Abstract
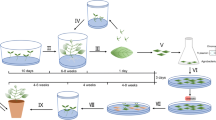
Compact growth is an important quality criterion in horticulture. Many Campanula species and cultivars exhibit elongated growth which is suppressed by chemical retardation and cultural practice during production to accommodate to the consumer’s desire. The production of compact plants via transformation with wild type Agrobacterium rhizogenes is an approach with great potential to produce plants that are non-GMO. Efficient transformation and regeneration procedures vary widely among both plant genera and species. Here we present a transformation protocol for Campanula. Hairy roots were produced on 26–90% of the petioles that were used for transformation of C. portenschlagiana (Cp), a C. takesimana × C. punctata hybrid (Chybr) and C. glomerata (Cg). Isolated hairy roots grew autonomously and vigorously without added hormones. The Cg hairy roots produced chlorophyll and generated plantlets in response to treatments with cytokinin (42 µM 2iP) and auxin (0.67 µM NAA). In contrast, regeneration attempts of transformed Cp and Chybr roots lead neither to the production of chlorophyll nor to the regeneration of shoots. Agropine A. rhizogenes strains integrate split T-DNA in TL- and TR-DNA fragments into the plant genome. In this study, regenerated plants of Cg did not contain TR-DNA, indicating that a selective pressure against this T-DNA fragment may exist in Campanula.



Similar content being viewed by others
References
Alpizar E et al (2008) Agrobacterium rhizogenes-transformed roots of coffee (Coffea arabica): conditions for long-term proliferation, and morphological and molecular characterization. Ann Bot 101:929–940
Bulgakov VP (2008) Functions of rol genes in plant secondary metabolism. Biotechnol Adv 26:318–324
Casanova E, Trillas MI, Moysset L, Vainstein A (2005) Influence of rol genes in floriculture. Biotechnol Adv 23:3–39
Chandra S (2012) Natural plant genetic engineer Agrobacterium rhizogenes: role of T-DNA in plant secondary metabolism. Biotechnol Lett 34:407–415
Choi PS, Kim YD, Choi KM, Chung HJ, Choi DW, Liu JR (2004) Plant regeneration from hairy-root cultures transformed by infection with Agrobacterium rhizogenes in Catharanthus roseus. Plant Cell Rep 22:828–831
Chriqui D, Guivarc’h A, Dewitte W, Prinsen E, van Onkelen H (1996) Rol genes and root initiation and development. Plant Soil 187:47–55
Christensen B, Sriskandarajah S, Serek M, Müller R (2008) Transformation of Kalanchoe blossfeldiana with rol-genes is useful in molecular breeding towards compact growth. Plant Cell Rep 27:1485–1495
Christensen B, Müller R, Sriskandarajah S (2009) Transformation of Hibiscus rosa-sinensis L. by Agrobacterium rhizogenes. J Hortic Sci Biotech 84:204–208
Clifford SC, Runkle ES, Langton FA, Mead A, Foster SA, Pearson S, Heins RD (2004) Height control of poinsettia using photoselective filters. HortScience 39:383–387
Dai W, Castillo C (2007) Factors affecting plant regeneration from leaf tissues of Buddleia species. HortScience 42:1670–1673
De Castro VL, Goes KP, Chiorato SH (2004) Developmental toxicity potential of paclobutrazol in the rat. Int J Environ Health Res 14:371–380
EU (2001) Directive 2001/18/EC of the European Parliament and of the Council of 12 March 2001 on the deliberate release into the environment of genetically modified organisms and repealing Council Directive 90/220/EEC. OJ L 106/1–38
Ferrante A, Trivellini A, Scuderi D, Romano D, Vernieri P (2015) Post-production physiology and handling of ornamental potted plants. Postharvest Biol Technol 100:99–108
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451
Intrieri MC, Buiatti M (2001) The horizontal transfer of Agrobacterium rhizogenes genes and the evolution of the genus Nicotiana. Mol Phylogenet Evol 20:100–110
Jensen L, Hegelund JN, Olsen A, Lütken H, Müller R (2016) A natural frameshift mutation in Campanula EIL2 correlates with ethylene insensitivity in flowers. BMC Plant Biol 16:117
Jian B et al (2009) Agrobacterium rhizogenes-mediated transformation of superroot-derived Lotus corniculatus plants: a valuable tool for functional genomics. BMC Plant Biol 9:78
Jouanin L, Guerche P, Pamboukdjian N, Tourneur C, Delbart FC, Tourneur J (1987) Structure of T-DNA in plants regenerated from roots transformed by Agrobacterium rhizogenes strain A4. Mol Gen Genet 206:387–392
Kyndt T et al (2015) The genome of cultivated sweet potato contains Agrobacterium T-DNAs with expressed genes: an example of a naturally transgenic food crop. Proc Natl Acad Sci USA 112:5844–5849
Latimer JG, Thomas PA (1991) Application of brushing for growth control of tomato transplants in a commercial setting. HortTechnology 1:109–110
Lima JE, Benedito VA, Figueira A, Peres LEP (2009) Callus, shoot and hairy root formation in vitro as affected by the sensitivity to auxin and ethylene in tomato mutants. Plant Cell Rep 28:1169–1177
Lütken H, Jensen LS, Topp SH, Mibus H, Müller R, Rasmussen SK (2010) Production of compact plants by overexpression of AtSHI in the ornamental Kalanchoë. Plant Biotechnol J 8:211–222
Lütken H, Laura M, Borghi C, Ørgaard M, Allavena A, Rasmussen S (2011) Expression of KxhKN4 and KxhKN5 genes in Kalanchoë blossfeldiana ‘Molly’ results in novel compact plant phenotypes: towards a cisgenesis alternative to growth retardants. Plant Cell Rep 30:2267–2279
Lütken H, Clarke JL, Muller R (2012a) Genetic engineering and sustainable production of ornamentals: current status and future directions. Plant Cell Rep 31:1141–1157
Lütken H, Jensen EB, Wallström SV, Müller R, Christensen B (2012b) Development and evaluation of a non-gmo breeding technique exemplified by Kalanchoë. Acta Hort 961:51–58
Lütken H, Wallström SV, Jensen EB, Christensen B, Müller R (2012c) Inheritance of rol -genes from Agrobacterium rhizogenes through two generations in Kalanchoë. Euphytica 188:397–407
Majumdar S, Garai S, Jha S (2011) Genetic transformation of Bacopa monnieri by wild type strains of Agrobacterium rhizogenes stimulates production of bacopa saponins in transformed calli and plants. Plant Cell Rep 30:941–954
Matveeva TV, Bogomaz DI, Pavlova OA, Nester EW, Lutova LA (2012) Horizontal gene transfer from genus Agrobacterium to the plant Linaria in nature. Mol Plant-Microbe Interact 25:1542–1551
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Ozyigit II, Dogan I, Tarhan EA (2013) Agrobacterium rhizogenes-mediated transformation and its biotechnological applications in crops. In: Hakeem KR, Ahmad P, Ozturk M (eds) Crop improvement. Springer, New York, pp 1–48
Park N, Tuan P, Li X, Kim Y, Yang T, Park S (2011) An efficient protocol for genetic transformation of Platycodon grandiflorum with Agrobacterium rhizogenes. Mol Biol Rep 38:2307–2313
Rademacher W (2000) Growth retardents: effects on gibberellin biosynthesis and other metabolic pathways. Annu Rev Plant Physiol Plant Mol Biol 51:501–531
Seglie L, Scariot V, Larcher F, Devecchi M, Chiavazza PM (2011) In vitro seed germination and seedling propagation in Campanula spp. Plant Biosyst 146:15–23
Sivanesan I, Lim M, Jeong B (2011) Somatic embryogenesis and plant regeneration from leaf and petiole explants of Campanula punctata Lam. var. rubriflora Makino. Plant Cell Tissue Organ Cult 107:365–369
Skoog F, Miller CO (1957) Chemical regulation of growth and organ formation in plant tissue cultures in vitro. Symp Soc Exp Biol 11:118–131
Slightom JL, Jouanin L, Leach F, Drong RF, Tepfer D (1985) Isolation and identification of TL-DNA/plant junctions in Convolvulus arvensis transformed by Agrobacterium rhizogenes strain A4. EMBO J 4:3069–3077
Sørensen MT, Danielsen V (2006) Effects of the plant growth regulator, chlormequat, on mammalian fertility. Int J Androl 29:129–133
Sriskandarajah S, Frello S, Jørgensen K, Serek M (2004) Agrobacterium tumefaciens-mediated transformation of Campanula carpatica: factors affecting transformation and regeneration of transgenic shoots. Plant Cell Rep 23:59–63
Sriskandarajah S, Mibus H, Serek M (2008) Regeneration and transformation in adult plants of Campanula species. Plant Cell Rep 27:1713–1720
Taneja J, Jaggi M, Wankhede D, Sinha A (2010) Effect of loss of T-DNA genes on MIA biosynthetic pathway gene regulation and alkaloid accumulation in Catharanthus roseus hairy roots. Plant Cell Rep 29:1119–1129
Vilaine F, Casse-Delbart F (1987) Independent induction of transformed roots by the TL and TR regions of the Ri plasmid of agropine type Agrobacterium rhizogenes. Mol Gen Genet 206:17–23
Zhou M-L, Zhu X-M, Shao J-R, Wu Y-M, Tang Y-X (2012) An protocol for genetic transformation of Catharanthus roseus by Agrobacterium rhizogenes A4. Appl Biochem Biotechnol 166:1674–1684
Acknowledgements
This project was funded by the innovation consortium “Innovative Plants” through the Danish Agency for Science, Technology and Innovation. A. rhizogenes strain A4 was kindly provided by Dr. David Tepfer, Laboratoire de Biologie de la Rhizosphère, INRA, Versailles, Cédex, France. Gartneriet PKM A/S is greatly acknowledged for providing plant material. We thank Christian Hald Madsen and Kell Kristiansen for interesting discussions.
Authors’ contribution
JNH and SVW developed the transformation procedure. JNH, HL and RM designed the experiments. JNH and UBL performed the experiments and analysed the data. JNH, HL and UBL wrote the manuscript. All authors approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hegelund, J.N., Lauridsen, U.B., Wallström, S.V. et al. Transformation of Campanula by wild type Agrobacterium rhizogenes . Euphytica 213, 51 (2017). https://doi.org/10.1007/s10681-017-1845-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-017-1845-0