank the Forestry Research Center for providing us with the seed. Thanks to Fitsum Sirak and Mulata Fiyisa for their support in data collection in the greenhouse. Thanks also to Amelework Zenebe, Etetu Maruta, Yeshi Ketema, Kalkidan Daba and Atalay Dagnew for all their kindness in providing us with pieces of information on the seed's history.
Funding
Open access publishing supported by the National Technical Library in Prague. This research was funded by Mendel University in Brno with contributions from the Czech Development Agency; Grant Number CzDA-ET-2019-001-DO-41040.
Author information
Authors and Affiliations
Contributions
Conceptualization, SAM.; methodology, SAM, HH, KH.; software, SAM.; investigation, SAM; writing—original draft preparation, SAM.; writing—review and editing, HH, KH.; supervision, SAM.; project administration, HH.; funding acquisition, HH. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alem, S., Habrová, H. & Houšková, K. Germination responses with different treatments, seed vigour and seedling growth of Pterolobium stellatum (Forssk.) provenances stored from 19 to 30 years. Genet Resour Crop Evol (2023). https://doi.org/10.1007/s10722-023-01784-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10722-023-01784-5