Abstract
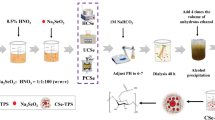
Selenium (Se) is an essential trace element for human beings and many other forms of life. Organic selenium from natural foods has greater bioavailability and is safer than inorganic selenium species. In this article, the structural properties and antioxidant activities of a Glycyrrhiza uralensis polysaccharide (GUP) after selenylation modification were investigated. The GUP was extracted by water decoction and ethanol precipitation and purified via protein elimination using the trichloroacetic acid method and column chromatography. The purified product was subsequently modified by the nitric acid-sodium selenite (HNO3-Na2SeO3) method. The selenized GUP (SeGUP) product was characterized by Fourier transform-infrared (FT-IR) spectroscopy, and its thermal stability, particle size, and antioxidant activities were investigated. FT-IR analysis indicated that the selenium in SeGUP existed mainly as O-Se-O. The thermal stability and particle size of SeGUP differed significantly from those of GUP. Moreover, compared to GUP, SeGUP exhibited greater antioxidant activities in vitro and in vivo. These results indicate that selenylation modification significantly enhances the antioxidant activity of SeGUP, increasing its potential for application as an antioxidant.

ᅟ





Similar content being viewed by others
References
Jassal, P.S., Kaur, G., Kaur, L.: Synergistic effect of curcuma longa and glycyrrhiza glabra extracts with copper ions on food spoilage bacteria. Int. J. Clin. Pharm. Net. 7, 371–375 (2015)
Yue, L., Wang, W., Wang, Y., Du, T., Shen, W., Tang, H., Wang, Y., Yin, H.: Bletilla Striata polysaccharide inhibits angiotensin II-induced ROS and inflammation via NOX4 and TLR2 pathways. Int. J. Biol. Macromol. 89, 376–388 (2016)
Wang, C., Xi, G.R., Shi, Y.R., Zhang, L.H.: Study on the anti-tumor effect in vivo of glycyrrhiza polysaccharide and its mechanism. Chin. J. Clin. Oncol. 8, 85–87 (2003) (in Chinese)
Zhang, C.H., Yu, Y., Liang, Y.Z., Chen, X.Q.: Purification, partial characterization and antioxidant activity of polysaccharides from Glycyrrhiza uralensis. Int. J. Biol. Macromol. 79, 681–686 (2015)
Kieliszek, M., Blazejak, S.: Selenium: significance, and outlook for supplementation. Nutrition. 29, 713–718 (2013)
Hurst, R., Hooper, L., Norat, T., Lau, R., Aune, D., Greenwood, D.C., Vieira, R., Collings, R., Harvey, L.J., Sterne, J.A., Beynon, R., Savović, J., Fairweather-Tait, S.J.: Selenium and prostate cancer: systematic review and meta-analysis. Am. J. Clin. Nutr. 96, 111–122 (2012)
Jayaprakash, V., Marshall, J.R.: Selenium and other antioxidants for chemoprevention of gastrointestinal cancers. Best Pract. Res. Clin. Gastroenterol. 25, 507–518 (2011)
Chiu, S.T., Hsieh, S.L., Yeh, S.P., Jian, S.J., Cheng, W., Liu, C.H.: The increase of immunity and disease resistance of the giant freshwater prawn, Macrobrachium rosenbergii by feeding with selenium enriched-diet. Fish Shellfish Immunol. 29, 623–629 (2010)
Indira Priyadarsini, K.G., Singh, B., Kunwar, A.: Current developments on synthesis, redox reactions and biochemical studies of selenium antioxidants. Curr. Opin. Chem. Biol. 7, 37–46 (2013)
Maseko, T., Howell, K., Dunshea, F.R., Ng, K.: Selenium-enriched Agaricus bisporus increases expression and activity of glutathione peroxidase-1 and expression of glutathione peroxidase-2 in rat colon. Food Chem. 146, 327–333 (2014)
Shi, L., Zhang, C., Yue, W., Shi, L., Zhu, X., Lei, F.: Short-term effect of dietary selenium-enriched yeast on semen parameters, antioxidant status and se concentration in goat seminal plasma. Anim. Feed Sci. Tech. 157, 104–108 (2010)
Qin, T., Ren, Z., Lin, D., Song, Y., Li, J., Ma, Y., Hou, X., Huang, Y.: Effects of selenizing Codonopsis pilosula polysaccharide on macrophage modulatory activities. J. Micobiol. Biotechnol. 26, 1358–1366 (2016)
Li, H., Wang, Y., Wang, C., Zhang, S., Li, S., Zhou, G., Wang, S., Zhang, J.: Extraction, selenylation modification and antitumor activity of the glucan from Castanea mollissima Blume. Glycoconj. J. 34, 207–217 (2017)
Qin, T., Chen, J., Wang, D., Hu, Y., Wang, M., Zhang, J., Nguyen, T.L., Liu, C., Liu, X.: Optimization of selenylation conditions for Chinese angelica polysaccharide based on immune-enhancing activity. Carbohydr. Polym. 92, 645–650 (2013)
Qiu, S., Chen, J., Chen, X., Fan, Q., Zhang, C., Wang, D., Li, X., Chen, X., Liu, C., Gao, Z.: Optimization of selenylation conditions for lycium barbarum polysaccharide based on antioxidant activity. Carbohydr. Polym. 103, 48–153 (2014)
Battin, E.E., Brumaghim, J.L.: Antioxidant activity of sulfur and selenium: a review of reactive oxygen species scavenging, glutathione peroxidase, and metal-binding antioxidant mechanisms. Cell Biochem. Biophys. 55, 1–23 (2009)
Zhao, B.T., Zhang, J., Yao, J., Song, S., Yin, Z.X., Gao, Q.Y.: Selenylation modification can enhance antioxidant activity of Potentilla anserina L. polysaccharide. Int. J. Biol. Macromol. 58, 320–328 (2013)
Hou, R., Chen, J., Yue, C., Li, X., Liu, J., Gao, Z., Liu, C., Lu, Y., Wang, D., Li, H., Hu, Y.: Modification of lily polysaccharide by selenylation and the immune-enhancing activity. Carbohydr. Polym. 142, 73–81 (2016)
Li, G., Wang, Z.: Sulfated esterifying technology of polysaccharide from Auricularia auricular and IR spectrum analysis. J. NE Forestry Uni. 12, 66–68 (2008) (in Chinese)
Yu, W., Yang, X.M., Liu, W.M., Liu, F.: Assay study on content of polysaccharides in ficus carica by phenol-sulfuric acid method. Food Sci. Technol. 10, 256–258 (2009)
Li, X., Hou, R., Yue, C., Liu, J., Gao, Z., Chen, J., Lu, Y., Wang, D., Liu, C., Hu, Y.: The selenylation modification of epimedium polysaccharide and isatis root polysaccharide and the immune-enhancing activity comparison of their modifiers. Biol. Trace Elem. Res. 171, 224–234 (2016)
Yen, G.C., Chen, H.Y.: Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agr. Food Chem. 43, 27–32 (1995)
Chen, H.Y., Yen, G.C.: Antioxidant activity and free radical-scavenging capacity of extracts from guava (Psidium guajava L.) leaves. Food Chem. 101, 686–694 (2007)
Halliwell, B., Gutteridge, J.M.C., Aruoma, O.I.: The deoxyribose method: a simple “test-tube” assay for determination of rate constants for reactions of hydroxyl radicals. Anal. Biochem. 165, 215–219 (1987)
Yokozawa, T., Dong, E., Natagawa, T., Kashiwagi, H., Nakagawa, H., Takeuchi, S., Chung, H.Y.: In vitro and in vivo studies on the radical-scavenging activity of tea. J. Agr. Food Chem. 46, 2143–2150 (1998)
Larrauri, J.A., Sanchez-Moreno, C., Saura-Calixto, F.: Effect of temperature on the free radical scavenging capacity of extracts from red and white grape pomace peels. J. Agr. Food Chem. 46, 2694–2697 (1998)
Tsai, S.Y., Huang, S.J., Mau, J.L.: Antioxidant properties of hot water extracts from Agrocybe cylindracea. Food Chem. 98, 670–677 (2006)
Jin, M., Lu, Z., Huang, M., Wang, Y., Wang, Y.: Effects of se-enriched polysaccharides produced by Enterobacter cloacae Z0206 on alloxan-induced diabetic mice. Int. J. Biol. Macromol. 50, 348–352 (2012)
Liu, J.Y., Feng, C.P., Li, X., Chang, M.C., Meng, J.L., Xu, L.J.: Immunomodulatory and antioxidative activity of Cordyceps militaris polysaccharides in mice. Int. J. Biol. Macromol. 86, 594–598 (2016)
Chen, T., Wang, J., Li, Y., Shen, J., Zhao, T., Zhang, H.: Sulfated modification and cytotoxicity of Portulaca oleracea L. polysaccharides. Glycoconj. J. 27, 635–642 (2010)
Li, C., Fu, X., Huang, Q., Luo, F.X., You, L.J.: Ultrasonic extraction and structural identification of polysaccharides from Prunella vulgaris and its antioxidant and antiproliferative activities. Eur. Food Res. Technol. 240, 49–60 (2015)
Zhang, J., Wang, F.X., Liu, Z.W., Zhang, S.T., Zhang, Y.Y., Liang, J.Y., Wang, Y.P.: Synthesis and characterization of seleno-Cynomorium songaricum Rupr. Polysaccharide. Nat. Prod. Res. 23, 1641–1651 (2009)
Liochev, S.I.: Reactive oxygen species and the free radical theory of aging. Free Radic. Biol. Med. 60, 1–4 (2013)
Ak, T., Gülçin, İ.: Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 174, 27–37 (2008)
Wang, J.L., Guo, H.Y., Zhang, J., Wang, X.F., Zhang, B.T., Yao, J., Wang, Y.P.: Sulfated modification, characterization and structure-antioxidant relationships of Artemisia sphaerocephala polysaccharides. Carbohydr. Polym. 81, 897–905 (2010)
Eklund, P.C., Langvik, O.K., Warna, J.P., Salmi, T.O., Willfor, S.M., Sjoholm, R.E.: Chemical studies on antioxidant mechanisms and free radical scavenging properties of lignans. Org. Bimol. Chem. 3, 3336–3347 (2005)
Ye, S., Liu, F., Wang, J.H., Wang, H., Zhang, M.P.: Antioxidant activities of an exopolysaccharide isolated and purified from marine Pseudomonas PF-6. Carbohydr. Polym. 87, 764–770 (2012)
Ramarathnam, N., Osawa, T., Ochi, H., Kawakishi, S.: The contribution of plant food antioxidants to human health. Trends Food Sci. Technol. 6, 75–82 (1995)
Wan, D., Zhou, X., Xie, C., Shu, X., Wu, X., Yin, Y.: Toxicological evaluation of ferrous N-carbamylglycinate chelate: acute, sub-acute toxicity and mutagenicity. Regul. Toxicol. Pharmacol. 73, 644–651 (2015)
Ye, M., Qiu, T., Peng, W., Chen, W.X., Ye, Y.W., Lin, Y.R.: Purification, characterization and hypoglycemic activity of extracellular polysaccharides from Lachnum calyculiforme. Carbohydr. Polym. 86, 285–290 (2011)
Pan, D., Mei, X.: Antioxidant activity of an exopolysaccharide purified from Lactococcus lactis subsp. lactis 12. Carbohydr. Polym. 80, 908–914 (2010)
Li, S.W., Luo, G.H., Qiao, F., Wang, F.J., Zhao, K.J., Wang, C.L.: Influence of Xanthomonas oryzae pv. oryzae PX099 inoculation on antioxidant enzyme activity and defense gene expression in CBB23. Chin. J. Appl. Environ. Biology. 19, 980–985 (2013) (in Chinese with English abstract)
Sun, Z.H., He, Z.X., Zhang, Q.L., Tan, Z.L., Han, X.F., Tang, S.X.: Effects of protein and/or energy restriction for six weeks on antioxidation capacity of plasma and gastrointestinal epithelial tissues of weaned kids. Livest. Sci. 149, 232–241 (2012)
Acknowledgements
We would like to thank all the staff at the Institute of Chinese Veterinary Medicine of Xinjiang Shihezi University for their assistances with these experiments. We would also like to thank Editage (www.editage.com) for English language editing and Publication Support.
The study was supported by the Talents of High Level Scientific Research Foundation [Grant no. RCZX201404].
Funding
The study was supported by the Talents of High Level Scientific Research Foundation [Grant no. RCZX201404].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All experiments related with animals were approved by the Animal Ethics Committee of Shihezi University (Approval No. AECSU2013–17).
Rights and permissions
About this article
Cite this article
Lian, KX., Zhu, XQ., Chen, J. et al. Selenylation modification: enhancement of the antioxidant activity of a Glycyrrhiza uralensis polysaccharide. Glycoconj J 35, 243–253 (2018). https://doi.org/10.1007/s10719-018-9817-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-018-9817-8