Abstract
It is necessary to clear the relationship between physical and vegetation factors on the processes governing dissolved ion inputs to the forest floor to estimate correctly the values of atmospheric input to the forest. This study identified the factors influencing the differences in dissolved ion inputs to the forest floor between coniferous evergreen and broad-leaved deciduous species by analyzing the phenological variations of dry deposition and canopy exchange calculated by the canopy budget model under a high-deposition site near the city of Tokyo and a low-deposition site 84 km further away. At low-deposition site, vegetation factors such as capture efficiency did not explain the differences in Na+ or Cl− dry deposition. Leaf physiological characteristics influenced the differences in the Mg2+ and Ca2+ canopy leaching values, and phenology, leaf wettability, and diffusion processes from water film into leaves influenced the differences in NH4+ and NO3− input processes between tree types. At the high-deposition site, differences in the dry deposition of Na+, SO42−, Cl−, Mg2+, Ca2+, NH4+, and NO3− between tree types were influenced by differences in capture efficiency between coniferous and broad-leaved canopies in the leafed period and by the absence of leaves in deciduous species after leaf fall. These results indicated that atmospheric deposition affected the capture efficiency of coniferous trees for dry deposition and enhanced the difference of dissolved ion inputs to the forest floor between coniferous and deciduous species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atmospheric deposition has caused a serious decline in forest health in Europe and Japan (Sase et al., 1998; Schütt & Cowling, 1985; Takamatsu et al., 2001) and the acidification of water and damage to fish and other aquatic life in lakes and rivers in Europe and North America (Baker, 1991; Rodhe, 1972). However, emissions of SO2 have decreased due to regulation and controls implemented in the 1970s (Smith et al., 2011), and emissions of NOx have decreased since the 1990s in Europe and since the 2000s in North America and Japan (Akimoto, 2003; EPA, 2022; Kopáček & Posch, 2011). Initial signs of the recovery from forest soil acidification, lake and river acidification, and biotic diversity have been reported in those regions due to the recovery of atmospheric conditions (Eimers et al., 2004; McHale et al., 2017; Sase et al., 2019; Wright et al., 2006). Therefore, correct and wide area estimation for values of atmospheric input to the forest is important to evaluate the recovery potential of the forest by reducing atmospheric deposition.
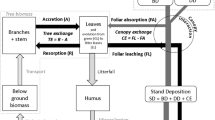
Stand deposition (SD) to the forest floor represents the total deposition supplied by throughfall (TF) and stemflow (SF). SD can be classified as wet deposition (WD; mainly precipitation), dry deposition (DD; gases and particles during dry periods), and canopy exchange (CE; canopy leaching or uptake) (Parker, 1983). WD is impacted by the strength and proximity of emission sources and meteorological factors, such as precipitation and wind speed. DD is affected by emission sources, meteorological factors, and vegetation factors such as the canopy’s ability to capture pollutants (Andersen & Hovmand, 1999; Lovett et al., 1996). CE encompasses the exchange of gases, such as nitrogen compounds (e.g., NH3, NO, and NO2), SO2, HNO3, and dissolved compounds (e.g., SO42−, NO3−, NH4+). The CE of gases occurs mainly via the stomata of vegetation (Sparks, 2009) and is controlled by stomatal conductance and gas concentrations. The CE of dissolved compounds is controlled by ion concentrations in leaves and precipitation characteristics (Lovett et al., 1996; Parker, 1983). Thus, the input of dissolved ions to the forest floor is influenced by both physical factors (e.g., the strength and proximity of emission sources and meteorological conditions) and vegetation factors (e.g., the canopy form and leaf ion concentrations). Understanding the influence of these physical and vegetation factors on dissolved ion input processes is necessary to clarify the differences in inputs to forests according to composition.
Physical factors can be investigated by comparing deposition trends in stands of the same species among different areas. For example, Imamura et al. (2020a) explained how physical factors (e.g., the distance from the center of Tokyo and elevation) affected the DD process in forests by comparing the WD and DD around Japanese cedar trees (Cryptomeria japonica (L.f.) D.Don) at seven sites within the Tokyo Metropolis. By contrast, to clarify vegetation factors, researchers have compared seasonal changes in DD and CE between coniferous and deciduous species at neighboring or nearby locations. Studies have performed seasonal comparisons of deciduous forests dominated by sugar maple (Acer saccharum Marsh.) and coniferous forests dominated by white pine (Pinus strobus L.) in Ontario, Canada (Neary & Gizyn, 1994); deciduous forests dominated by sugar maple and coniferous forests dominated by balsam fir (Abies balsamea (L.) Mill.) in Quebec, Canada (Houle et al., 1999); and deciduous forests dominated by silver birch (Betula pendula Roth) and coniferous forests dominated by Corsican pine (Pinus nigra ssp. laricio Maire) in Merksplas, Belgium (De Schrijver et al., 2004; Table 1). From these previous studies, the influence of vegetation factors (e.g., leaf capture efficiency, physiological activity, and leafless period in deciduous species) on the processes governing dissolved element inputs to the forest floor could be investigated by comparing seasonality between coniferous and deciduous species at neighboring locations. However, reported vegetation factors differed among previous studies.
De Schrijver et al. (2007) summarized the differences in annual SD between coniferous and deciduous stands reported in 38 case studies of sites with different atmospheric conditions. They found that, among stands, the Na+ SD increased with open field deposition; there were no relationships of the variations in the SDs of K+, Ca2+, and Mg2+ with open field deposition; moreover, the NH4+ SD had a stronger correlation with open field deposition than did the NO3− SD (De Schrijver et al., 2007). From these results, we hypothesized that open field deposition, which is driven by physical factors, also affects the differences in DD and CE between coniferous and deciduous species, which are influenced by vegetation factors. The difference in atmospheric deposition could affect the different results about vegetation factors among previous studies. The present study aimed to identify factors influencing the differences in annual SDs between coniferous evergreen and broad-leaved deciduous species by comparing DD and CE between species during different phenological phases at sites that experience high and low atmospheric deposition values.
Materials and methods
Site description
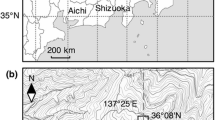
We established forest sites in Kanto, Japan, experiencing either lower atmospheric deposition (Chichibu) or higher atmospheric deposition (Tanashi) (Fig. 1 and Table 1). The Chichibu site was located at The University of Tokyo Chichibu Forest, 83.7 km from the center of Tokyo (Imamura et al., 2020a) and about 100 km from Tokyo Bay. The site hosts a natural deciduous mixed forest (35°56′N, 138°48′E; 1264 m a.s.l.) and coniferous Japanese cedar (C. japonica) plantation (35°56′N, 138°49′E; 1040 m a.s.l.) (Fig. 1) (Imamura et al., 2020b). The deciduous mixed forest is dominated by Japanese beech (Fagus japonica Maxim.), Siebold’s beech (Fagus crenata Blume), and Hemlock fir (Tsuga sieboldii Carrière). The Tanashi site was established in The University of Tokyo Tanashi Forest (35°44′N, 139°32′E) (Fig. 1) (Shi et al., 2014). The site is 14.3 km from the center of Tokyo and 22.6 km from Tokyo Bay (Imamura et al., 2020a). The present study focused on naturally seriated secondary deciduous stands of Sawtooth oak (Quercus acutissima Carruth.) and Japanese cedar (C. japonica) evergreen plantations at the Tanashi site.
Precipitation, throughfall, and stemflow measurements
Wet-only precipitation was collected using an automatic wet-only sampler at the First Nursery meteorological station at Tanashi (Imamura et al., 2018). Bulk precipitation was collected using a bulk sampler at the Koakasawa meteorological station at Chichibu and at the meteorological station at Tanashi. At Chichibu, six throughfall collectors arranged in two parallel lines of three samplers each were installed 1 m from broad-leaved deciduous species (F. crenata) (Fig. 2) (Imamura et al., 2012). Under the stand of coniferous evergreen species (C. japonica), throughfall samplers were placed at random, with six per site. Water samples were collected from the same three collectors to take into account the distance from the trunks (Fig. 2). SF was collected at each study plot. At Tanashi, under a broad-leaved deciduous (Q. acutissima) tree and C. japonica tree, TF samples were collected using five bulk samplers (Fig. 2). F. crenata and Q. acutissima belong to Fagaceae (the Beech family). Water samples were collected from all collectors. SF collectors were installed in three individuals of each tree type. Bulk precipitation, TF, and SF were measured and sampled at least once a month from October 2010 to September 2012 at Chichibu and Tanashi. Stemflow volumes were calculated by dividing the volume of collected water by the canopy projection area at each site. Table 1 summarizes the study plot characteristics, including tree age, tree height, basal area, and leaf area index (LAI).
Canopy projection and locations of bulk throughfall samplers at the observation sites at (a) Chichibu and (b) Tanashi. Individual Fagus crenata, Quercus acutissima, and Cryptomeria japonica trees are denoted as Fc, Qa, and Cj, respectively. Stemflow water was collected from Cj1, Cj2, and Cj3 and from Qa1, Qa2, and Qa3, respectively, at Tanashi
The pH and electrical conductivity of the samples were measured during each sampling period. The concentrations of dissolved inorganic ions (Na+, Cl−, SO42−, K+, Mg2+, Ca2+, NH4+, and NO3−) were measured using ion chromatography (LC-10A; Shimadzu Corp., Kyoto, Japan) and flame emission spectrometry (Z-2310; Hitachi High-Tech Science Corp., Tokyo, Japan). For quality control, each sample was assessed by comparing the measured and calculated conductivities of the water samples and the ion balances.
Meteorological and air quality measurements
Meteorological observations of air temperature and relative humidity were conducted atop a 23-m-high meteorological tower at the deciduous forest site in Chichibu and a 26-m-high meteorological tower at the C. japonica plantation in Tanashi. Air temperature and relative humidity were measured using a thermo-hygrograph at Chichibu (CS500; Campbell Scientific, Inc., Logan, USA) and Tanashi (Hobo U23-002; Onset Computer Corp., Bourne, MA, USA).
NO2 and NOx (NO plus NO2) filter samples were collected monthly from July 26, 2011, to July 5, 2012, on the meteorological tower in Chichibu using a passive sampler (OG-SN-S; Ogawa & Co., Ltd, Kobe, Japan). NO was calculated by subtraction. Water extracted from the exposed filter and blank filter was analyzed using the Saltzman method with a spectrophotometer (U-1800; Hitachi High-Tech Corporation, Tokyo, Japan). The limit of quantitation (LOQ) was calculated as ten times the standard deviation of the blanks (MacDougall et al., 1980). At Tanashi, these concentrations were obtained from national monitoring data obtained from July 14, 2011, to July 3, 2012, at the nearest monitoring site (located 1.2 km from Tanashi) (National Institute for Environmental Studies, 2022).
Estimation of dry deposition and canopy exchange
The TF and SF volumes were averaged across all collector samples at each site. The volume-weighted mean ion concentrations in TF and SF were then calculated. TF and SF depositions (mmol m−2) were calculated using the above datasets. During periods without observations, WD was calculated using bulk deposition and the bulk:wet-only concentration ratio at Tanashi (Imamura et al., 2018). Bulk deposition was used as a proxy for WD at Chichibu during all sampling periods. DD and CE were calculated with the canopy budget model (CBM) using WD, TF, and SF (Adriaenssens et al., 2013; Staelens et al., 2008). The DDs of K+, Mg2+, and Ca2+ were estimated assuming that aerosols containing K+, Mg2+, and Ca2+ were deposited onto the forest canopy at a rate equal to that of particulate Na+. Using the net throughfall (NTF):WD ratio of the Na+ tracer ion, the DD rates of K+, Mg2+, and Ca2+ were calculated as follows:
where X is K+, Mg2+, or Ca2+.
The CL values of K+, Mg2+, and Ca2+ were calculated by subtracting DD from NTF. The CL of basic cations (BC; K+ + Mg2+ + Ca2+) should equal the CU of H+ (Cronan & Reiners, 1983) and NH4+ (Roelofs et al., 1985) based on the ion charge balance of the canopy (Eq. 2.2):
H+ has an exchange capacity six times larger than that of NH4+ (Draaijers et al., 1998), which is accounted for by the relative uptake efficiency factor (xH = 6) in Eq. 2.3 to calculate the CU of NH4+ (De Schrijver et al., 2004):
The CU of H+ was calculated by subtracting NH4 from CUNH4+H. The DDs of NH4+ and H+ were calculated by subtracting CU from NTF.
The CU of NO3− was calculated based on the TF fluxes of NH4+ and NO3−, using an efficiency factor of NH4+ versus NO3− uptake (xNH4) with a value of six (de Vries et al., 2001; Eq. 2.4).
Na+, Cl−, and SO42− were defined only in terms of DD, i.e., not in terms of CL or CU.
Definition of seasons
At Chichibu, long-term video data indicate that the leaf-out period for the F. crenata canopy lasts from May to November (Fujiwara & Saito, 2005); these 7 months were considered the growing season, and the remaining 5 months were considered the dormant season (Fig. 3). At Tanashi, the leaf-out period for Q. acutissima lasted from April until November based on visual observations; these 8 months were defined as the growing season, and the remaining 4 months were considered the dormant season (Fig. 3).
Phenological periods at (a) Chichibu and (b) Tanashi. The leaf emergence period lasted 1 month (May at Chichibu and April at Tanashi). The fully leafed period was 4 months (June–September) at Chichibu and 5 months (May–September) at Tanashi. The leaf senescence period was 2 months (October–November) at both sites. The leafless period was 5 months (December–April) at Chichibu and 4 months (December–March) at Tanashi
The 2-year average annual DD and CE values were separated into four phenological phases (Staelens et al., 2007; Van Stan et al., 2012). The leaf emergence and leaf senescence periods were defined as the first and last 2 months of the growing season, respectively, for all species (Fig. 3). The other months of the growing season were defined as the fully leafed period. The leafless period was equal to the duration of the dormant season (Fig. 3).
Data analysis
To elucidate the differences in annual SDs between coniferous and deciduous species, annual SDs between coniferous and deciduous stands were compared at original study sites (Chichibu and Tanashi) and previous study sites (Ontario; Neary & Gizyn, 1994, Quebec; Houle et al., 1999, Merksplas; De Schrijver et al., 2004). Previous studies have performed seasonal comparisons of deciduous and coniferous forests. A low atmospheric deposition site was defined as a lower wet deposition site compared to that at Chichibu. By contrast, a higher atmospheric deposition site was defined as a higher wet deposition site compared to that at Tanashi. DD and CE in annual, growing, and dormant seasons were calculated by the CBM at Chichibu, Tanashi, Ontario, Quebec, and Merksplas. In addition, only at Chichibu and Tanashi, monthly averaged DD and CE were calculated by the same method in the leaf emergence, fully leafed, leaf senescence, and leafless periods. A paired t-test was used to test the significant difference in annual and seasonal SD, DD, and CE between coniferous and deciduous stands.
Results
Meteorology and air quality
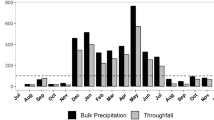
Figure 4 presents the seasonal variations in meteorological parameters of the canopies at Chichibu and Tanashi from October 2010 to September 2012. The annual mean humidity was 79% and 70% at Chichibu and Tanashi, respectively. The relative humidity was higher at Chichibu than Tanashi throughout the year, particularly in summer when the relative humidity at Chichibu exceeded 80% from June to September.
At Chichibu, the average absorption of the field blank for NO2 and NOx was 0.16 (n = 4) and 0.21 (n = 4), respectively. The LOQ of absorption was 0.02 and 0.57 for NO2 and NOx. Whereas all values of absorption for NO2 were above the LOQ, those for NOx were below LOQ. Therefore, the average NO and NO2 concentrations were below the LOQ and 1.2 ppb, respectively, at Chichibu. At Tanashi, the average NO and NO2 concentrations were 5.5 ppb and 16.6 ppb, respectively. The NO2 concentrations were > 10 times higher at Tanashi than Chichibu.
Differences in stand deposition between deciduous and coniferous species
At Chichibu, there were no differences in the 2-year average annual SD and DD values for Na+ (p = 0.05) and SO42− (p = 0.61) between deciduous and coniferous species (Tables 2 and 3). By contrast, the average annual SD and DD values of Cl− were significantly higher for coniferous than deciduous species (p < 0.05) (Tables 2 and 3). At Tanashi, the average annual SD and DD values of Na+, Cl−, and SO42− were significantly higher for coniferous than deciduous species (p < 0.001) (Tables 2 and 3). At Chichibu, the DD values were significantly higher for coniferous than deciduous species for Na+ and Cl− in the leaf senescence period (p < 0.05) and Cl− and SO42− in the leafless period (p < 0.05) (Fig. 5). The SO42− DD value was significantly higher for deciduous than coniferous species in the fully leafed and leaf senescence periods (p < 0.05) (Fig. 5). At Tanashi, the DD values were significantly higher for coniferous than deciduous species during all phenological periods except the leaf senescence period for Na+ (p < 0.05) and Cl− (p < 0.01) and the fully leafed and leafless periods for SO42− (p < 0.01) (Fig. 5).
The 2-year average dry deposition values (mmol m−2 month−1) of Na+, Cl−, SO42−, K+, Mg2+, Ca2+, H+, NH4+, and NO3− during each canopy phenological phase for deciduous species and coniferous species (Cryptomeria japonica) at Chichibu and Tanashi. The leaf emergence, fully leafed, leaf senescence, and leafless periods are defined in Fig. 3. *A paired t-test, p < 0.05, **t-test, p < 0.01, ***t-test, p < 0.001
The annual K+ SD value was non-significantly higher for deciduous than coniferous species at Chichibu (p = 0.50) and significantly at Tanashi (p < 0.01) (Table 2). The annual Mg2+ SD value did not differ between deciduous and coniferous species at Chichibu (p = 0.09) but was significantly higher for coniferous than deciduous species at Tanashi (p < 0.01) (Table 2). The annual SD value of Ca2+ was significantly higher for coniferous than deciduous species at both Chichibu and Tanashi (p < 0.01) (Table 2). At Chichibu, the annual CL values of K+, Mg2+, and Ca2+ were > 20 times higher than the DD values for both coniferous and deciduous species (Tables 3 and 4). At Tanashi, the annual K+ DD value was about one-tenth the CL value for coniferous species, and the Mg2+ and Ca2+ DD values were about half the respective CL values (Tables 3 and 4). The Mg2+ CL value was significantly higher for deciduous than coniferous species in the leaf emergence period at Chichibu (p < 0.01) (Fig. 6). At Tanashi, the CL values of K+ (p < 0.01) and Mg2+ (p < 0.05) were significantly higher for deciduous than coniferous species in the leaf emergence and fully leafed periods (Fig. 6). By contrast, the Mg2+ CL value was significantly higher for coniferous than deciduous species in the leafless period at Chichibu (p < 0.05) and Tanashi (p < 0.001) (Fig. 6). The Ca2+ CL value was significantly higher for coniferous than deciduous species in the fully leafed, leaf senescence, and leafless periods at Chichibu (p < 0.05) and in the leaf emergence and leafless periods at Tanashi (p < 0.05) (Fig. 6). Coniferous species showed significantly higher DD values of Mg2+ (p < 0.01) and Ca2+ (p < 0.01) than deciduous species during all phenological periods except the leaf senescence period at Tanashi (Fig. 5).
The 2-year average canopy leaching values (mmol m−2 month−1) of K+, Mg2+ and Ca2+ during each canopy phenological phase for deciduous species and coniferous species (Cryptomeria japonica) at Chichibu and Tanashi. The leaf emergence, fully leafed, leaf senescence, and leafless periods are defined in Fig. 3. * A paired t-test, p < 0.05, **t-test, p < 0.01, ***t-test, p < 0.001
The annual H+ SD value was significantly higher for coniferous than deciduous species at Chichibu and Tanashi (p < 0.001) (Table 2). At Chichibu and Tanashi, SF represented 78% and 83%, respectively, of the annual H+ SD value for coniferous species and 5% and 6%, respectively, for deciduous species (Table 2).
The annual NH4+ SD value was significantly higher for deciduous than coniferous species at Chichibu (p < 0.01) (Table 2). By contrast, coniferous species showed significantly higher annual SD values of NH4+ (p < 0.01) and NO3− (p < 0.001) than deciduous species at Tanashi (Table 2). At Chichibu, the annual NH4+ DD value was non-significantly higher than the CU value for deciduous species (p = 0.54) (Tables 3 and 5). At Tanashi, the annual DD values of NH4+ and NO3− were significantly higher than the respective CU values for coniferous and deciduous species (p < 0.001 and p < 0.05, respectively), and the DD values were significantly higher for coniferous than deciduous species (p < 0.01 and p < 0.001, respectively) (Tables 3 and 5). The NH4+ CU value was significantly higher for deciduous than coniferous species in the leaf emergence and fully leafed periods at Chichibu and Tanashi (p < 0.05) (Fig. 7). The NH4+ DD value was significantly higher for deciduous than coniferous species in the leaf emergence, fully leafed, and leaf senescence periods at Chichibu (p < 0.05) (Fig. 5). By contrast, the NH4+ DD value was significantly higher for coniferous than deciduous species in the fully leafed and leafless periods at Tanashi (p < 0.01), while the NH4+ DD value was significantly higher for deciduous than coniferous species in the leaf emergence period (p < 0.01) (Fig. 5). In addition, at Chichibu, the NO3− DD and CU values were significantly higher for deciduous than coniferous species in the leaf emergence period (p < 0.05) (Figs. 5 and 7). At Tanashi, the NO3− DD value was higher for coniferous than deciduous species during all phenological periods except the leaf senescence period (p < 0.01) (Fig. 5).
The 2-year average canopy uptake values (mmol m−2 month−1) of H+, NH4+, and NO3− during each canopy phenological phase for deciduous species and coniferous species (Cryptomeria japonica) at Chichibu and Tanashi. The leaf emergence, fully leafed, leaf senescence, and leafless periods are defined in Fig. 3. * A paired t-test, p < 0.05, **t-test, p < 0.01, ***t-test, p < 0.001
Discussion
Sodium and chloride ions
At the low-atmospheric-deposition site (Chichibu), there were no differences in the annual Na+ DD or SD values between coniferous and deciduous species (Tables 2 and 3) because significant differences were observed only in the leaf senescence period (Fig. 5). Similarly, in Ontario, Canada, where the Na+ WD value was lower than that at Chichibu (Table 1), the annual SD value did not differ significantly between species (Neary & Gizyn, 1994) (Table 2) because the DD values did not differ between species in either the growing or dormant seasons (Table 3). In addition, at a low-atmospheric-deposition site in Quebec, Canada (Table 1), the Na+ SD and DD values were similar between coniferous and deciduous species (Houle et al., 1999) (Tables 2 and 3). These results suggest that differences in the annual SD values of Na+ between coniferous and deciduous species are minimally impacted by vegetation factors such as capture efficiency at low-deposition sites.
The annual Cl− SD value was higher for coniferous than deciduous species at Chichibu (Table 2). This resulted from the significantly higher Cl− DD value for coniferous than deciduous species during the leaf senescence and leafless periods (Fig. 5), which could be influenced by HCl gas concentrations at Chichibu. In Ontario and Quebec, where the Cl− WD values were lower than that at Chichibu (Table 1), there were no significant differences in annual Cl− SD values between species (Houle et al., 1999; Neary & Gizyn, 1994) (Table 2) because there were no differences in the DD values between species in the growing and dormant seasons (Table 3). Similar to Na+, the differences in annual Cl− SD values between coniferous and deciduous species are minimally influenced by vegetation factors such as capture efficiency at low-deposition sites.
At the high-atmospheric-deposition site (Tanashi), the annual Na+ and Cl− SD values were significantly higher for coniferous than deciduous species (Table 2) as a result of the higher DD values for the former species in the fully leafed and leafless periods (Fig. 5). In Merksplas, Belgium, where the Na+ WD value was higher than that at Tanashi (Table 1), the annual Na+ SD value was significantly higher for coniferous than deciduous species (De Schrijver et al., 2004) (Table 2) because the DD value was significantly higher for the former species in both the growing and dormant seasons (Table 3). Vegetation factors impacting DD include tree height (Griffith et al., 2015; Lovett & Reiners, 1986), LAI (Augusto et al., 2002), basal area (Zhang et al., 2022), vegetation type (evergreen vs. deciduous) (Erisman & Draaijers, 2003), and leaf shape (Woodcock, 1953). Tree height was higher for deciduous than coniferous species, and the difference in LAI was small between species at Tanashi and Quebec (Table 1). These results suggest that higher stand capture efficiency based on wide basal area and spiral leaf shape of coniferous canopies drive the differences in Na+ and Cl− SD values between species in the growing (i.e., leafed) season. In the dormant season, the absence of leaves on deciduous species and differences in tree basal area drive the differences in the annual Na+ and Cl− SD values between deciduous and coniferous species.
Potassium, magnesium, and calcium ions
The annual K+ SD value was significantly higher for deciduous than coniferous species at Tanashi (Table 2) as a result of the low K+ DD value (Table 3) and increased K+ CL value for deciduous species in the leaf emergence and fully leafed periods (Fig. 6). Although not significant, the same trend was observed at Chichibu (Fig. 6, Tables 2 and 3). Similarly, three other studies found higher annual K+ SD values for deciduous than coniferous species because the K+ CL values were higher for deciduous than coniferous species in the growing season, even though these correlations were not significant (De Schrijver et al., 2004; Houle et al., 1999; Neary & Gizyn, 1994) (Tables 2 and 4). Similar to Chichibu and Tanashi, the previous studies also reported lower annual K+ DD than CL values (Tables 3 and 4). These trends indicate that differences in the annual K+ SD value between coniferous and deciduous species are influenced by the CL value of deciduous species in the growing season, especially the leaf emergence and fully leafed periods, even at sites with different atmospheric deposition values. In Japan, deciduous broad-leaved species generally have higher K+ levels in their leaves than C. japonica (Table 6). In addition, broad leaves are susceptible to the leaching of K+ (Rothe et al., 2002). The higher K+ CL value in deciduous than coniferous species in the leaf emergence and fully leafed periods was assumed to be due to differences in leaf K+ concentrations between deciduous and coniferous species. Therefore, differences in the K+ SD value between coniferous and deciduous species are influenced by the physiological state and leaf K+ concentration of tree species, regardless of the atmospheric deposition value.
At Chichibu, the Mg2+ CL value was higher for deciduous species in the leaf emergence period and coniferous species in the leafless period; meanwhile, the Ca2+ CL was significantly higher for coniferous than deciduous species in the fully leafed and leafless periods (Fig. 6). Moreover, the annual Mg2+ SD value did not differ between tree species, and the Ca2+ SD value was significantly higher for coniferous than deciduous species (Table 2). The same trends were observed at low-deposition sites in Ontario and Quebec, Canada (Houle et al., 1999; Neary & Gizyn, 1994) (Table 2). This resulted from the higher Mg2+ CL value for deciduous species in the growing period and coniferous species in the dormant season (Table 4). In Ontario, the Ca2+ CL value was non-significantly higher for coniferous than deciduous species in the growing or dormant season; in Quebec, the Ca2+ CL value was significantly higher for coniferous species in the dormant season (Table 4). Leaf Mg2+ and Ca2+ concentrations did not differ between deciduous species and C. japonica (Table 6); however, broad leaves are more susceptible to leaching of Mg2+ than Ca2+ (Rothe et al., 2002). The lack of differences in annual Mg2+ SD values between coniferous and deciduous species resulted from differences in the Mg2+ CL values of each species between the growing and dormant seasons at low-deposition sites. By contrast, the higher annual Ca2+ SD value for coniferous than deciduous species resulted from higher CL from coniferous than deciduous species throughout the year. These results indicate that differences in annual Mg2+ and Ca2+ SD values between coniferous and deciduous species are influenced by the physiological characteristics of leaves at low-deposition sites.
The annual Mg2+ and Ca2+ DD values were > 20-fold lower than the respective CL values at the low-deposition site, Chichibu, but were only half the respective CL values at the high-deposition site (Tanashi; Tables 3 and 4). In addition, the Mg2+ and Ca2+ DD values were higher for coniferous than deciduous species during all phenological periods, except the leaf senescence period at Tanashi (Fig. 5). These trends indicate that Mg2+ and Ca2+ DD values also influence the differences in annual Mg2+ and Ca2+ SD values between coniferous and deciduous species at high-deposition sites. At a high-deposition site in Merksplas, Belgium (Table 1), the annual Mg2+ and Ca2+ SD values were higher for coniferous than deciduous species, although not significantly (De Schrijver et al., 2004) (Table 2); the Mg2+ and Ca2+ DD values were higher than the respective CL values and were also higher for coniferous than deciduous species throughout the year (Tables 3 and 4). These findings indicate that differences in annual Mg2+ and Ca2+ SD values between coniferous and deciduous species are affected by the CL and DD values at high-deposition sites. Mg2+ and Ca2+ CL and DD values between coniferous and deciduous species are influenced by the physiological characteristics of leaves and by stand capture efficiency, respectively.
Hydrogen ion
The higher annual H+ SD for coniferous than deciduous species was the result of higher H+ SF for coniferous than deciduous species (Table 2). Generally, conifers supply high amounts of H+ via SF, due to the low pH of SF resulting from the comparatively higher dissolved organic concentrations of conifers (Inagaki et al., 1995; Parker, 1983; Thieme et al., 2019) and lower bark pH (Asplund et al., 2015) compared to deciduous species. Measurements in Ontario, Canada, revealed lower H+ SF values for conifers than in this study (Table 2) because the five most dominant species were selected for SF sampling in the former study (Table 1), which also found higher H+ SF levels for coniferous than deciduous species (Neary & Gizyn, 1994) (Table 2). Other studies conducted in Quebec, Canada, and Merksplas, Belgium, did not consider inputs of H+ from SF in the evaluation of H+ SD values, and the SD of conifers was not greater than that of deciduous species (De Schrijver et al., 2004; Houle et al., 1999) (Table 2). Overall, the findings indicate that differences in the annual H+ SD value between coniferous and deciduous species are caused by differences in H+ SF between species, regardless of atmospheric deposition values.
Sulfur, ammonium, and nitrate ions
The CU of nitrogen is controlled by leaf physiological activities (e.g., stomatal opening and photosynthesis rates) (Krupa, 2003; Lovett et al., 1996; Parker, 1983) and passive diffusion processes from water film into leaves (Hansen, 1996; Lovett et al., 1996; Schaefer et al., 1988). In addition, some studies reported canopy nitrification by using a dual isotope approach (Guerrieri et al., 2015; Templer et al., 2015; Watanabe et al., 2016). However, CBM does not account for possible nitrogen transformations occurring in tree canopies by epiphytes and/or microbes associated with foliage (Guerrieri et al., 2021). Therefore, this chapter is just focused on leaf physiological activities and passive diffusion processes for CU of nitrogen.
Birch showed the highest uptake rates in the leaf developing stage by the 15NH4+-labelled test (Adriaenssens et al., 2011). In addition, Adriaenssens et al. (2012) reported a strong negative net TF flux for NO3− in a beech canopy during the leaf development period, which was related to NO3−-N assimilation. At both Chichibu and Tanashi, we found that the NH4+ CU value was significantly higher for deciduous than coniferous species during the leaf emergence period (Fig. 7). This trend is considered the result of increased CU by leaf physiological activities of deciduous species in the leaf emergence period. In addition, water films on leaves, which form when particles are dry-deposited under relatively humid conditions, reduce cuticular resistance (Burkhardt & Eiden, 1994). Gaseous deposition of NH3, NOy, and SO2 from the atmosphere to the plant surface is increased in the presence of such films; as the gases dissolve into the water in the film, their concentrations rise, thereby enhancing passive diffusion from water film into the needle (Adriaenssens et al., 2012; Sase et al., 2008). De Schrijver et al. (2004) suggested that CU by deciduous species increases in the growing season when cuticles are thinner and wettability is greater. At the low-deposition site (Chichibu), we found that the SO42− and NH4+ DD values were significantly higher for deciduous than coniferous species in the fully leafed and leaf senescence periods (Fig. 5). The relative humidity during summer was higher at Chichibu than Tanashi because a large area around Chichibu is forested (Fig. 4). In addition, fog occurs frequently at Chichibu (Imamura et al., 2020b). Therefore, the increased SO42− and NH4+ DD values for deciduous species in the growing season are attributable to the result of the presence of water films on leaves. In addition, the NH4+ CU value for deciduous species also could be increased in the growing season via passive diffusion from water film into leaves (Fig. 7).
While the NH4+ CU value was significantly higher for deciduous than coniferous species in the leaf emergence period, the NH4+ DD value was significantly higher for deciduous than coniferous species during the growing season (Figs. 5 and 7). Therefore, the annual NH4+ SD value has been higher for deciduous than coniferous species because the annual NH4+ DD value was higher than the annual NH4+ CU value for deciduous species. The annual SO42− SD value did not differ between coniferous and deciduous species (Table 2); this was the result of a higher SO42− DD value for deciduous species in the fully leafed and leaf senescence periods and higher value for coniferous species in the leafless period (Fig. 5). At a low-deposition site in Quebec, Canada (Table 1), the significantly higher annual NH4+ SD value for deciduous than coniferous species resulted from a higher NH4+ DD value for the former species throughout the year (Houle et al., 1999) (Tables 2 and 3). Differences in the annual SO42− SD value between deciduous and coniferous species were influenced by leaf wettability, and those of NH4+ were influenced by leaf uptake (a phenological factor), as well as the DD and CU driven by leaf wettability and diffusion processes from water film into leaves at the low-deposition site.
The NO3− CU value was higher for deciduous than coniferous species in the leaf emergence period at Chichibu (Fig. 7). However, there was no difference between species in the NO3− DD value during other periods (Fig. 5). While the NH4+ DD value for deciduous species increased in fully leafed and leaf senescence periods (relatively humid conditions), the NO3− DD value did not increase during same periods at Chichibu (Fig. 5). This is because water films had no impact on the diffusion processes, possibly due to low gas concentrations (NO: below the LOQ; NO2: 1.2 ppb). In Quebec, CU of NO3− was reported in the leaf senescence period. In addition, the annual NO3− SD value was significantly higher for deciduous than coniferous species because the NO3− DD value was higher for the former species throughout the year (Houle et al., 1999) (Tables 2 and 3). The gas concentrations in 1999 in Quebec (NO: 2.3 ppb; NO2: 8.0 ppb; National Air Pollution Surveillance Program, 2022) were higher than those at Chichibu during the present study; thus, water film formation increased the DD of NO3− for deciduous species. Overall, differences in the annual NO3− SD value between deciduous and coniferous species were influenced by both phenological factors and diffusion processes, although atmospheric gas concentrations also impact diffusion.
At Tanashi, the significantly higher annual SO42− SD value for coniferous than deciduous species (Table 2) resulted from the higher SO42− DD values for coniferous than deciduous species in the fully leafed and leafless periods (Fig. 5). The NH4+ and NO3− CU values were higher in the leaf emergence period (Fig. 5), and the annual NH4+ and NO3− DD values were significantly higher than the respective CU values for both species (Tables 3 and 5). In addition, the NH4+ and NO3− DD values were higher for coniferous than deciduous species in the fully leafed and leafless periods (Fig. 5). Therefore, the annual NH4+ and NO3− SD values were significantly higher for coniferous than deciduous species (Table 2). In Ontario, Canada, where the SO42− WD value was higher than that at Tanashi (Table 1), the annual SO42− SD value was significantly higher for coniferous than deciduous species (Neary & Gizyn, 1994) (Table 2) because DD was significantly higher for coniferous than deciduous species in the growing season (Table 3). Similarly, the annual SO42− SD value was non-significantly higher for coniferous than deciduous species in Quebec (Houle et al., 1999) (Table 2) because the DD was higher for the former species in the growing and dormant seasons (Table 3). In Merksplas, Belgium, where the NH4+ and NO3− WD values were higher than those in Tanashi (Table 1), the annual NH4+ and NO3− DD values were higher than the respective CU values (Tables 3 and 5). In addition, the annual NH4+ and NO3− DD and SD values were higher for coniferous than deciduous species (De Schrijver et al., 2004) (Tables 2 and 3). These results indicate that the annual SO42−, NH4+, and NO3− SD values are higher for coniferous than deciduous species as a result of increased DD (due to a higher capture efficiency) for coniferous species in the growing season, as well as the absence of leaves on deciduous species in the dormant season at high-deposition sites.
Conclusion
This research explained the factors affecting the difference of dissolved ion inputs to the forest floor between coniferous and deciduous species by comparing seasonal variations of dry deposition and canopy exchange at two different atmospheric deposition conditions. Whereas this research is regional and based on limited observation data, this research cleared that the atmospheric deposition affected to vegetation factor, especially the capture efficiency of coniferous trees for Na+, Cl−, Mg2+, Ca2+, SO42−, NH4+, and NO3−. In contrast, atmospheric deposition had no impact on canopy leaching of K+, Mg2+, and Ca2+ and neutralization between species. This suggests that information on atmospheric depositions in the study area could be important to estimate correctly different values of dissolved ion inputs to forest floor between deciduous and coniferous forests.
Data availability
Data is supplied by supplementary file.
Change history
12 March 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10661-024-12515-3
References
Adriaenssens, S., Staelens, J., Wuyts, K., De Schrijver, A., Van Wittenberghe, S., Wuytack, T., Kardel, F., Verheyen, K., Samson, R., & Boeckx, P. (2011). Foliar nitrogen uptake from wet deposition and the relation with leaf wettability and water storage capacity. Water, Air, and Soil Pollution, 219, 43–57. https://doi.org/10.1007/s11270-010-0682-8
Adriaenssens, S., Hansen, K., Staelens, J., Wuyts, K., De Schrijver, A., Baeten, L., Boeckx, P., Samson, R., & Verheyen, K. (2012). Throughfall deposition and canopy exchange processes along a vertical gradient within the canopy of beech (Fagus sylvatica L.) and Norway spruce (Picea abies (L.) Karst). Science of the Total Environment, 420, 168–182. https://doi.org/10.1016/j.scitotenv.2011.12.029
Adriaenssens, S., Staelens, J., Baeten, L., Verstraeten, A., Boeckx, P., Samson, R., & Verheyen, K. (2013). Influence of canopy budget model approaches on atmospheric deposition estimates to forests. Biogeochemistry, 116, 215–229. https://doi.org/10.1007/s10533-013-9846-0
Akimoto, H. (2003). Global air quality and pollution. Science, 302, 1716–1719. https://doi.org/10.1126/science.1092666
Andersen, H. V., & Hovmand, M. F. (1999). Review of dry deposition measurements of ammonia and nitric acid to forest. Forest Ecology and Management, 114, 5–18. https://doi.org/10.1016/S0378-1127(98)00378-8
Asplund, J., Ohlson, M., & Gauslaa, Y. (2015). Tree species shape the elemental composition in the lichen Hypogymnia physodes transplanted to pairs of spruce and beech trunks. Fungal Ecology, 16, 1–5. https://doi.org/10.1016/j.funeco.2015.03.006
Augusto, L., Ranger, J., Binkley, D., & Rothe, A. (2002). Impact of several common tree species of European temperate forests on soil fertility. Annals of Forest Science, 59, 233–253. https://doi.org/10.1051/forest:2002020
Baker, J. P. (1991). Biological effects of changes in surface water acid-base chemistry. In P. M. Irving (Ed.), Acidic deposition: State of science and technology (pp. 119–123). The U.S. National Acid Precipitation Assessment Program.
Burkhardt, J., & Eiden, R. (1994). Thin water films on coniferous needles. A new device for the study of water vapour condensation and gaseous deposition to plant surfaces and particle samples. Atmospheric Environment, 28, 2001–2011. https://doi.org/10.1016/1352-2310(94)90469-3
Cronan, C. S., & Reiners, W. A. (1983). Canopy processing of acidic precipitation by coniferous and hardwood forests in New England. Oecologia, 59, 216–223. https://doi.org/10.1007/BF00378839
De Schrijver, A., Nachtergale, L., Staelens, J., Luyssaert, S., & De Keersmaeker, L. (2004). Comparison of throughfall and soil solution chemistry between a high-density Corsican pine stand and a naturally regenerated silver birch stand. Environmental Pollution, 131, 93–105. https://doi.org/10.1016/j.envpol.2004.01.019
de Vries, W., Reinds, G. J., van der Salm, C., Draaijers, G. P. J., Bleeker, A., Erisman, J. W., Auée, J., Gundersen, P., Kristensen, H. L., van Dobben, H., de Zwart, D., Derome, J., Voogd, J. C. H., & Vel, E. M. (2001). Intensive monitoring of forest ecosystems in Europe Technical Report 2001. In Forest Intensive Monitoring Coordinating Institute, 2001. Retrieved November 17, 2023 from https://www.icp-forests.org/pdf/TRLII2001.pdf
Draaijers, G. P. J., Erisman, J. W., Lövblad, G., Spranger, T., & Vel, E. (1998). Quality and uncertainty aspects ot torest deposition estimation using throughfall, stemflow and precipitation measurements. Retrieved July 11, 2023 from https://publications.tno.nl/publication/34641023/vuCqh6/MEP-R-1998-093.pdf
Eimers, M. C., Dillon, P. J., & Watmough, S. A. (2004). Long-term (18-year) changes in sulphate concentrations in two Ontario headwater lakes and their inflows in response to decreasing deposition and climate variations. Hydrological Processes, 18, 2617–2630. https://doi.org/10.1002/hyp.5570
EPA. (2022). Air pollutant emissions trends data. National Tier 1 CAPS Trends (xlsx). Criteria pollutants National Tier 1 for 1970 - 2021. Retrieved July 26, 2022, from https://www.epa.gov/air-emissions-inventories/air-pollutant-emissions-trends-data
Erisman, J. W., & Draaijers, G. (2003). Deposition to forests in Europe: Most important factors influencing dry deposition and models used for generalisation. Environmental Pollution, 124, 379–388. https://doi.org/10.1016/S0269-7491(03)00049-6
Fujiwara, A., & Saito, K. (2005). Analysis of long term fixed point daily video recordings using a robot camera for observing tree phenology. Journal of the Japanese Institute of Landscape Architecture, 68, 927–930. https://doi.org/10.5632/jila.68.927
Griffith, K. T., Ponette-González, A. G., Curran, L. M., & Weathers, K. C. (2015). Assessing the influence of topography and canopy structure on Douglas fir throughfall with LiDAR and empirical data in the Santa Cruz mountains, USA. Environmental Monitoring and Assessment, 187, 270. https://doi.org/10.1007/s10661-015-4486-6
Guerrieri, R., Vanguelova, E. I., Michalski, G., Heaton, T. H. E., & Mencuccini, M. (2015). Isotopic evidence for the occurrence of biological nitrification and nitrogen deposition processing in forest canopies. Global Change Biology, 21, 4613–4626. https://doi.org/10.1111/gcb.13018
Guerrieri, R., Templer, P., & Magnani, F. (2021). Canopy exchange and modification of nitrogen fluxes in forest ecosystems. Current Forestry Reports, 7, 115–137. https://doi.org/10.1007/s40725-021-00141-y
Hansen, K. (1996). In-canopy throughfall measurements of ion fluxes in Norway spruce. Atmospheric Environment, 30, 4065–4076. https://doi.org/10.1016/1352-2310(95)00444-0
Houle, D., Ouimet, R., Paquin, R., & Laflamme, J.-G. (1999). Interactions of atmospheric deposition with a mixed hardwood and a coniferous forest canopy at the Lake Clair Watershed (Duchesnay, Quebec). Canadian Journal of Forest Research, 29, 1944–1957. https://doi.org/10.1139/x99-212
Imamura, N., Tanaka, N., Ohte, N., & Yamamoto, H. (2012). Nutrient transfer with rainfall in the canopies of a broad-leaved deciduous forest in Okuchichibu. Journal of the Japanese Forestry Society, 94, 74–83. https://doi.org/10.4005/jjfs.94.74
Imamura, N., Iwai, N., Tanaka, N., & Ohte, N. (2018). A comparison between wet-only and bulk deposition at two forest sites in Japan. Asian Journal of Atmospheric Environment, 12, 67–77. https://doi.org/10.5572/ajae.2018.12.1.067
Imamura, N., Katata, G., Kajino, M., Kobayashi, M., Itoh, Y., & Akama, A. (2020b). Fogwater deposition of radiocesium in the forested mountains of East Japan during the Fukushima Daiichi Nuclear Power Plant accident: A key process in regional radioactive contamination. Atmospheric Environment, 224, 117339. https://doi.org/10.1016/j.atmosenv.2020.117339
Imamura, N., Levia, D. F., Nanko, K., Tanaka, N., & Ohte, N. (2020a). Geographic factors explain the variability of atmospheric deposition of sulfur and nitrogen onto coniferous forests within and beyond the Tokyo metropolis. Water, Air, & Soil Pollution, 231, 105. https://doi.org/10.1007/s11270-020-4467-4
Inagaki, M., Sakai, M., & Ohnuki, Y. (1995). The effects of organic carbon on acid rain in a temperate forest in Japan. Water, Air, & Soil Pollution, 85, 2345–2350. https://doi.org/10.1007/BF01186184
Kopáček, J., & Posch, M. (2011). Anthropogenic nitrogen emissions during the Holocene and their possible effects on remote ecosystems. Global Biogeochemical Cycles, 25, 1–16. https://doi.org/10.1029/2010GB003779
Krupa, S. V. (2003). Effects of atmospheric ammonia (NH3) on terrestrial vegetation: A review. Environmental Pollution, 124, 179–221. https://doi.org/10.1016/S0269-7491(02)00434-7
Lovett, G. M., & Reiners, W. A. (1986). Canopy structure and cloud water deposition in subalpine coniferous forests. Tellus, 38B, 319–327. https://doi.org/10.1111/j.1600-0889.1986.tb00257.x
Lovett, G. M., Nolan, S. S., Driscoll, C. T., & Fahey, T. J. (1996). Factors regulating throughfall flux in a New Hampshire forested landscape. Canadian Journal of Forest Research, 26, 2134–2144. https://doi.org/10.1139/x26-242
MacDougall, D., Crummett, W. B., et al. (1980). Guidelines for data acquisition and data quality evaluation in environmental chemistry. Analytical Chemistry, 52, 2242–2249. https://doi.org/10.1021/ac50064a004
McHale, M. R., Burns, D. A., Siemion, J., & Antidormi, M. R. (2017). The response of soil and stream chemistry to decreases in acid deposition in the Catskill Mountains, New York, USA. Environmental Pollution, 229, 607–620. https://doi.org/10.1016/j.envpol.2017.06.001
National Air Pollution Surveillance Program. (2022). Available from the Government of Canada Open Data Portal at open.canada.ca. Retrieved June 7, 2022 from https://open.canada.ca/data/en/dataset/1b36a356-defd-4813-acea-47bc3abd859b
National Institute for Environmental Studies. (2022). Environmental observatory data of continuous monitoring of air pollution. Retrieved June 7, 2022 from https://tenbou.nies.go.jp/download
Neary, A. J., & Gizyn, W. I. (1994). Throughfall and stemflow chemistry under deciduous and coniferous forest canopies in south central Ontario. Canadian Journal of Forest Research, 24, 1089–1100. https://doi.org/10.1139/x94-145
Parker, G. G. (1983). Throughfall and stemflow in the forest nutrient cycle. Advances in Ecological Research, 13, 57–133. https://doi.org/10.1016/S0065-2504(08)60108-7
Rodhe, H. (1972). A study of the sulfur budget for the atmosphere over Northern Europe. Tellus a: Dynamic Meteorology and Oceanography, 24, 128. https://doi.org/10.3402/tellusa.v24i2.10627
Roelofs, J. G. M., Kempers, A. J., Houdijk, A. L. F. M., & Jansen, J. (1985). The effect of air-borne ammonium sulphate on Pinus nigra var. maritima in the Netherlands. Plant and Soil, 84, 45–56. https://doi.org/10.1007/BF02197866
Rothe, A., Huber, C., Kreutzer, K., & Weis, W. (2002). Deposition and soil leaching in stands of Norway spruce and European Beech: Results from the Hoglwald research in comparison with other European case studies. Plant and Soil, 240, 33–45. https://doi.org/10.1023/A:1015846906956
Sase, H., Takamatsu, T., Yoshida, T., & Inubushi, K. (1998). Changes in properties of epicuticular wax and the related water loss in Japanese cedar (Cryptomeria japonica) affected by anthropogenic environmental factors. Canadian Journal of Forest Research, 28, 546–556. https://doi.org/10.1139/x98-021
Sase, H., Takahashi, A., Sato, M., Kobayashi, H., Nakata, M., & Totsuka, T. (2008). Seasonal variation in the atmospheric deposition of inorganic constituents and canopy interactions in a Japanese cedar forest. Environmental Pollution, 152, 1–10. https://doi.org/10.1016/j.envpol.2007.06.023
Sase, H, Takahashi, M., Matsuda, K., Sato, K., Tanikawa, T., Yamashita, N., Ohizumi, T., Ishida, T., Kamisako, M., Kobayashi, R., Uchiyama, S., Saito, T., Morohashi, M., Fukuhara, H., Kaneko, S., Inoue, T., Yamada, T., Takenaka, C., Tayasu, I., … Ohta, S. (2019). Response of river water chemistry to changing atmospheric environment and sulfur dynamics in a forested catchment in central Japan. Biogeochemistry, 142, 357–374. https://doi.org/10.1007/s10533-019-00540-1
Schaefer, D. A., Reiners, W. A., & Olson, R. K. (1988). Factors controlling the chemical alteration of throughfall in a subalpine balsam fir canopy. Environmental and Experimental Botany, 28, 175–189. https://doi.org/10.1016/0098-8472(88)90027-5
Schrijver, A. D., Geudens, G., Augusto, L., Staelens, J., Mertens, J., Wuyts, K., Gielis, L., & Verheyen, K. (2007). The effect of forest type on throughfall deposition and seepage flux: A review. Oecologia, 153, 663–674. https://doi.org/10.1007/s00442-007-0776-1
Schütt, P., & Cowling, E. (1985). Waldsterben, a general decline of forests in central Europe: Symptoms, deveropment, and possible causes. Plant Disease, 69, 548–558.
Shi, J., Ohte, N., Tokuchi, N., Imamura, N., Nagayama, M., Oda, T., & Suzuki, M. (2014). Nitrate isotopic composition reveals nitrogen deposition and transformation dynamics along the canopy-soil continuum of a suburban forest in Japan. Rapid Communications in Mass Spectrometry, 28, 2539–2549. https://doi.org/10.1002/rcm.7050
Smith, S. J., Van Aardenne, J., Klimont, Z., Andres, R. J., Volke, A., & Delgado Arias, S. (2011). Anthropogenic sulfur dioxide emissions: 1850–2005. Atmospheric Chemistry and Physics, 11, 1101–1116. https://doi.org/10.5194/acp-11-1101-2011
Sparks, J. P. (2009). Ecological ramifications of the direct foliar uptake of nitrogen. Oecologia, 159, 1–13. https://doi.org/10.1007/s00442-008-1188-6
Staelens, J., De Schrijver, A., & Verheyen, K. (2007). Seasonal variation in throughfall and stemflow chemistry beneath a European beech (Fagus sylvatica) tree in relation to canopy phenology. Canadian Journal of Forest Research, 37, 1359–1372. https://doi.org/10.1139/X07-003
Staelens, J., Houle, D., De Schrijver, A., Neirynck, J., & Verheyen, K. (2008). Calculating dry deposition and canopy exchange with the canopy budget model: Review of assumptions and application to two deciduous forests. Water, Air, and Soil Pollution, 191, 149–169. https://doi.org/10.1007/s11270-008-9614-2
Takamatsu, T., Sase, H., & Takada, J. (2001). Some physiological properties of Cryptomeria japonica leaves from Kanto, Japan: Potential factors causing tree decline. Canadian Journal of Forest Research, 31, 663–672. https://doi.org/10.1139/cjfr-31-4-663
Templer, P. H., Weathers, K. C., Lindsey, A., Lenoir, K., & Scott, L. (2015). Atmospheric inputs and nitrogen saturation status in and adjacent to Class I wilderness areas of the northeastern US. Oecologia, 177, 5–15. https://doi.org/10.1007/s00442-014-3121-5
Thieme, L., Graeber, D., Hofmann, D., Bischoff, S., Schwarz, M. T., Steffen, B., Meyer, U. N., Kaupenjohann, M., Wilcke, W., Michalzik, B., & Siemens, J. (2019). Dissolved organic matter characteristics of deciduous and coniferous forests with variable management: Different at the source, aligned in the soil. Biogeosciences, 16, 1411–1432. https://doi.org/10.5194/bg-16-1411-2019
Van Stan, J. T., Levia, D. F., Inamdar, S. P., Lepori-Bui, M., & Mitchell, M. J. (2012). The effects of phenoseason and storm characteristics on throughfall solute washoff and leaching dynamics from a temperate deciduous forest canopy. Science of the Total Environment, 430, 48–58. https://doi.org/10.1016/j.scitotenv.2012.04.060
Watanabe, K., Kohzu, A., Suda, W., Yamamura, S., Takamatsu, T., Takenaka, A., Koshikawa, M. K., Hayashi, S., & Watanabe, M. (2016). Microbial nitrification in throughfall of a Japanese cedar associated with archaea from the tree canopy. SpringerPlus, 5, 1596. https://doi.org/10.1186/s40064-016-3286-y
Woodcock, A. H. (1953). Salt nuclei in marine air as a function of altitude and wind force. Journal of Meteorology, 10, 362–371. https://doi.org/10.1175/1520-0469(1953)010%3c0366:SNIMAA%3e2.0.CO;2
Wright, R. F., Aherne, J., Bishop, K., Camarero, L., Cosby, B. J., Erlandsson, M., Evans, C. D., Forsius, M., Hardekopf, D. W., Helliwell, R., Hruška, J., Jenkins, A., Kopáček, J., Moldan, F., Posch, M., & Rogora, M. (2006). Modelling the effect of climate change on recovery of acidified freshwaters: Relative sensitivity of individual processes in the MAGIC model. Science of the Total Environment, 365, 154–166. https://doi.org/10.1016/j.scitotenv.2006.02.042
Zhang, S., De Frenne, P., Landuyt, D., & Verheyen, K. (2022). Impact of tree species diversity on throughfall deposition in a young temperate forest plantation. Science of the Total Environment, 842, 156947. https://doi.org/10.1016/j.scitotenv.2022.156947
Acknowledgements
We gratefully acknowledge suggestions from Dr. Masakazu Suzuki (Emeritus Professor, The University of Tokyo). The authors thank to technical staffs in The University of Tokyo Chichibu forest and Tanashi forest for supporting observation works. The authors also wish to thank Ms. Chiai Kosaku and Dr. Jun Shi for their support in field observation at Tanashi.
Funding
This work was supported by the Japan Society for the Promotion of Science: JSPS KAKENHI (grant nos. 22780139, 24658133, and 20K15563).
Author information
Authors and Affiliations
Contributions
Conceptualization: Naohiro Imamura, and Nobuhito Ohte; methodology: Naohiro Imamura, Nobuhito Ohte, and Nobuaki Tanaka.; formal analysis and investigation: Naohiro Imamura, Nobuhito Ohte, and Nobuaki Tanaka.; writing—original draft preparation: Naohiro Imamura; writing—review and editing: Naohiro Imamura, Nobuhito Ohte, and Nobuaki Tanaka; funding acquisition: Naohiro Imamura, Nobuhito Ohte, and Nobuaki Tanaka.
Corresponding author
Ethics declarations
Ethics approval
All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Imamura, N., Ohte, N. & Tanaka, N. Factors influencing the difference in dissolved ion inputs to the forest floor between deciduous and coniferous stands: comparison under high and low atmospheric deposition conditions. Environ Monit Assess 196, 1 (2024). https://doi.org/10.1007/s10661-023-12132-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-12132-6