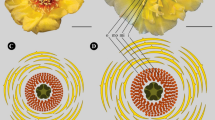
Abstract
Plant surface microstructure and its ultrastructural cuticular patterns, especially of the petal surface, is diverse in nature, and has associated diverse biological functions. These microstructures can be primary as well as secondary, i.e. hierarchical in development. Since the discovery of these structures over a century ago, many functional aspects about them, including biological, chemical, and physical, have been constantly redefined over the years. Funneliform flowers, an example of synorganization in corolla, are known to be a derived trait in angiosperms. As for pollinator interaction, gamopetalous corolla is a definite advantage because it provides a sturdy seat or a sturdy receptacle for the pollinator depending on the type of pollinator. Petal folds likewise play a role in increasing the total size of the interacting surface of the petal with the pollinator without losing compactness in the bargain. We illustrate that petal folds have multiple evolutionary occurrences in Acanthaceae as well as in Asterids and therefore provide a strong reason for their functional association with the corresponding types of floral forms. We present observations on petal folds and their surface microstructure in two Ruellia species studied, and note that the microstructure differs in different regions containing petal folds—denser on out-folds compared to the in-folds—throughout the throat of the funneliform corolla. We then discuss: (a) how these secondary microstructures differentiating in the two types of folds could be associated with varied functional aspects, and (b) what might be governing this differential development of cuticular secondary microstructure, through some hypotheses.




Similar content being viewed by others
Data availability
The manuscript does not use reportable data, all observations presented in the article are the authors’ own and in this sense original.
References
Baker EA (1970) The morphology and composition of isolated plant cuticles. New Phytol 69:1053–1058. https://doi.org/10.2307/2430987
Bargel H, Koch K, Cerman Z, Neinhuis C (2006) Structure-function relationships of the plant cuticle and cuticular waxes: a smart material? Funct Plant Biol 33:893–910. https://doi.org/10.1071/FP06139
Barthlott W (1980) Morphogenese und mikromorphologie komplexer cuticular-faltungsmuster an blu¨ten-trichomen von Antirrhinum L. (Scrophulariaceae). Berichte Der Deutschen Botanischen Gesellschaft 93:379–390. https://doi.org/10.1111/j.1438-8677.1980.tb03348.x
Barthlott W (1981) Epidermal and seed surface characters of plants: systematic applicability and some evolutionary aspects. Nord J Bot 1:345–355. https://doi.org/10.1111/j.1756-1051.1981.tb00704.x
Barthlott W, Mail M, Bhushan B, Koch K (2017) Plant surfaces: structures and functions for biomimetic innovations. Nano-Micro Lett 9:23. https://doi.org/10.1007/s40820-016-0125-1
Bräuer P, Neinhuis C, Voigt D (2017) Attachment of honeybees and greenbottle flies to petal surfaces. Arthropod-Plant Interact 11(2):171–192
Berry E, Geeta R (2019) Variation in surface microstructure of the corolla with respect to pigment pattern in some Acanthaceae. Phytomorphology 69:25–31
Buschhaus C, Peng C, Jetter R (2013) Very-long-chain 1, 2-and 1, 3-bifunctional compounds from the cuticular wax of Cosmos bipinnatus petals. Phytochemistry 91:249–256
Buschhaus C, Hager D, Jetter R (2015) Wax layers on Cosmos bipinnatus petals contribute unequally to total petal water resistance. Plant Physiol 167:80–88
Cao Y, Jana S, Bowen L, Tan X, Liu H, Rostami N, Brown J, Jakubovics NS, Chen J (2019) Hierarchical rose petal surfaces delay the early-stage bacterial biofilm growth. Langmuir 35(45):14670–14680
Cheng G, Huang H, Zhou L, He S, Zhang Y (2019) Chemical composition and water permeability of the cuticular wax barrier in rose leaf and petal: a comparative investigation. Plant Physiol Biochem 135:404–410
Endress PK (2016) Development and evolution of extreme synorganization in angiosperm flowers and diversity: a comparison of Apocynaceae and Orchidaceae. Ann Bot 117:749–767
Feng L, Zhang Y, Cao Y, Ye X, Jiang L (2011) The effect of surface microstructures and surface compositions on the wettabilities of flower petals. Soft Matter 7:2977–2980. https://doi.org/10.1039/C0SM01032D
Garcia JE, Shrestha M, Ospina-Rozo L, Dekiwadia C, Field MR, Ma JS, Tran N, Dyer AG, Fox K, Greentree AD (2020) Iridescence and hydrophobicity have no clear delineation that explains flower petal micro-surface. Sci Rep 101–2.
Glover BJ, Whitney HM (2010) Structural colour and iridescence in plants: the poorly studied relations of pigment colour. Ann Bot 105(4):505–511
Goodwin SM, Kolosova N, Kish CM, Wood KV, Dudareva N, Jenks MA (2003) Cuticle characteristics and volatile emissions of petals in Antirrhinum majus. Physiol Plant 117(3):435–443
Graham VA (1988) Delimitation and infra-generic classification of Justicia (Acanthaceae). Kew Bull 43:551–624
Hawkeswood TJ, Sommung B (2016) Pollination of Ruellia simplex C. Wright (Acanthaceae) by the Giant Tropical Bee, Apis dorsata (Fabr., 1793) (Hymenoptera: Apidae) in Bangkok. Thailand Calodema 438:1–5
Hu Z, Liu Z, Zhao J, Yu X, Lu B (2021) Rose-petals-derived hemispherical micropapillae carbon with cuticular folds for super potassium storage. Electrochim Acta 368:137629
Jeffree CE (2006) Annual Plant Reviews Volume 23: Biology of the Plant Cuticle. ed M. Riederer and C. Müller. Blackwell Publishing, Oxford, pp 11–125
Kannangara R, Branigan C, Liu Y, Penfield T, Rao V, Mouille G, Hofte H, Pauly M, Riechmann JL, Broun P (2007) The transcription factor WIN1/SHN1 regulates cutin biosynthesis in Arabidopsis thaliana. Plant Cell 19:1278–1294. https://doi.org/10.1105/tpc.106.047076
Kay QON, Daoud HS, Stirton CH (1981) Pigment distribution, light reflection and cell structure in petals. Bot J Linn Soc 83:57–83. https://doi.org/10.1111/j.1095-8339.1981.tb00129.x
Koch K, Bohn HF, Barthlott W (2009) Hierarchically sculptured plant surfaces and superhydrophobicity. Langmuir 25:14116–14120. https://doi.org/10.1021/la9017322
Kolattukudy PE (1980) Biopolyester membranes of plants: cutin and suberin. Science 208:990–1000. https://doi.org/10.1126/science.208.4447.990
Lai PH, Huang LM, Pan ZJ, Jane WN, Chung MC, Chen WH, Chen HH (2020) PeERF1, a SHINE-like transcription factor, is involved in nanoridge development on lip epidermis of Phalaenopsis flowers. Front Plant Sci 10:1709. https://doi.org/10.3389/fpls.2019.01709
Liu F, Xiong X, Wu L, Fu D, Hayward A, Zeng X, Cao Y, Wu Y, Li Y, Wu G (2014) BraLTP1, a lipid transfer protein gene involved in epicuticular wax deposition, cell proliferation and flower development in Brassica napus. PLoS ONE 9:e110272. https://doi.org/10.1371/journal.pone.0110272
Mabberley DJ (2008) Mabberley’s plant-book: a portable dictionary of plants, their classification and uses, 3rd edn. Cambridge University Press, Cambridge
McDade LA, Daniel TF, Kiel CA (2008) Toward a comprehensive understanding of phylogenetic relationships among lineages of Acanthaceae sl (Lamiales). Am J Bot 95:1136–1152. https://doi.org/10.3732/ajb.0800096
Neinhuis C, Barthlott W (1997) Characterization and distribution of water-repellent, self-cleaning plant surfaces. Ann Bot 79:667–677
Panikashvili D, Shi JX, Bocobza S, Franke RB, Schreiber L, Aharoni A (2010) The Arabidopsis DSO/ABCG11 transporter affects cutin metabolism in reproductive organs and suberin in roots. Mol Plant 3:563–575. https://doi.org/10.1093/mp/ssp103
Pfündel EE, Agati G, Cerovic ZG (2008) Optical properties of plant surfaces. Biol Plant Cuticle 3:216–239
Pighin JA, Zheng H, Balakshin LJ, Goodman IP, Western TL, Jetter R, Kunst L, Samuels AL (2004) Plant cuticular lipid export requires an ABC transporter. Science 306:702–704. https://doi.org/10.1126/science.1102331
Priestley JH (1943) The cuticle in angiosperms. Bot Rev 9:593. https://doi.org/10.1007/BF02872505
Samuels L, Kunst L, Jetter R (2008) Sealing plant surfaces: cuticular wax formation by epidermal cells. Annu Rev Plant Biol 59:683–707. https://doi.org/10.1146/annurev.arplant.59.103006.093219
Shi JX, Malitsky S, De Oliveira S, Branigan C, Franke RB, Schreiber L, Aharoni A (2011) SHINE transcription factors act redundantly to pattern the archetypal surface of Arabidopsis flower organs. PLoS Genet 7(5):e1001388
Takeda S, Iwasaki A, Matsumoto N, Uemura T, Tatematsu K, Okada K (2013) Physical interaction of floral organs controls petal morphogenesis in Arabidopsis. Plant Physiol 161:1242–1250. https://doi.org/10.1104/pp.112.212084
To A, Joubes J, Barthole G, Lecureuil A, Scagnelli A, Jasinski S, Lepiniec L, Baud S (2012) WRINKLED transcription factors orchestrate tissue-specific regulation of fatty acid biosynthesis in Arabidopsis. Plant Cell 24:5007–5023. https://doi.org/10.1105/tpc.112.106120
Tripp EA, Manos PS (2008) Is floral specialization an evolutionary dead-end? Pollination system transitions in Ruellia (Acanthaceae). Evolut Int J Org Evolut 62:1712–1737. https://doi.org/10.1111/j.1558-5646.2008.00398.x
Whitney H, Kolle M, Alvarez-Fernandez R, Steiner U, Glover B (2009) Contributions of iridescence to floral patterning. Commun Integr Biol 2:230–232
Whitney HM, Bennett KV, Dorling M, Sandbach L, Prince D, Chittka L, Glover BJ (2011) Why do so many petals have conical epidermal cells? Ann Bot 108:609–616
Wilson SB, Flory AL (2012) FloraGator: A novel, interactive and online multiple entry key for identifying plant families. HortTechnology 22:410–412
Wolff C (1996) The art of manipulating fabric. Krause Publications, Iola, p 312
Yeats TH, Rose JK (2013) The formation and function of plant cuticles. Plant Physiol 163:5–20. https://doi.org/10.1104/pp.113.222737
Yu YM, Li XX, Xie D, Wang H (2021) Horizontal orientation of zygomorphic flowers: significance for rain protection and pollen transfer. Plant Biol 23:156–161
Acknowledgements
EB is supported by a Maharishi Kanad Postdoctoral Fellowship, Institution of Eminence-Delhi University. AKC is supported by Academia Sinica and Taiwan Ministry of Science and Technology. All authors acknowledge the scientific environment provided by the Department of Botany, University of Delhi for free interaction and open observations. The authors thank the editor and the two reviewers for critical and constructive comments that helped improve the manuscript.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
EB conceived the idea with considerable inputs from AKC; EB and AKC came out with the hypotheses; while RG substantially contributed to the intellectual development of the same; EB led the writing of the manuscript with considerable inputs from AKC. RG gave final critical comments on the draft. All authors gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Berry, E., Choudhary, A.K. & Geeta, R. Why do some funneliform flowers have petal folds accompanied with hierarchical surface microstructure?. Evol Ecol 37, 385–399 (2023). https://doi.org/10.1007/s10682-022-10217-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-022-10217-1