Abstract
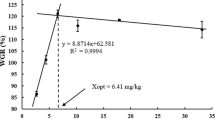
The aim of this study was to investigate the effect of dietary vitamin A on juvenile Chinese perch (Siniperca chuatsi). Chinese perch were fed with five experimental diets containing 0, 20, 40, 60, and 80 mg VA·kg−1 for 8 weeks. Results showed that dietary vitamin A significantly influenced the fish's growth, feed utilization, glucose and lipid metabolism, appetite, and antioxidant capacity. Vitamin A-supplemented groups had higher weight gain rate (WGR) and specific growth rate (SGR) compared to the control group. Feed conversion ratio (FCR) was also lower in the vitamin A-supplemented groups. Dietary vitamin A had no significant effect on the survival rate (SR). Compared to the control group, fish fed with vitamin A had increased feed intake (FI), and the expression of appetite-promoting genes (npy and agrp) was significantly higher in the 40 mg VA·kg−1 group. Vitamin A also enhanced the utilization of dietary protein by Chinese perch. The serum glucose content of the fish fed with 40 mg VA·kg−1 diet was significantly higher than that of the control group and 20 mg VA·kg−1 diet, indicating that the promoting effect of VA on gluconeogenesis was greater than that on glycolysis. Additionally, dietary vitamin A increased the expression of lipid metabolism-related genes (hl and fas) and antioxidant genes (nrf2 and gpx) in the fish. These results suggest that the optimal vitamin A requirement of juvenile Chinese perch bream was estimated to be 37.32 mg VA·kg−1 based on broken-line regression analysis of WGR. In conclusion, this study provides valuable insights into the potential benefits of dietary vitamin A on the growth, metabolism, and antioxidant capacity of Chinese perch.








Similar content being viewed by others
Data availability
All data generated and analyzed during this study are presented in this article.
Code availability
Not applicable.
References
Ahwach SM, Thomas M, Onstead-Haas L et al (2015) The glutathione mimic ebselen inhibits oxidative stress but not endoplasmic reticulum stress in endothelial cells. Life Sci 134:9–15. https://doi.org/10.1016/j.lfs.2015.05.004
Ballantyne JS (2001) Amino acid metabolism. Fish Physiology 20:77–107. https://doi.org/10.1016/s1546-5098(01)20004-1
Baranowski M (2008) Biological role of liver X receptors. J Physiol Pharmacol Off J Pol Physiol Soc 59 Suppl 7:31. https://doi.org/10.2170/physiolsci.RP013108
Battisti EK, Marasca S, Durigon EG et al (2017) Growth and oxidative parameters of Rhamdia quelen fed dietary levels of vitamin A. Aquaculture 474:11–17. https://doi.org/10.1016/j.aquaculture.2017.03.025
Berry DC, Noy N (2012) Signaling by vitamin A and retinol-binding protein in regulation of insulin responses and lipid homeostasis. Biochim Biophys Acta 1821:168–176. https://doi.org/10.1016/j.bbalip.2011.07.002
Blomhoff R, Blomhoff HK (2006) Overview of retinoid metabolism and function. J Neurobiol 66:606–630. https://doi.org/10.1002/neu.20242
Bruzelius K, Sundler R, Pagmantidis V, Akesson B (2010) Regulation of selenoprotein mRNA expression by hormones and retinoic acid in bovine mammary cells. J Trace Elem Med Biol 24:251–256. https://doi.org/10.1016/j.jtemb.2010.08.001
Campeche D, Catharino R, Godoy H, Cyrino JE (2009) Vitamin A in diets for Nile tilapia. Scientia Agricola - SCI AGRIC 66:. https://doi.org/10.1590/S0103-90162009000600005
Chen X-L, Kunsch C (2004) Induction of cytoprotective genes through Nrf2/antioxidant response element pathway: a new therapeutic approach for the treatment of inflammatory diseases. Curr Pharm Des 10:879–891. https://doi.org/10.2174/1381612043452901
Clagett-Dame M, DeLuca HF (2002) The role of vitamin A in mammalian reproduction and embryonic development. Annu Rev Nutr 22:347–381. https://doi.org/10.1146/annurev.nutr.22.010402.102745E
Costas B, Aragão C, Ruiz-Jarabo I et al (2011) Feed deprivation in Senegalese sole (Solea senegalensis Kaup, 1858) juveniles: effects on blood plasma metabolites and free amino acid levels. Fish Physiol Biochem 37:495–504. https://doi.org/10.1007/s10695-010-9451-2
D’Ambrosio DN, Clugston RD, Blaner WS (2011) Vitamin A metabolism: an update. Nutrients 3:63–103. https://doi.org/10.3390/nu3010063
Dawood MAO, Koshio S, Esteban MÁ (2018) Beneficial roles of feed additives as immunostimulants in aquaculture: a review. Rev Aquacult 10:950–974. https://doi.org/10.1111/raq.12209
Enes P, Panserat S, Kaushik S, Oliva-Teles A (2009) Nutritional regulation of hepatic glucose metabolism in fish. Fish Physiol Biochem 35:519–539. https://doi.org/10.1007/s10695-008-9259-5
Fernández I, Gisbert E (2011) The effect of vitamin A on flatfish development and skeletogenesis: a review. Aquaculture 315:34–48. https://doi.org/10.1016/j.aquaculture.2010.11.025
Gaspar C, Silva-Marrero JI, Fàbregas A et al (2018) Administration of chitosan-tripolyphosphate-DNA nanoparticles to knockdown glutamate dehydrogenase expression impairs transdeamination and gluconeogenesis in the liver. J Biotechnol 286:5–13. https://doi.org/10.1016/j.jbiotec.2018.09.002
He Y, Gong L, Fang Y et al (2013) The role of retinoic acid in hepatic lipid homeostasis defined by genomic binding and transcriptome profiling. BMC Genomics 14:575. https://doi.org/10.1186/1471-2164-14-575
Hemre G-I, Mommsen TP, Krogdahl Å (2002) Carbohydrates in fish nutrition: effects on growth, glucose metabolism and hepatic enzymes: Carbohydrates in fish nutrition. Aquac Nutr 8:175–194. https://doi.org/10.1046/j.1365-2095.2002.00200.x
Hernandez LHH, Teshima S, Koshio S et al (2007) Effects of vitamin A on growth, serum anti-bacterial activity and transaminase activities in the juvenile Japanese flounder, Paralichthys olivaceus. Aquaculture 262:444–450. https://doi.org/10.1016/j.aquaculture.2006.10.012
Huang Q, Kwaku A, Du T et al (2018) Dietary vitamin A requirement of Sillago sihama Forskál. Aquac Res 49:2587–2596. https://doi.org/10.1111/are.13725
Hudson RC, Daniel RM (1993) l-glutamate dehydrogenases: distribution, properties and mechanism. Comp Biochem Physiol B 106:767–792. https://doi.org/10.1016/0305-0491(93)90031-Y
Jeyakumar SM, Sheril A, Vajreswari A (2017) Vitamin A Improves Hyperglycemia and Glucose-Intolerance through Regulation of Intracellular Signaling Pathways and Glycogen Synthesis in WNIN/GR-Ob Obese Rat Model. Prev Nutr Food Sci 22:172–183. https://doi.org/10.3746/pnf.2017.22.3.172
Joseph SB, Laffitte BA, Patel PH et al (2002) Direct and indirect mechanisms for regulation of fatty acid synthase gene expression by liver X receptors*. J Biol Chem 277:11019–11025. https://doi.org/10.1074/jbc.M111041200
Klyuyeva AV, Belyaeva OV, Goggans KR et al (2021) Changes in retinoid metabolism and signaling associated with metabolic remodeling during fasting and in type I diabetes. J Biol Chem 296:100323. https://doi.org/10.1016/j.jbc.2021.100323
Lamb TD, Pugh EN (2004) Dark adaptation and the retinoid cycle of vision. Prog Retin Eye Res 23:307–380. https://doi.org/10.1016/j.preteyeres.2004.03.001
Li Y, Li R, Chen W, Chen G (2016) Vitamin A status and its metabolism contribute to the regulation of hepatic genes during the cycle of fasting and refeeding in rats. J Nutr Biochem 30:33–43. https://doi.org/10.1016/j.jnutbio.2015.11.012
Liang XF, Kiu JK, Huang BY (1998) The role of sense organs in the feeding behaviour of Chinese perch. J Fish Biol 52:1058–1067. https://doi.org/10.1111/j.1095-8649.1998.tb00603.x
Liang XF, Oku H, Ogata HY et al (2001) Weaning Chinese perch Siniperca chuatsi (Basilewsky) onto artificial diets based upon its specific sensory modality in feeding: Weaning Chinese perch onto artificial diets. Aquac Res 32:76–82. https://doi.org/10.1046/j.1355-557x.2001.00006.x
Lin Z, Han F, Lu J et al (2020) Influence of dietary phospholipid on growth performance, body composition, antioxidant capacity and lipid metabolism of Chinese mitten crab, Eriocheir sinensis. Aquaculture 516:734653. https://doi.org/10.1016/j.aquaculture.2019.734653
Liu B, Zhao Z, Brown PB et al (2016) Dietary vitamin A requirement of juvenile Wuchang bream (Megalobrama amblycephala) determined by growth and disease resistance. Aquaculture 450:23–30. https://doi.org/10.1016/j.aquaculture.2015.06.042
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mangelsdorf DJ, Evans RM (1995) The RXR heterodimers and orphan receptors. Cell 83:841–850. https://doi.org/10.1016/0092-8674(95)90200-7
Manolescu D-C, Sima A, Bhat PV (2010) All-trans retinoic acid lowers serum retinol-binding protein 4 concentrations and increases insulin sensitivity in diabetic mice. J Nutr 140:311–316. https://doi.org/10.3945/jn.109.115147
Mazurais D, Glynatsi N, Darias MJ et al (2009) Optimal levels of dietary vitamin A for reduced deformity incidence during development of European sea bass larvae (Dicentrarchus labrax) depend on malformation type. Aquaculture 294:262–270. https://doi.org/10.1016/j.aquaculture.2009.06.008
McClintick JN, Crabb DW, Tian H et al (2006) Global effects of vitamin A deficiency on gene expression in rat liver: evidence for hypoandrogenism. J Nutr Biochem 17:345–355. https://doi.org/10.1016/j.jnutbio.2005.08.006
Mohammedi K, Patente TA, Bellili-Muñoz N et al (2016) Glutathione peroxidase-1 gene (GPX1) variants, oxidative stress and risk of kidney complications in people with type 1 diabetes. Metabolism 65:12–19. https://doi.org/10.1016/j.metabol.2015.10.004
Oelze M, Kröller-Schön S, Steven S et al (2014) Glutathione peroxidase-1 deficiency potentiates dysregulatory modifications of endothelial nitric oxide synthase and vascular dysfunction in aging. Hypertension 63:390–396. https://doi.org/10.1161/HYPERTENSIONAHA.113.01602
Papp LV, Lu J, Holmgren A, Khanna KK (2007) From selenium to selenoproteins: synthesis, identity, and their role in human health. Antioxid Redox Signal 9:775–806. https://doi.org/10.1089/ars.2007.1528
Polakof S, Panserat S, Soengas JL, Moon TW (2012) Glucose metabolism in fish: a review. J Comp Physiol B 182:1015–1045. https://doi.org/10.1007/s00360-012-0658-7
Raja Gopal Reddy M, Jeyakumar SM, Vajreswari A (2022) Consumption of vitamin A-deficient diet elevates endoplasmic reticulum stress marker and suppresses high fructose-induced orexigenic gene expression in the brain of male Wistar rats. Nutr Neurosci 25:1872–1880. https://doi.org/10.1080/1028415X.2021.1911048
Reue K, Doolittle MH (1996) Naturally occurring mutations in mice affecting lipid transport and metabolism. J Lipid Res 37:1387–1405. https://doi.org/10.1016/S0022-2275(20)39126-4
Robbins KR (1986) A Method, SAS Program, and Example for Fitting the Broken-Line to Growth Data. https://trace.tennessee.edu/utk_agresreport/70/
Schwartz MW, Woods SC, Porte D et al (2000) Central nervous system control of food intake. Nature 404:661–671. https://doi.org/10.1038/35007534
Scott DK, Mitchell JA, Granner DK (1996) Identification and characterization of a second retinoic acid response element in the phosphoenolpyruvate carboxykinase gene promoter (∗). J Biol Chem 271:6260–6264. https://doi.org/10.1074/jbc.271.11.6260
Stoney PN, McCaffery P (2016) A vitamin on the mind: new discoveries on control of the brain by vitamin A. World Rev Nutr Diet 115:98–108. https://doi.org/10.1159/000442076
Tan KP, Kosuge K, Yang M, Ito S (2008) NRF2 as a determinant of cellular resistance in retinoic acid cytotoxicity. Free Radical Biol Med 45:1663–1673. https://doi.org/10.1016/j.freeradbiomed.2008.09.010
Williams S (1984) Official Methods of Analysis of the Association of Official Analytical Chemists. Assoc Off Anal Chem. https://lib.ugent.be/en/catalog/rug01:000071630
Wu P, Zhang L, Jiang W et al (2022) Dietary vitamin A improved the flesh quality of grass carp (Ctenopharyngodon idella) in relation to the enhanced antioxidant capacity through Nrf2/Keap 1a signaling pathway. Antioxidants (basel) 11:148. https://doi.org/10.3390/antiox11010148
Yang Q, Ding M, Tan B et al (2017) Effects of dietary vitamin A on growth, feed utilization, lipid metabolism enzyme activities, and fatty acid synthase and hepatic lipase mRNA expression levels in the liver of juvenile orange spotted grouper, Epinephelus coioides. Aquaculture 479:501–507. https://doi.org/10.1016/j.aquaculture.2017.06.024
Yang Q, Zhou X, Jiang J, Liu Y (2008) Effect of dietary vitamin A deficiency on growth performance, feed utilization and immune responses of juvenile Jian carp (Cyprinus carpio var. Jian). Aquaculture Res 39:902–906. https://doi.org/10.1111/j.1365-2109.2008.01945.x
Zhang Y, Liang X-F, He S et al (2021) Metabolic responses of Chinese perch (Siniperca chuatsi) to different levels of dietary carbohydrate. Fish Physiol Biochem 47:1449–1465. https://doi.org/10.1007/s10695-021-00965-2
Zile MH, Cullum ME (1983) The function of vitamin A: current concepts. Proc Soc Exp Biol Med 172:139–152. https://doi.org/10.3181/00379727-172-41537
Acknowledgements
The research would like to thank the Innovation Base for Chinese Perch Breeding, Key Lab of Freshwater Animal Breeding, Ministry of Agriculture, Wuhan, China, for providing the breeding site. Thanks to the others who contributed to the experiment.
Funding
The Key Research & Development Program of Hubei Province(2022BBA0051); Key Research and Development Project of Hubei Province (2020BBA035).
Author information
Authors and Affiliations
Contributions
Xu-Fang Liang: experimental design; Junliang Chen: carrying out experiments, collecting samples and analyzing data; Ming Zeng: writing and revising manuscripts. Di Peng, Ruipeng Xie and Dongliang Wu: participated in sample collection.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
The animal study was reviewed and approved by the Ethics committee of the Institute of Laboratory Animal Centre, Huazhong Agriculture University.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflicts of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Zeng, M., Liang, XF. et al. Dietary supplementation of VA enhances growth, feed utilization, glucose and lipid metabolism, appetite, and antioxidant capacity of Chinese perch (Siniperca chuatsi). Fish Physiol Biochem 50, 225–237 (2024). https://doi.org/10.1007/s10695-023-01221-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01221-5