Abstract
Intraspecific variation in body size, both among populations and between sexes, is an important factor influencing life-history strategies. This variation might be the response to different environmental conditions, as well as natural and sexual selection, and can result in differences in behavior and reproductive strategies among populations. Here, we use the dyeing poison frog (Dendrobates tinctorius) as a model to investigate how interpopulation variation in body size and sexual size dimorphism affects reproductive strategies. As body size increased, sexual size dimorphism also increased, i.e., females were larger than males, and more so in populations with overall larger frogs. This indicates that there is a stronger selection for body size in females than in males, likely as a response to divergent reproductive investment between the sexes. Females from larger-bodied populations produced larger clutches, but the overall number of froglets produced per clutch did not differ among populations. We discuss potential causes and mechanisms that might be responsible for the observed divergence in body size, sexual size dimorphism, and reproductive strategies among populations that likely represent local adaptations. Our findings demonstrate the importance of cross-population studies, cautioning against drawing general conclusions about a species’ ecology without accounting for intraspecific variation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phenotypic and genotypic variation within and among populations of single species (intraspecific variation) are essential for adaptation to environmental change and evolution by natural selection (Des Roches et al. 2018). The study of spatio-temporal intraspecific variation can, therefore, provide insight into local adaptation and population divergence. Yet, intraspecific variation has historically been overlooked in most conservation efforts, typically directed only at protecting species diversity (Des Roches et al. 2021; Hughes et al. 1997).
Body size is an important factor influencing life-history strategies (Miles and Dunham 1992) that commonly display intraspecific variation among populations (Ashton 2001; Chown and Gaston 2010; Gavini et al. 2020) and between sexes of the same population (i.e., sexual size dimorphism, hereafter SSD) (Zhang and Lu 2013). While variation between populations is commonly explained by local adaptation or phenotypic plasticity to environmental differences, such as latitude and altitude (Liang et al. 2023; Morrison and Hero 2003), SSD is usually explained by a complex combination of selective pressures acting differently on the sexes. For example, in male-biased SSD the selection pressure is typically associated with larger males having an advantage in intrasexual competition for access to females (Blanckenhorn 2005; Shine 2003), and should be stronger than the selection pressure on female fertility. Female-biased SSD, in contrast, is usually explained by the fecundity advantage hypothesis, whereby selection favors larger females that produce larger litters (Liang et al. 2023; Nali et al. 2014). Further, numerous animal groups follow a macroecological pattern known as Rensch’s rule, according to which SSD increases with body size (hyperallometry) when males are the larger sex, and decreases with body size (hypoallometry) when females are larger (Rensch 1950). Although the mechanisms leading to this pattern are still not fully understood, intra- and intersexual selection are often attributed as the main driving forces (Székely et al. 2004). However, Rensch’s rule does not always hold, and numerous studies have found the inverse, i.e., SSD increasing with female size in species where females are the larger sex (Dugo-Cota et al. 2022; Liao et al. 2015; Teder and Tammaru 2005).
Intraspecific variation in body size can translate into differences in behavior (Gavini et al. 2020), performance traits (Brecko et al. 2008), microhabitat use (Isaac 2005), or mating systems (Baur et al. 2020) between populations and sexes. Thus, understanding intraspecific variation can give us important information about a species’ ability for phenotypic and behavioral plasticity, and how they might cope with environmental change. Furthermore, increasing differences in body size can generate assortative mating and/or mating incompatibilities among populations, which can lead to reproductive isolation and speciation (Montiglio et al. 2016).
The dyeing poison frog (Dendrobates tinctorius) is an excellent model organism for studying intraspecific variation in body size and its potential impact on fecundity and reproductive strategies. D. tinctorius populations are partially isolated and characterized by great phenotypic variation among them, especially in terms of aposematic color and pattern (Noonan and Gaucher 2006; Wollenberg et al. 2008). In fact, phenotypic differences are so extreme that they have historically been described as distinct species (Wollenberg et al. 2006). More recent studies have also found population differences in toxicity (Lawrence et al. 2019, 2023), while many other traits, such as clutch size, remain unexplored.
Although variation in body size and SSD (females are bigger) in a D. tinctorius population has been described in the past (Rojas and Endler 2013), very little attention has been paid to their ecological and evolutionary implications. Moreover, such variation has not been studied across different populations. Importantly, a better understanding of intraspecific variation in body size and SSD could help us identify the different selective pressures acting on the sexes and populations, as well as give us a first insight into possible ongoing speciation events.
Here, we quantified intraspecific variation in body size and SSD across four populations of D. tinctorius in the wild and tested the consistency of Rensch’s rule. Moreover, we tested how these differences potentially translate into divergences in their reproductive strategies by conducting breeding experiments with frogs from five populations. In line with the fecundity advantage hypothesis, we would expect populations with larger female body size to produce larger clutches (Han and Fu 2013; Nali et al. 2014). However, the terrestrial egg-laying nature of the species and the need for paternal care (i.e., males transport hatched tadpoles to water bodies, in most cases one by one (Rojas 2014; Rojas and Pašukonis 2019) could act as a constraint on the evolution of clutch size (Han and Fu 2013; Lange et al. 2021; Gould et al. 2022). Although less common, female body size could also be positively correlated with egg size (Wells 2019), which could represent a direct reproductive advantage if we assume higher survival rates of larger eggs in the face of, for example, desiccation. However, a negative correlation between clutch and egg size is often found due to physiological trade-offs (Liao et al. 2014; Wells 2019; Gould et al. 2022). We predicted that under standardized conditions, if larger females produce larger clutches, those will have relatively smaller eggs with faster development, lower survival, and smaller offspring (Marshall and Bolton 2007; Gould et al. 2022). Contrary, if larger females produce smaller clutches with larger eggs, we expect clutches to have slower development, higher survival, and larger offspring (Gould et al. 2022) (Fig. 1).
Schematic representation of our hypothesis related to reproductive investment depending on maternal body size in the dyeing poison frog (Dendrobates tinctorius). A large female could invest in either a large clutch of rather small eggs or, instead, in a small clutch of large eggs. Each scenario leads to different outcomes for offspring development and fitness
Materials and methods
Study species, study areas, and data collection
D. tinctorius is endemic to the Eastern Guiana Shield and, although it can be found at elevations between 0 and 600 m, they are often restricted to elevated ridges or plateaus, creating a patchy distribution of populations (Noonan and Gaucher 2006). They are diurnal and show complex reproductive behaviors, including paternal care (Rojas and Pašukonis 2019). Eggs are laid by the female on ground leaf litter or under/inside hollow fallen logs, and then taken care of by the male. Clutch size typically consists of four to five eggs in one of the populations (Rojas 2014), but no information is available for others. Like in other poison frogs, males transport newly hatched tadpoles to small pools formed in vegetation structures (i.e., phytotelmata), where they remain unattended until metamorphosis (Rojas and Pašukonis 2019).
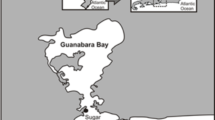
Between January and March 2023, we sampled 230 frogs in four populations located in French Guiana: (1) Kaw, (2) Mataroni, (3) Mont Fortuné and Petit-Matoury, and (4) Nouragues Nature Reserve (Fig. 2; Table 1). Each population harbored individuals with a distinct color morph (Fig. 2), with frogs in Nouragues being more variable in coloration compared to the other populations (Rojas and Endler 2013; JP Lawrence & B Rojas et al., unpublished data). Although the study sites in Mont Fortuné and Petit-Matoury are fragmented by a large road, we categorized them as one population because the two areas are within close spatial proximity (ca. 1.5 km) and there were no statistically significant differences in body mass and no discernible differences (i.e., to a human observer) in coloration. Moreover, sample sizes from these sites were smaller (35 and 34 frogs) than in the other sites, making it necessary to merge them for analysis.
The location of a our study areas and of the Amazonian Experimental Ecology Platform (PLEEA) within French Guiana, and b within South America (red square). Data were either collected for breeding experiments (blue dots), to obtain field observations (orange dot), or for both purposes (red dots). Moreover, the map schematically shows the morphological differences among individuals from the different dyeing poison frog (Dendrobates tinctorius) populations
In each study area, we walked transects that covered an area between 10 and 15 km in length, and systematically captured all frogs that we encountered. We hand-captured frogs, changing gloves for each new individual. Frogs were photographed on millimeter paper for snout-vent length (SVL) measurement and individual identification (to avoid double-counting individuals that were captured multiple times), weighed, and sexed based on the shape and size of their fingertips (Rojas and Endler 2013). All frogs were released right after at the capture site.
Breeding experiments
For breeding experiments, we collected 54 frogs between January 2020 and August 2022 from five populations (Fig. 2): (1) Mt Bruyére (2 females and 2 males forming 2 unique pairs), (2) Kaw (9 females and 6 males forming 18 unique pairs; 5 females and 5 males were used for multiple pairings), (3) Mataroni (7 females and 7 males forming 11 unique pairs; 2 females and 3 males were used for multiple pairings), (4) Ouanary (7 females and 8 males forming 11 unique pairs; 4 females and 3 males were used for multiple pairings), and (5) Petit-Matoury (3 females and 3 males forming 3 unique pairs). We followed the same capture and handling procedures as described above. Wild-caught frogs were placed in captivity at the Amazonian Experimental Ecology Platform (PLEEA), located in Montabo, French Guiana (Fig. 2). They were kept in an open greenhouse under natural climatic conditions (mean daily temperature was 26 °C across all months when reproduction trials were conducted), with a shade sail installed to mimic the forest cover, and automatic sprinklers to keep the greenhouse in humid conditions by watering 3 times a day. The adult frogs were fed three times a week with Drosophila flies (dusted with calcium and vitamins). Frogs were placed in Exoterra© mesh terrariums (60 cm × 45 cm × 90 cm) in pairs from the same population.
We checked terraria three times per week for egg deposition (this date was defined as egg laying day), noting the number of eggs laid. The eggs were then transferred to petri dishes (60 mm) where they were left until they hatched. We acknowledge that this prevented paternal care, potentially affecting tadpole development and survival, but deemed this necessary to disentangle between genetic factors and parental care. We noted the date and number of tadpoles hatched per clutch and took a picture of the tadpoles on the day of hatching to estimate their size, measured from the snout to the tip of the tail. We then placed each tadpole individually in a cup filled with 250 ml of rainwater with some dead leaves. They were fed three times a week ad libitum with a mixture of spirulina and nettle powder, until froglets metamorphosed. We then transferred the froglets to a separate terrarium. We noted the date and the number of froglets that emerged. For pairs that laid multiple clutches over time, we recorded the clutch order.
Statistical analysis
Field observations
We used the ‘scaled mass index’ of body condition \(\hat{\rm M}_{\rm i}\) as a measure of body condition, following Peig and Green (2009). It can be computed as follows
where Mi is the body mass and Li the SVL of individual i; bSMA is the scaling exponent estimated by a standardized major axis regression of log transformed M on L; L0 is the arithmetic mean SVL for the study population; and \(\hat{\rm M}_{\rm i}\) is the predicted body mass for individual i when the linear body measure is standardized to L0 (Peig and Green 2009).
To investigate sex and population differences in SVL, body mass, and body condition (response variable in separate analyses), we used linear models with a Gaussian distribution. We included population, sex, and their two-way interaction as predictor variables (Table 2). We conducted post-hoc pairwise comparisons to investigate differences between females and males in each population (Table S1) using the R package ‘emmeans’ using Tukey P-value correction for multiple testing (Lenth et al. 2019). Moreover, we estimated sex ratios as the number of males per female and used one-proportion z-tests separately for each population to investigate if the proportion of males (out of all individuals) differed from 0.5. Finally, we ran a generalized linear model with a binomial distribution to investigate if the proportion of males (out of all individuals; response variable) differed among population (predictor).
Breeding experiments
We initially used generalized linear models with a Poisson distribution and a log link to investigate (1) the number of clutches produced per pair (n = 45 pairs; all pairs were housed for approximately the same amount of time), including population and maternal SVL as predictor variables (Table S2). We then used generalized linear mixed models (GLMM) of the R package ‘lme4’ (Bates et al. 2015) to evaluate population differences in (2) egg laying frequency, measured as the number of days between clutch deposition (only possible for pairs that produced more than one clutch, n = 242 clutches), (3) number of eggs per clutch (clutch size, n = 290), (4) number of tadpoles hatched per clutch (n = 299 clutches), (5) time to hatching (n = 217 clutches), defined as the number of days from egg deposition till tadpole hatching, (6) tadpole size (total length from snout to the tip of the tail) at hatching (n = 738 tadpoles), which was used as proxy for offspring size, (7) time to metamorphosis (n = 485 froglets), defined as the number of days from egg deposition to metamorphosis of froglets, and (8) survival (n = 273 clutches), defined as the proportion of eggs surviving from laying to metamorphosis of froglets (Table S2). We included population, maternal SVL (to investigate among-female variation), their two-way interaction, and clutch order (for analysis 2–8) as predictor variables and maternal ID as a random intercept in all analyses (Table S2). For the analysis of tadpole size, time to metamorphosis, and survival, we additionally included clutch size as predictor, and for the analysis of time to hatching, clutch size and average tadpole size at hatching as additional predictors (Table S2). For count data (clutch size, tadpole number, time to hatching) we used a Poisson distribution and a log link. To analyze survival, we used a binomial distribution and a logit link, and to analyze tadpole size, we used a Gaussian distribution and an identity link.
Model selection
For all analyses, we used a set of candidate models including all possible combinations of the fixed effects, including the above-mentioned two-way interaction. There was no collinearity (Pearson’s r < 0.6 and variance inflation factors < 3) between independent variables within the same model (Zuur et al. 2010). We scaled and centered all numeric fixed effects to avoid convergence issues and to be able to compare the relative effect sizes. Model selection was based on Akaike’s information criterion corrected for small sample size (AICc) (Burnham et al. 2011), using the r package ‘MuMIn’ (Barton 2020), selecting the model with the lowest AICc value. Parameters that included zero within their 95% CI were considered uninformative (Arnold 2010). For all analyses where population was retained in the most parsimonious model, we conducted post-hoc pairwise comparisons (as described above) to detect differences among populations. We evaluated model fit by calculating the marginal R2, i.e., the variation explained by the fixed effects, and the conditional R2, i.e., the variation explained by fixed and random effects (Nakagawa and Schielzeth 2013). Model assumptions were verified by plotting residuals versus fitted values (Zuur and Ieno 2016). All statistical analyses were conducted in R version 4.2.3 (R Core Team 2017).
Results
Field observations
SVL and body mass differed between sexes and among the four populations (Fig. 3; Tables 1 and 2). Frogs were largest in Mataroni, intermediate in Nouragues, and smallest in Kaw and Mt Fortuné/Petit-Matoury, with no statistically significant difference in SVL and body mass between the latter two populations (Fig. 3a; Table 2, Fig. S1). On average, frogs from Mataroni were 1.2-fold larger (in terms of SVL) than frogs from Nouragues, and 1.4-fold larger compared to the other populations. Similarly, frogs in Mataroni were 1.7-fold heavier (in terms of body mass) compared to Nouragues, and 2.6-fold heavier compared to the other populations. Sexual size dimorphism was greater in frogs from populations with larger individuals, i.e., in Mataroni and Nouragues, compared to frogs from populations with smaller individuals (Kaw and Mt Fortuné/Petit Matoury; Fig. 3a,b; Table 2, Table S1). Differences in SVL and body mass between females and males were significant in all populations, except Mt Fortuné/Petit Matoury (Table 2, Table S1). There were no differences regarding body condition between males and females or between populations (Fig. 3c; Table 2). Observed sex ratios were generally female-biased (Table 1), with the proportion of males (out of all frogs) differing from 0.5 in Mataroni (X-squared = 8.16, P-value = 0.004) and Mt Fortuné/Petit Matoury (X-squared = 4.70, P-value = 0.03), but not in Nouragues (X-squared = 1.27, P-value = 0.26) and Kaw (X-squared = 0.52, P-value = 0.47). When tested for all populations combined, the proportion of males differed significantly from 0.5 (X-squared = 13.15, P-value < 0.001). However, there were no statistically significant differences regarding the proportion of males among populations (Table 2).
Breeding experiments
We obtained 316 clutches from 45 breeding pairs, consisting of 28 females and 26 males (individuals were paired with multiple mates). Breeding pairs produced 1 to 43 clutches (mean ± SD: 7.1 ± 8.9 clutches). The number of clutches per pair was best explained by female SVL and population (Table S2), with pairs containing larger females (within populations) producing more clutches and pairs from Kaw producing more clutches compared to all other populations (Fig. 4a, Tables S3, S4). Pairs laid a clutch every (mean ± SD) 18 ± 45 days (Fig. 4b; Table 3). The number of days between clutches was best explained by clutch order and population, with clutch frequency declining over time (within pairs) and pairs in Kaw laying clutches at higher frequency compared to frogs from Ouanary (Fig. 4b, Table S2). There was no statistically significant difference between the other populations, although frogs from Ouanary tended (p < 0.10) to have a lower clutch laying frequency compared to frogs from Bruyere and Petit-Matoury (Table S4).
Showing the predicted population estimate (large symbols) of the a number of clutches produced per pair, b days between clutch deposition per pair, c number of (i) eggs laid, (ii) tadpoles hatched, and (iii) frogs metamorphosed per clutch, and d time to tadpole hatching, based on breeding experiments of 45 pairs of dyeing poison frog (Dendrobates tinctorius) from 5 different populations. Moreover, the predicted effect (line) of clutch size on e time to metamorphosis and f clutch survival (proportion of eggs surviving from laying to metamorphosis). The 95% confidence intervals are shown as bars (a–d) or shading (e, f), and small dots (all plots) show the raw data. Note that plot B is on logarithmic scale
Clutch size ranged from 1 to 12 eggs (mean ± SD: 4.3 ± 1.8 eggs) and was best explained by clutch order and population, with frogs from Mataroni (largest average SVL and body mass) producing larger clutches compared to all other populations (Fig. 4c, Tables S2, S4). There was no statistically significant difference between the other populations (Table S5). Clutch order was uninformative (Table S3). The number of tadpoles hatched per clutch was best explained by clutch order (Table S2), but this effect was uninformative (Table S3). Population and maternal SVL were not included in the best model (Table S2). Time to hatching ranged from 4 to 28 days (mean ± SD: 14 ± 4 days) and was best explained by population and average tadpole size at hatching (Table S2). Time to hatching was positively correlated with tadpole size (estimate ± SE: 0.105 ± 0.020, 95% CI: 0.067, 0.144), and eggs from Petit-Matoury hatched sooner compared to all other populations (Fig. 4d, Table S5). There were no statistically significant differences regarding time to hatching for pairwise comparisons between the other populations (Table S5). Tadpole size at hatching ranged from 8.1 to 21.0 mm (mean ± SD: 15.2 ± 1.4 mm) and was best explained by the model including population (Table S2). However, the effect was uninformative with no statistically significant differences regarding tadpole size for pairwise comparisons between populations and the intercept only model was within delta AIC < 2 (Tables S3, S4). Time to metamorphosis ranged from 53 to199 days (mean ± SD: 82 ± 15 days) and was positively correlated with clutch size (Fig. 4e) and clutch order (Table S3). When additionally analyzing average time to metamorphosis per clutch and including average tadpole size (we did not have these data on individual tadpole level), tadpole size was uninformative in explaining variation in time to metamorphosis (Table S3). Finally, survival (proportion of eggs per clutch surviving to metamorphosis) ranged from 0 to 1 (mean ± SD: 0.44 ± 0.37) and was negatively correlated with clutch size (Fig. 4f) and clutch order (Table S3). However, clutch size only explained 2% of the variation in survival and clutch order was uninformative, whereas maternal ID explained 19% (Table S2). Generally, maternal ID was important in explaining variation in clutch laying frequency (R2: 0.94), tadpole size (R2: 0.33), and time to metamorphosis (R2: 0.59; Table S2).
Discussion
Here, we investigated intraspecific variation in body size and SSD in four populations of a polytypic frog and tested whether these differences translate into distinctive life-history traits. Broadly, we found that as body size and mass increase in a population, sexual size dimorphism also increases, opposite to Rensch’s rule. Females from larger-bodied populations produced larger clutches, but the overall number of froglets produced per clutch did not differ among populations. Moreover, we found population differences regarding the number of clutches produced, days between clutch laying, and time to tadpole hatching that were unrelated to average body size among populations. We discuss potential causes and mechanisms responsible for the observed divergence in body size, SSD, and reproductive strategies among populations.
Variation in body size, sexual size dimorphism, and sex ratios among populations
Body size varied strongly among populations, with frogs from Mataroni being 40% larger compared to individuals from Kaw and Mt Fortuné/Petit-Matoury. We can only speculate regarding the reasons for this large variation, which might be related to genetic drift, differences in climate, e.g., precipitation and seasonality, and resource availability (Cvetković et al. 2009; Henry et al. 2023; Olalla-Tárraga and Rodríguez 2007). Genetic drift as a random source of allelic variation could lead to non-adaptive change in body sizes, due to increased differentiation between populations with reduced gene flow (Slatkin 1987). It is unlikely that population differences in body size are related to thermal conditions in D. tinctorius, as there are similar and constant temperatures across the region. It is likely that vertebrate communities varied between the studied populations, resulting from different levels of habitat disturbance (Cantera et al. 2022). This might have indirectly affected resource availability, prey and predator communities, and interspecific competition, creating selective pressures on morphological traits, including body size. However, without underlying data this remains purely speculative. We found no measurable differences in body condition among populations. It is hard to draw conclusions from this finding, as we did not measure resource availability, which moreover could be invested in both, reproduction or growth (Lardner and Loman 2003). Future experimental studies providing varying amounts of food to frogs from different populations could shed light on how females allocate resources differently into reproduction or growth, and if differences in growth and body size have a genetic basis.
Most anuran species exhibit female-biased SSD (Shine 1979), a pattern also found in all populations of our study species except Mt Fortuné/Petit Matoury. However, the degree of allometry between male and female body size differed between populations, being greater in the populations with larger females (Mataroni and Nouragues). These findings are consistent with the inverse of Rensch’s rule, which has been reported in multiple studies with female-biased SSD (Dugo-Cota et al. 2022; Liao et al. 2015; Teder and Tammaru 2005). Therefore, our results suggest differences in selective pressures acting on female and male body size both within and between populations. Advantages associated with increased fecundity are commonly recognized as a leading selecting force in anurans shaping female-biased SSD (Nali et al. 2014). However, a significant correlation between female body size and fecundity-related traits (i.e., clutch size) was only found when comparing populations, but not within populations (see below). Apart from differences in body size and SSD, individuals from the different populations also differ in coloration and toxicity (Lawrence et al. 2019, 2023), which might be the result of varying predation pressure among populations.
We found female-biased adult sex ratios in two of the four investigated populations, raising the question of whether skewed sex ratios have a genetic basis or are a result of environmental factors. Observed differences in sex ratios might be related to data collection during slightly different times of the year (though all data was collected between January and March), potentially affecting the activity of males and females. This is unlikely as unpublished data confirm the low proportion of males in Mataroni (MC; personal observation).To test a potential genetic basis, e.g. if there are differences in sex ratios at birth, future breeding experiments could determine the sex of offspring, and establish whether there are differences in development time and survival between male and female tadpoles; similar to studies conducted in wood frogs (Lithobates sylvaticus) (Warne and Crespi 2015). Moreover, by measuring estrogen levels in females from different populations, we could investigate the possibility that females can affect the sex ratios of their clutches via endogenous sex steroids (Nakamura 2010). An environmental explanation for female-biased sex ratios is that males might generally face a higher predation pressure compared to females, because they move larger distances when transporting tadpoles (Pašukonis et al. 2019, 2022), potentially making them more conspicuous to predators (Rojas and Endler 2013). In line with having the largest SSD, frogs from Mataroni also had the most female-biased sex ratio, which might be related to their comparatively higher fecundity. It is conceivable that males in this population experienced higher predation rates (compared to males from other populations) if they spent more time transporting tadpoles, either due to larger clutches or due to taking care of the clutches of multiple females (paternal care for multiple clutches was observed by BR and shown for the closely related D. auratus (Summers 1989). Alternatively, the spatial distribution and abundance of phytotelmata might have differed between populations, forcing males to move greater distances, thereby affecting their mortality risk. To test this hypothesis, it would be of interest to describe the distribution, abundance, and type of phytotelmata (Fouilloux et al. 2021) among the different populations.
Variation in reproductive investment
Females from Mataroni (the population with the largest body size) laid larger clutches compared to all other populations but did not produce larger tadpoles. These two traits, offspring number and size, determine the maternal reproductive investment and are often negatively correlated (Gould et al. 2022), though this relationship might be more complicated (Brown and Shine 2009). Given that population differences best explained the variation in clutch size (and we did not find a positive correlation between clutch size and female body size), this pattern appears to be related to differences among populations, although the mechanisms for this remain unclear.
Moreover, we found a negative correlation between clutch size and offspring survival, resulting in the same offspring recruitment among the different populations, regardless of initial clutch size. That is, a possible fecundity advantage in Mataroni frogs did not translate into a higher reproductive success per clutch in the breeding experiments. This suggests that a reduction in the viability of tadpoles seems to be a limiting factor in the evolution of clutch size. The reasons for increased mortality with larger clutch size remain unclear, but might be related to a higher risk of fungal infection (Rojas and Pašukonis 2019). In poison frogs, egg care has been proven to reduce dehydration, fungal infections in embryos, and predation events (Rocha et al. 2021). Thus, paternal care might potentially ameliorate the observed increasing mortality with clutch size in the wild.
We did not find a positive correlation between female body size and clutch/offspring size within populations, similar to other studies (Dziminski and Roberts 2006), but after accounting for population differences, larger females produced more clutches. Larger females might increase their reproductive success by laying more clutches rather than by adjusting clutch or offspring size, though this needs more investigation as clutch frequency showed no clear pattern related to maternal body size. Finally, we observed a positive correlation between tadpole size and time to hatching, but not to metamorphosis. Other studies also found larger eggs (which produced larger tadpoles) to take longer to hatch or reach early larval stages, but this size effect disappeared towards metamorphosis (Dziminski and Roberts 2006; Marshall and Bolton 2007). This could be due to offspring from larger eggs taking longer to resorb their yolk sacs. In addition, and contrary to our predictions, smaller clutches reached metamorphosis quicker. This last result was apparent for the Mt Fortuné/Petit Matoury population, where frogs produced the smallest clutches and had the quickest developmental rate, though this result must be taken with caution due to the low number of breeding pairs. Finally, clutch order was negatively correlated with clutch laying frequency and positively with time to metamorphosis (and there was a trend that it negatively affected survival). Combined, this suggests a reduced reproductive investment by females in later clutches, potentially due to depletion of body fat reserves, as shown in other species (Wilkinson and Gibbons 2005).
Limitations of the breeding experiments
Apart from the above-mentioned limitations regarding the lack of information about differences in habitat composition, resource availability, and genetic variation, there were three shortcomings related to the breeding experiments. First, using tadpole size at hatching as a proxy for offspring size might create biases, because these measures are not necessarily correlated (Carvajal-Castro et al. 2021). Nevertheless, we argue that using tadpole size as measure for offspring size was reasonable, because all tadpoles were raised under standardized conditions. Second, we had only 2 and 3 pairs from Bruyere and Petit-Matoury, respectively, for our breeding experiments. Although we obtained 14 and 11 clutches from these pairs, they could be considered genetic replicates, cautioning against general interpretations regarding the findings from these two populations. Third, it might be possible that experimental conditions might have been more favorable for frogs from a certain population, which might lead to observations that do not reflect natural patterns in the wild.
Conclusions and future perspectives
Our study revealed considerable intraspecific variation in sexual size dimorphism and reproductive traits among dyeing poison frog populations. Independent of the underlying mechanisms, our findings demonstrate the importance of cross-population studies when making inferences about a species. For example, much of the published research on D. tinctorius to date has focused on three populations, with most of it from a single site (Born et al. 2010; Fouilloux et al. 2021; Lawrence et al. 2019; Pašukonis et al. 2019; Rojas 2014; Rojas and Endler 2013; Rojas and Pašukonis 2019). While there is no doubt that detailed long-term studies from single field sites provide valuable knowledge about ecological patterns and processes (e.g., Brown et al. 2011; Mayer et al. 2020), we have to be careful when drawing general conclusions about a species’ ecology without accounting for intraspecific variation. Understanding intraspecific variation among populations can give us relevant information on the demographic resilience and evolutionary potential of different populations, which will be relevant for wildlife management and conservation.
Much remains to be learned about the selective pressures driving morphological differences across D. tinctorius populations. Studies regarding habitat use and selection among populations, as well as identifying variations in resource availability and prey and predator communities, could help to identify behavioral differences and the causes of intraspecific variation. Similarly, second-generation common garden experiments could help to disentangle and clarify the contribution of genetic factors versus phenotypic plasticity on morphological trait variability, such as growth rate and body size. Finally, hybridization experiments could shed more light on the level of divergence between populations and regarding patterns of potential speciation.
Data availability
Relevant raw data is available on the public repository figshare: https://doi.org/10.6084/m9.figshare.23607537.v1.
Code availability
We used standard statistical analyses that are described in sufficient detail to be replicated using provided raw data.
References
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s information criterion. J Wildl Manag 74:1175–1178
Ashton KG (2001) Body size variation among mainland populations of the western rattlesnake (Crotalus viridis). Evolution 55:2523–2533
Blanckenhorn WU (2005) Behavioral causes and consequences of sexual size dimorphism. Ethology 111:977–1016
Barton K (2020) Package ‘MuMIn’
Brown GP, Shine R (2009) Beyond size–number trade-offs: clutch size as a maternal effect. Philos Trans R Soc B Biol Sci 364:1097–1106
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Grothendieck G, Eigen C, Rcpp L (2015) Package ‘lme4’
Brown GP, Phillips BL, Shine R (2011) The ecological impact of invasive cane toads on tropical snakes: field data do not support laboratory-based predictions. Ecology 92:422–431
Brecko J, Huyghe K, Vanhooydonck B, Herrel A, Grbac I, Van Damme R (2008) Functional and ecological relevance of intraspecific variation in body size and shape in the lizard Podarcis melisellensis (Lacertidae). Biol J Linn Soc 94:251–264
Born M, Bongers F, Poelman EH, Sterck FJ (2010) Dry-season retreat and dietary shift of the dart-poison frog dendrobates tinctorius (Anura: Dendrobatidae). Phyllomedusa J Herpetol 9:37–52
Burnham KP, Anderson DR, Huyvaert KP (2011) AIC model selection and multimodel inference in behavioral ecology: some background, observations, and comparisons. Behav Ecol Sociobiol 65:23–35
Buchanan K, Burt de Perera T, Carere C, Carter T, Hailey A, Hubrecht R, Jennings D, Metcalfe N, Pitcher T, Peron F (2012) Guidelines for the treatment of animals in behavioural research and teaching. Anim Behav 83:301
Baur J, Roy J, Schäfer MA, Puniamoorthy N, Blanckenhorn WU, Rohner PT (2020) Intraspecific mating system evolution and its effect on complex male secondary sexual traits: Does male–male competition increase selection on size or shape? J Evol Biol 33:297–308
Cantera I, Coutant O, Jézéquel C, Decotte J-B, Dejean T, Iribar A, Vigouroux R, Valentini A, Murienne J, Brosse S (2022) Low level of anthropization linked to harsh vertebrate biodiversity declines in Amazonia. Nat Commun 13:3290
Chown SL, Gaston KJ (2010) Body size variation in insects: a macroecological perspective. Biol Rev 85:139–169
Carvajal-Castro JD, Vargas-Salinas F, Casas-Cardona S, Rojas B, Santos JC (2021) Aposematism facilitates the diversification of parental care strategies in poison frogs. Sci Rep 11:19047
Cvetković D, Tomašević N, Ficetola GF, Crnobrnja-Isailović J, Miaud C (2009) Bergmann’s rule in amphibians: combining demographic and ecological parameters to explain body size variation among populations in the common toad Bufo bufo. J Zool Syst Evol Res 47:171–180
Des Roches S, Pendleton LH, Shapiro B, Palkovacs EP (2021) Conserving intraspecific variation for nature’s contributions to people. Nat Ecol Evol 5:574–582
Des Roches S, Post DM, Turley NE, Bailey JK, Hendry AP, Kinnison MT, Schweitzer JA, Palkovacs EP (2018) The ecological importance of intraspecific variation. Nat Ecol Evol 2:57–64
Dugo-Cota A, Vilà C, Rodríguez A, Gonzalez‐Voyer A (2022) Influence of microhabitat, fecundity, and parental care on the evolution of sexual size dimorphism in caribbean Eleutherodactylus frogs. Evolution 76:3041–3053
Dziminski M, Roberts J (2006) Fitness consequences of variable maternal provisioning in quacking frogs (Crinia georgiana). J Evol Biol 19:144–155
Fouilloux CA, Serrano Rojas SJ, Carvajal-Castro JD, Valkonen JK, Gaucher P, Fischer MT, Pašukonis A, Rojas B (2021) Pool choice in a vertical landscape: tadpole‐rearing site flexibility in phytotelm‐breeding frogs. Ecol Evol 11:9021–9038
Gavini SS, Quintero C, Tadey M (2020) Intraspecific variation in body size of bumblebee workers influences anti-predator behaviour. J Anim Ecol 89:658–669
Gould J, Beranek C, Valdez J, Mahony M (2022) Quantity versus quality: a balance between egg and clutch size among Australian amphibians in relation to other life-history variables. Austral Ecol 47:685–697
Han X, Fu J (2013) Does life history shape sexual size dimorphism in anurans? A comparative analysis. BMC Evol Biol 13:1–11
Hughes JB, Daily GC, Ehrlich PR (1997) Population diversity: its extent and extinction. Science 278:689–692
Henry E, Santini L, Huijbregts MA, Benítez-López A (2023) Unveiling the environmental drivers of intraspecific body size variation in terrestrial vertebrates. Glob Ecol Biogeogr 32:267–280
Isaac JL (2005) Potential causes and life-history consequences of sexual size dimorphism in mammals. Mamm Rev 35:101–115
Lange L, Bégué L, Brischoux F, Lourdais O (2021) The costs of being a good dad: egg-carrying and clutch size impair locomotor performance in male midwife toads (Alytes obstetricans). Biol J Linn Soc 132:270–282
Lardner B, Loman J (2003) Growth or reproduction? Resource allocation by female frogs Rana temporaria. Oecologia 137:541–546
Lawrence JP, Rojas B, Blanchette A, Saporito RA, Mappes J, Fouquet A, Noonan BP (2023) Linking predator responses to Alkaloid variability in Poison frogs. J Chem Ecol 49:195–204
Lawrence JP, Rojas B, Fouquet A, Mappes J, Blanchette A, Saporito RA, Bosque RJ, Courtois EA, Noonan BP (2019) Weak warning signals can persist in the absence of gene flow. Proc Natl Acad Sci 116:19037–19045
Lenth R, Singmann H, Love J, Buerkner P, Herve M (2019) Package ‘emmeans’
Liang T, Dai W, Zhang Z, Bempah G, Shi L, Lu C (2023) Altitudinal gradients and body size variation among Chinese lizards in different terrains. J Zool 320:7–17
Liao WB, Liu WC, Merilä J (2015) Andrew meets Rensch: sexual size dimorphism and the inverse of Rensch’s rule in Andrew’s toad (Bufo andrewsi). Oecologia 177:389–399
Liao WB, Lu X, Jehle R (2014) Altitudinal variation in maternal investment and trade-offs between egg size and clutch size in the Andrew’s toad. J Zool 293:84–91
Miles DB, Dunham AE (1992) Comparative analyses of phylogenetic effects in the life-history patterns of iguanid reptiles. Am Nat 139:848–869
Morrison C, Hero JM (2003) Geographic variation in life-history characteristics of amphibians: a review. J Anim Ecol 72:270–279
Marshall DJ, Bolton TF (2007) Effects of egg size on the development time of non-feeding larvae. Biol Bull 212:6–11
Montiglio PO, Wey TW, Chang AT, Fogarty S, Sih A (2016) Multiple mating reveals complex patterns of assortative mating by personality and body size. J Anim Ecol 85:125–135
Mayer M, Frank SC, Zedrosser A, Rosell F (2020) Causes and consequences of inverse density-dependent territorial behaviour and aggression in a monogamous mammal. J Anim Ecol 89:577–588
Nakamura M (2010) The mechanism of sex determination in vertebrates—Are sex steroids the key-factor? J Exp Zool part A Ecol Genet Physiol 313A:381–398
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142
Noonan BP, Gaucher P (2006) Refugial isolation and secondary contact in the dyeing Poison frog dendrobates tinctorius. Mol Ecol 15:4425–4435
Nali RC, Zamudio KR, Haddad CFB, Prado CP (2014) Size-dependent selective mechanisms on males and females and the evolution of sexual size dimorphism in frogs. Am Nat 184:727–740
Olalla-Tárraga MÁ, Rodríguez MÁ (2007) Energy and interspecific body size patterns of amphibian faunas in Europe and North America: anurans follow Bergmann’s rule, urodeles its converse. Glob Ecol Biogeogr 16:606–617
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118:1883–1891
Pašukonis A, Loretto M-C, Rojas B (2019) How far do tadpoles travel in the rainforest? Parent-assisted dispersal in poison frogs. Evol Ecol 33:613–623
Pašukonis A, Serrano-Rojas SJ, Fischer M-T, Loretto M-C, Shaykevich DA, Rojas B, Ringler M, Roland AB, Marcillo-Lara A, Ringler E (2022) Contrasting parental roles shape sex differences in poison frog space use but not navigational performance. Elife 11:e80483
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Rensch B (1950) Die Abhängigkeit Der Relativen Sexualdifferenz Von Der Körpergrösse. Bonn Zool Beitr 1:58–69
Rojas B (2014) Strange parental decisions: fathers of the dyeing Poison frog deposit their tadpoles in pools occupied by large cannibals. Behav Ecol Sociobiol 68:551–559
Rojas B, Endler JA (2013) Sexual dimorphism and intra-populational colour pattern variation in the aposematic frog Dendrobates tinctorius. Evol Ecol 27:739–753
Rojas B, Pašukonis A (2019) From habitat use to social behavior: natural history of a voiceless Poison frog, Dendrodates tinctorius. PeerJ 7:e7648
Rocha S, Lima AP, Kaefer IL (2021) Key roles of paternal care and climate on offspring survival of an Amazonian poison frog. Anais Da Academia Brasileira De Ciências 93:e20210067
Shine R (1979) Sexual selection and sexual dimorphism in the Amphibia. Copeia 1979:297–306
Slatkin M (1987) Gene Flow and the Geographic structure of natural populations. Science 236:787–792
Summers K (1989) Sexual selection and intra-female competition in the green poison-dart frog, dendrobates auratus. Anim Behav 37:797–805
Shine R (2003) Reproductive strategies in snakes. Proc R Soc Lond B Biol Sci 270:995–1004
Székely T, Freckleton RP, Reynolds JD (2004) Sexual selection explains Rensch’s rule of size dimorphism in shorebirds. Proc Natl Acad Sci 101:12224–12227
Teder T, Tammaru T (2005) Sexual size dimorphism within species increases with body size in insects. Oikos 108:321–334
Warne RW, Crespi EJ (2015) Larval growth rate and sex determine resource allocation and stress responsiveness across life stages in juvenile frogs. J Exp Zool Part A Ecol Genet Physiol 323:191–201
Wells KD (2019) The ecology and behavior of amphibians. University of Chicago press, Chicago
Wilkinson LR, Gibbons JW (2005) Patterns of reproductive allocation: clutch and egg size variation in three freshwater turtles. Copeia 2005:868–879
Wollenberg KC, Mora-Ferrer LÖTTERSS, Veith M (2008) Disentangling composite colour patterns in a poison frog species. Biol J Linn Soc 93:433–444
Wollenberg KC, Veith M, Noonan BP, Lötters S (2006) Polymorphism versus species richness—systematics of large dendrobates from the eastern Guiana shield (Amphibia: Dendrobatidae). Copeia 2006:623–629
Zhang L, Lu X (2013) Sexual size dimorphism in anurans: ontogenetic determination revealed by an across-species comparison. Evol Biol 40:84–91
Zuur AF, Ieno EN (2016) A protocol for conducting and presenting results of regression-type analyses. Methods Ecol Evol 7:636–645
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Acknowledgements
We are thankful to the staff of CNRS Guyane and the National Forests Office for logistic support and valuable information regarding field sites, and to the Nouragues Research Station, which benefits from “Investissement d’Avenir” grants managed by Agence Nationale de la Recherche (AnaEE France ANR-11- INBS-0001; Labex CEBA ANR-10-LABX-25-01). BR is part of a multi-researcher partnership with the Nouragues Nature Reserve (Convention Cadre de Partenariat N°01-2019) aimed at improving and spreading knowledge about amphibians. We also would like to thank Cyril Gaertner, Cristina Marqués Ferri, Constance Laccours, Camille Gerard, and Philippe Gaucher for their precious help on PLEEA. Finally, we would like to thank two anonymous reviewers for clarifying and improving the quality of our manuscript.
Funding
Open access funding provided by Inland Norway University Of Applied Sciences. B.R., L.S.J., C.D., and M.M. received funding from the Research Council of Finland (Academy Research Fellowship, Decision Nos. 318404 and 319949 to BR). LS received funds for research activities abroad from the International Relations Office of the University of Veterinary Medicine Vienna (KUWI_MA). This research was supported by the French National Agency for Research (ANR) grant RANAPOSA (ref. ANR-20-CE02-0003) and from an “Investissement d’Avenir” grant CEBA (ref. ANR-10-LABX-25-01) to M.C., and from a doctoral grant from the Ministère de l’enseignement supérieur, de la recherche et de l’innovation to U.L.C.
Author information
Authors and Affiliations
Contributions
All authors developed the ideas for this study. LSJ, MM, and CD conducted field observations, and ULC and MC conducted breeding experiments. MM conducted the analyses, and LSJ and MM wrote the first draft of the manuscript. All authors substantially contributed to the following drafts.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
All capture and handling procedures were approved by the Prefet de la Région Guyane (R03-2022-12-28-00004). Protocols employed at Nouragues Research Station were also approved by the Reserve’s scientific committee. For breeding experiments, we obtained permits to collect frogs from the Prefet de la Région Guyane (Collection permit R03-2021-03-03-004). Authorization to maintain animals in captivity was also given by the Prefet de la Région Guyane (Agréement A973-02-002). Authorization to perform breeding experiments was provided by the Ministère de l’enseignement supérieur et de la Recherche (référence APAFIS #38380-2022090708128291). Our study met the ASAB/ABS Guidelines for the treatment of animals in research and teaching ASAB/ABS (Buchanan et al. 2012). None of the frogs were injured during capture and handling and were successfully released at the site of capture after handling (except the individuals kept for breeding experiments). No subsequent long-term effects of capture and handling were observed (based on the frogs in captivity).
Consent for publication
All authors agreed with the content and gave explicit consent to submit and have obtained consent from the responsible authorities at the organization where the work has been carried out.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schlippe Justicia, L., Mayer, M., Lorioux-Chevalier, U. et al. Intraspecific divergence of sexual size dimorphism and reproductive strategies in a polytypic poison frog. Evol Ecol 38, 121–139 (2024). https://doi.org/10.1007/s10682-023-10280-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-023-10280-2