Abstract
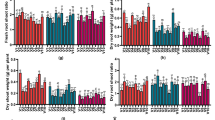
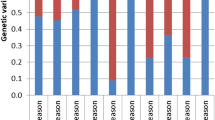
Genetic architecture of seedling drought tolerance is complex and needs to be better understood. To address this challenge, we developed a protocol to identify the most promising drought-tolerant genotypes at the seedling stage in winter wheat. A population of 146 recombinant inbred lines (F9) derived from a cross between wheat cultivars, ‘Harry’ (seedling drought tolerant) and ‘Wesley’ (seedling drought susceptible) were used in this study. All genotypes were sown in three replications in a randomized complete block design under controlled conditions in a greenhouse. Seven traits were scored and grouped into tolerance traits; days to wilting, leaf wilting, and stay green and survival traits; days to regrowth, regrowth, drought survival rate, and recovery after irrigation. Three selection indices were calculated (1) tolerance index, (2) survival index, and (3) drought tolerance index (DTI). The same set of genotypes were also tested for grain yield in two low rainfall environments for two seasons. High genetic variation was found among all genotypes for all seedling traits scored in this study. Correlations between tolerance and survival traits were weak or did not exist. Heritability estimates ranged from 0.53 to 0.88. DTI had significant phenotypic and genotypic correlations with all seedling traits. Genotypes were identified with a high drought tolerance at the seedling stage combined with high grain yield in low rainfall. Breeding for tolerance and survival traits should be taken into account for improving winter wheat drought tolerance at seedling stage. The selected genotypes can be used for to further improve drought tolerance in high yielding wheat for Nebraska.







Similar content being viewed by others
Abbreviations
- v-set:
-
Verification set
- DTW:
-
Days to wilting
- LW:
-
Leaf wilting
- S_LW:
-
Sum of leaf wilting
- SG:
-
Stay green
- DTR:
-
Days to recovery
- RB:
-
Regrowth biomass
- DSR:
-
Drought survival rate
- RAI:
-
Recovery after irrigation
- RI:
-
Recovery index
- TI:
-
Tolerance index
- DTI:
-
Drought tolerance index
References
Abid M, Tian Z, Ata-Ul-Karim ST et al (2016) Nitrogen nutrition improves the potential of wheat (Triticum aestivum L.) to alleviate the effects of drought stress during vegetative growth periods. Front Plant Sci 7:981. https://doi.org/10.3389/fpls.2016.00981
Andrade-Sanchez P, Gore MA, Heun JT et al (2014) Development and evaluation of a field-based high-throughput phenotyping platform. Funct Plant Biol 41:68. https://doi.org/10.1071/FP13126
Araus JL, Cairns JE (2014) Field high-throughput phenotyping: the new crop breeding frontier. Trends Plant Sci 19:52–61. https://doi.org/10.1016/j.tplants.2013.09.008
Awlia M, Nigro A, Fajkus J et al (2016) High-throughput non-destructive phenotyping of traits that contribute to salinity tolerance in Arabidopsis thaliana. Front Plant Sci 7:1414. https://doi.org/10.3389/fpls.2016.01414
Bai G, Ge Y, Hussain W et al (2016) A multi-sensor system for high throughput field phenotyping in soybean and wheat breeding. Comput Electron Agric 128:181–192. https://doi.org/10.1016/j.compag.2016.08.021
Baker RJ (1986) Selection indices in plant breeding. CRC Press, Boca Raton
Barmeier G, Schmidhalter U (2016) High-throughput phenotyping of wheat and barley plants grown in single or few rows in small plots using active and passive spectral proximal sensing. Sensors (Basel). https://doi.org/10.3390/s16111860
Blum A (2011) The moisture environment. In: Plant breeding for water-limited environments. Springer, New York, pp 1–9. https://link.springer.com/chapter/10.1007/978-1-4419-7491-4_1
Bowne JB, Erwin TA, Juttner J et al (2012) Drought responses of leaf tissues from wheat cultivars of differing drought tolerance at the metabolite level. Mol Plant 5:418–429. https://doi.org/10.1093/mp/ssr114
Butler DG, Cullis BR, Gilmour AR, Gogel BJ (2009) ASReml-R reference manual. http://www.citeulike.org/user/vprieto/article/10128936
Christopher JT, Christopher MJ, Borrell AK et al (2016) Stay-green traits to improve wheat adaptation in well-watered and water-limited environments. J Exp Bot 67:5159–5172. https://doi.org/10.1093/jxb/erw276
Drira M, Hanin M, Masmoudi K et al (2016) Comparison of full-length and conserved segments of wheat dehydrin DHN-5 overexpressed in Arabidopsis thaliana showed different responses to abiotic and biotic stress. Funct Plant Biol 43:1048. https://doi.org/10.1071/FP16134
Falconer D, Mackay T (1996) Introduction to quantitative genetics, 4th edn. Prentice Hall, Harlow
Gallagher JN, Biscoe PV, Hunter B (1976) Effects of drought on grain growth. Nature 264:541–542. https://doi.org/10.1038/264541a0
Gebre-Mariam H, Larter EN (1996) Genetic response to index selection for grain yield, kernel weight and per cent protein in four wheat crosses. Plant Breed 115:459–464. https://doi.org/10.1111/j.1439-0523.1996.tb00957.x
Grewal KS, Buchan GD, Tonkin PJ (1990) Estimation of field capacity and wilting point of some New Zealand soils from their saturation percentages. New Zeal J Crop Hortic Sci 18:241–246. https://doi.org/10.1080/01140671.1990.10428101
Harb A, Krishnan A, Ambavaram MMR, Pereira A (2010) Molecular and physiological analysis of drought stress in Arabidopsis reveals early responses leading to acclimation in plant growth. Plant Physiol 154:1254–1271. https://doi.org/10.1104/pp.110.161752
Hussain W, Stephen Baenziger P, Belamkar V et al (2017) Genotyping-by-sequencing derived high-density linkage map and its application to QTL mapping of flag leaf traits in bread wheat. Sci Rep 7:16394. https://doi.org/10.1038/s41598-017-16006-z
Hussain W, Guttieri MJ, Belamkar V et al (2018) Registration of a bread wheat recombinant inbred line mapping population derived from a cross between ‘Harry’ and ‘Wesley’. J Plant Regist. https://doi.org/10.3198/jpr2017.11.0085crmp
Izanloo A, Condon AG, Langridge P et al (2008) Different mechanisms of adaptation to cyclic water stress in two South Australian bread wheat cultivars. J Exp Bot 59:3327–3346. https://doi.org/10.1093/jxb/ern199
Laurie SM, Calitz FJ, Adebola PO, Lezar A (2013) Characterization and evaluation of South African sweet potato (Ipomoea batatas (L.) LAM) land races. South Afr J Bot 85:10–16. https://doi.org/10.1016/j.sajb.2012.11.004
Lawlor DW, Cornic G (2002) Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant, Cell Environ 25:275–294. https://doi.org/10.1046/j.0016-8025.2001.00814.x
Li L, van Staden J (1998) Effects of plant growth regulators on the antioxidant system in callus of two maize cultivars subjected to water stress. Plant Growth Regul 24:55–66. https://doi.org/10.1023/A:1005954532397
Lyon DJ, Hergert GW (2012) Nitrogen fertility in semiarid dryland wheat production is challenging for beginning organic farmers. http://digitalcommons.unl.edu/panhandleresext
Maathuis FJM, Filatov V, Herzyk P et al (2003) Transcriptome analysis of root transporters reveals participation of multiple gene families in the response to cation stress. Plant J 35:675–692
Mart KB, Veneklaas EJ, Bramley H (2016) Osmotic potential at full turgor: an easily measurable trait to help breeders select for drought tolerance in wheat. Plant Breed 135:279–285. https://doi.org/10.1111/pbr.12367
Mueller SC, Teuber LR (2007) Alfalfa growth and development. In: Irrigated alfalfa management for mediterranean and desert zones. http://alfalfa.ucdavis.edu/IrrigatedAlfalfa/pdfs/UCAlfalfa8289GrowthDev_free.pdf
Muir CD, Thomas-Huebner M (2015) Constraint around quarter-power allometric scaling in wild tomatoes (Solanum sect. Lycopersicon; Solanaceae). Am Nat 186:421–433. https://doi.org/10.1086/682409
Mwadzingeni L, Shimelis H, Dube E et al (2016) Breeding wheat for drought tolerance: progress and technologies. J Integr Agric 15:935–943. https://doi.org/10.1016/S2095-3119(15)61102-9
Neuman H, Galpaz N, Cunningham FX et al (2014) The tomato mutation nxd1 reveals a gene necessary for neoxanthin biosynthesis and demonstrates that violaxanthin is a sufficient precursor for abscisic acid biosynthesis. Plant J 78:80–93. https://doi.org/10.1111/tpj.12451
Nezhadahmadi A, Prodhan ZH, Faruq G (2013) Drought tolerance in wheat. Sci World J 2013:610721. https://doi.org/10.1155/2013/610721
Omotobora BO (2013) Evaluation of selected sweetpotato (Ipomoea batatas) accessions for drought tolerance. University of South Africa. http://hdl.handle.net/10500/10490
Omotobora BO, Adebola PO, Modise DM et al (2014) Greenhouse and field evaluation of selected Sweetpotato (Ipomoea batatas (L.) LAM) accessions for drought tolerance in South Africa. Am J Plant Sci 05:3328–3339. https://doi.org/10.4236/ajps.2014.521348
Pearce RS (1985) The membranes of slowly drought-stressed wheat seedlings: a freeze-fracture study. Planta 166:1–14. https://doi.org/10.1007/BF00397380
Peterson JC (1992) Similarities among test sites based on cultivar performance in the hard red winter wheat region. Crop Sci 32:907. https://doi.org/10.2135/cropsci1992.0011183X003200040014x
Rebetzke GJ, Jimenez-Berni JA, Bovill WD et al (2016) High-throughput phenotyping technologies allow accurate selection of stay-green. J Exp Bot 67:4919–4924. https://doi.org/10.1093/jxb/erw301
Reynolds P, Ortiz-Monasterio J, McNab A et al (2001) Application of physiology in wheat breeding M physiology in wheat breeding. CIMMYT, Mexico, D.F
Ritchie SW, Nguyen HT, Holaday AS (1990) Leaf water content and gas-exchange parameters of two wheat genotypes differing in drought resistance. Crop Sci 30:105. https://doi.org/10.2135/cropsci1990.0011183X003000010025x
Rivero RM, Kojima M, Gepstein A et al (2007) Delayed leaf senescence induces extreme drought tolerance in a flowering plant. Proc Natl Acad Sci USA 104:19631–19636. https://doi.org/10.1073/pnas.0709453104
Roth F, Link W (2010) Selection on freezing-tolerance of faba bean (Vicia faba L.): improvement of methods and results. In: 60. Jahrestagung der Vereinigung der Pflanzenzüchter und Saatgutkaufleute Österreichs, 24–26 November 2009, Raumberg-Gumpenstein. pp 31–37
Salgado JD, Madden LV, Paul PA (2015) Quantifying the effects of fusarium head blight on grain yield and test weight in soft red winter wheat. Phytopathology 105:295–306. https://doi.org/10.1094/PHYTO-08-14-0215-R
Sallam A, Hamed E-S, Hashad M, Omara M (2014) Inheritance of stem diameter and its relationship to heat and drought tolerance in wheat (Triticum aestivum L.). J Plant Breed Crop Sci 6:11–23. https://doi.org/10.5897/jpbcs11.017
Sallam A, Hashad M, Hamed E-S, Omara M (2015a) Genetic variation of stem characters in wheat and their relation to kernel weight under drought and heat stresses. J Crop Sci Biotechnol. https://doi.org/10.1007/s12892-015-0014-z
Sallam A, Martsch R, Moursi YS (2015b) Genetic variation in morpho-physiological traits associated with frost tolerance in faba bean (Vicia faba L.). Euphytica 205:395–408. https://doi.org/10.1007/s10681-015-1395-2
Sallam A, Dhanapal AP, Liu S (2016) Association mapping of winter hardiness and yield traits in faba bean (Vicia faba L.). Crop Pasture Sci 67:55. https://doi.org/10.1071/cp15200
Sanad MNME, Campbell KG, Gill KS (2016) Developmental program impacts phenological plasticity of spring wheat under drought. Bot Stud 57:35. https://doi.org/10.1186/s40529-016-0149-3
Sankar B, Jaleel CA, Manivannan P et al (2008) Relative efficacy of water use in five varieties of Abelmoschus esculentus (L.) Moench. under water-limited conditions. Colloids Surf B Biointerfaces 62:125–129. https://doi.org/10.1016/j.colsurfb.2007.09.025
Scandalios JG (1993) Oxygen stress and superoxide dismutases. Plant Physiol 101:7–12
Tahara M, Carver BF, Johnson RC, Smith EL (1990) Relationship between relative water content during reproductive development and winter wheat grain yield. Euphytica 49:255–262. https://doi.org/10.1007/BF00036297
Thomas H, Howarth CJ (2000) Five ways to stay green. J Exp Bot. https://doi.org/10.1093/jexbot/51.suppl_1.329
Timmusk S, Abd El-Daim IA, Copolovici L et al (2014) Drought-tolerance of wheat improved by rhizosphere bacteria from harsh environments: enhanced biomass production and reduced emissions of stress volatiles. PLoS ONE 9:e96086. https://doi.org/10.1371/journal.pone.0096086
Utz FH (1991) PLABSTAT: A computer program for statistical analysis of plant breeding experiments. 44. https://fsc.uni-hohenheim.de/fileadmin/einrichtungen/plant-breeding/plabstat_manual_ger.pdf
van Rensburg L, Krüger GHJ (1994) Evaluation of components of oxidative stress metabolism for use in selection of drought tolerant cultivars of Nicotiana tabacum L. J Plant Physiol 143:730–737. https://doi.org/10.1016/S0176-1617(11)81166-1
Yordanov I, Velikova V, Tsonev T (2000) Plant responses to drought, acclimation, and stress tolerance. Photosynthetica 38:171–186. https://doi.org/10.1023/A:1007201411474
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421. https://doi.org/10.1111/j.1365-3180.1974.tb01084.x
Zhu J-K (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273. https://doi.org/10.1146/annurev.arplant.53.091401.143329
Acknowledgements
We thank Marry Guttieri for her contribution in developing HW RILs population. The authors thank Prof. Dr. Kent Eskridge, University of Nebraska-Lincoln, for his valuable recommendations on the design of greenhouse experiments.
Funding
This work is partially funded by the Minister of Higher Education, Egypt and the University of Nebraska-Lincoln, USA.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sallam, A., Mourad, A.M.I., Hussain, W. et al. Genetic variation in drought tolerance at seedling stage and grain yield in low rainfall environments in wheat (Triticum aestivum L.). Euphytica 214, 169 (2018). https://doi.org/10.1007/s10681-018-2245-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-018-2245-9