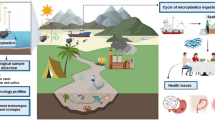
Abstract
Owing to their widespread distribution and high bioaccumulation, microplastics (MPs) and mercury (Hg) are considered major threats to the ocean. MP interacts with Hg because of its high adsorption properties. However, their toxicological interactions with marine organisms, especially combined effects at the molecular level, are poorly understood. This study investigated the single and combined effects of MP and Hg on the metabolic profile of the brackish water flea Diaphanosoma celebensis. A total of 238 metabolites were significantly affected by MP, Hg, or MP + Hg. Metabolite perturbation patterns showed that toxicity of Hg and MP + Hg was similar and that of MP was not significant. Among the 223 metabolites affected by Hg, profiles of 32 unannotated metabolites were significantly different from those of MP + Hg, and combined effects of MP + Hg decreased the effect of Hg on 25 of these metabolites. Only 11 annotated metabolites were significantly affected by Hg or MP + Hg and were related to carbohydrate, lipid, vitamin, and ecdysteroid metabolism. Ten metabolites were decreased by Hg and MP + Hg and were not significantly different between the exposure groups. Enrichment analysis showed that galactose, starch, and sucrose metabolism were the most affected pathways. These findings suggest that MP has negligible toxic effect, and Hg can induce energy depletion, membrane damage, and disruption of growth, development, and reproduction. Although the impact of MP was negligible, the combined effects of MP + Hg could be metabolite specific. This study provides better understanding of the combined effects of MP and Hg on marine organisms.





Similar content being viewed by others
References
Ašmonaitė, G., Tivefälth, M., Westberg, E., Magnér, J., Backhaus, T., & Carney Almroth, B. (2020). Microplastics as a Vector for exposure to hydrophobic organic chemicals in fish: a comparison of two polymers and silica particles spiked with three model compounds. Frontiers in Environmental Science, 8, 87. https://doi.org/10.3389/fenvs.2020.00087
Bae, C., & Lee, Y. M. (2018). Antioxidant responses in brackish water flea Diaphanosoma celebensis - Exposed to Mercury. Journal of Marine Life Science, 3(2), 74–80.
Barboza, L. G. A., Vieira, L. R., Branco, V., Carvalho, C., & Guilhermino, L. (2018). Microplastics increase mercury bioconcentration in gills and bioaccumulation in the liver, and cause oxidative stress and damage in Dicentrarchus labrax juveniles. Scientific Reports. https://doi.org/10.1038/s41598-018-34125-z
Bartonitz, A., Anyanwu, I. N., Geist, J., Imhof, H. K., Reichel, J., Graßmann, J., Drewes, J. E., & Beggel, S. (2020). Modulation of PAH toxicity on the freshwater organism G. roeseli by microparticles. Environmental Pollution., 260, 113999. https://doi.org/10.1016/j.envpol.2020.113999
Bell, M. V., & Tocher, D. R. (2009). Biosynthesis of Polyunsaturated Fatty Acids in Aquatic Ecosystems: General Pathways and New Directions. In M. Kainz, M. T. Brett, & M. T. Arts (Eds.), Lipids in Aquatic Ecosystems (pp. 211–236). Springer New York. https://doi.org/10.1007/978-0-387-89366-2_9
Beltrame, M. O., De Marco, S. G., & Marcovecchio, J. E. (2010). Effects of zinc on molting and body weight of the estuarine crab Neohelice granulata (Brachyura: Varunidae). Science of the Total Environment, 408, 531–536. https://doi.org/10.1016/j.scitotenv.2009.09.051
Bhagat, J., Nishimura, N., & Shimada, Y. (2021). Toxicological interactions of microplastics/nanoplastics and environmental contaminants: Current Knowledge and Future Perspectives. Journal of Hazardous Materials, 405, 123913. https://doi.org/10.1016/j.jhazmat.2020.123913
Birch, Q. T., Potter, P. M., Pinto, P. X., Dionysiou, D. D., & Al-Abed, S. R. (2020). Sources, transport, measurement and impact of nano and microplastics in urban watersheds. Reviews in Environmental Science and Bio/technology, 19, 275–336. https://doi.org/10.1007/s11157-020-09529-x
Brennecke, D., Duarte, B., Paiva, F., Caçador, I., & Canning-Clode, J. (2016). Microplastics as vector for heavy metal contamination from the marine environment. Estuarine, Coastal and Shelf Science, 178, 189–195. https://doi.org/10.1016/j.ecss.2015.12.003
Cappello, T., Brandão, F., Guilherme, S., Santos, M. A., Maisano, M., Mauceri, A., Canário, J., Pacheco, M., & Pereira, P. (2016). Insights into the mechanisms underlying mercury-induced oxidative stress in gills of wild fish (Liza aurata) combining 1H NMR metabolomics and conventional biochemical assays. Science of the Total Environment, 548–549, 13–24. https://doi.org/10.1016/j.scitotenv.2016.01.008
Cole, M., Lindeque, P., Halsband, C., & Galloway, T. S. (2011). Microplastics as contaminants in the marine environment: A review. Marine Pollution Bulletin, 62(12), 2588–2597. https://doi.org/10.1016/j.marpolbul.2011.09.025
De Coen, W. M., Janssen, C. R., & Segner, H. (2001). The use of biomarkers in Daphnia magna toxicity testing V. In vivo alterations in the carbohydrate metabolism of Daphnia magna exposed to sublethal concentrations of mercury and lindane. Ecotoxicology and Environmental Safety, 48(3), 223–234. https://doi.org/10.1006/eesa.2000.2009
Derraik, J. G. B. (2002). The pollution of the marine environment by plastic debris: A review. Marine Pollution Bulletin, 44(9), 842–852. https://doi.org/10.1016/s0025-326x(02)00220-5
Frat, L., Chertemps, T., Pesce, E., Bozzolan, F., Dacher, M., Planelló, R., Herrero, O., Llorente, L., & Siaussat, D. (2021). Single and mixed exposure to cadmium and mercury in Drosophila melanogaster: Molecular responses and impact on post-embryonic development. Ecotoxicology and Environmental Safety, 220, 112377. https://doi.org/10.1016/j.ecoenv.2021.112377
Gil-Solsona, R., Gil-Solsona, R., Nácher-Mestre, J., Lacalle-Bergeron, L., Vicente Sancho, J., Calduch-Giner, J. A., Hernández, F., & Pérez-Sánchez, J. (2017). Untargeted metabolomics approach for unraveling robust biomarkers of nutritional status in fasted gilthead sea bream (Sparus aurata). PeerJ, 5, e2920. https://doi.org/10.7717/peerj.2920
Gworek, B., Bemowska-Kałabun, O., Kijenska, M., & Wrzosek-Jakubowska, J. (2016). Mercury in marine and oceanic waters—a review. Water, Air, & Soil Pollution, 227(10), 371. https://doi.org/10.1007/s11270-016-3060-3
Hanau, S., Almugadam, S. H., Sapienza, E., Cacciari, B., Manfrinato, M. C., Trentini, A., & Kennedy, J. F. (2020). Schematic overview of oligosaccharides, with survey on their major physiological effects and a focus on milk ones. Carbohydrate Polymer Technologies and Applications, 1, 100013. https://doi.org/10.1016/j.carpta.2020.100013
Hishikawa, D., Valentine, W. J., Iizuka-Hishikawa, Y., Shindou, H., & Shimizu, T. (2017). Metabolism and functions of docosahexaenoic acid-containing membrane glycerophospholipids. FEBS Letters, 591(18), 2730–2744. https://doi.org/10.1002/1873-3468.12825
Hosamani, N., Reddy, S. B., & Reddy, R. P. (2017). Crustacean Molting: Regulation and Effects of Environmental Toxicants. Journal of Marine Science: Research & Development, 7, 236. https://doi.org/10.4172/2155-9910.1000236
In, S., Yoon, H.-W., Yoo, J.-W., Cho, H., Kim, R.-O., & Lee, Y.-M. (2019). Acute toxicity of bisphenol A and its structural analogues and transcriptional modulation of the ecdysone-mediated pathway in the brackish water flea Diaphanosoma celebensis. Ecotoxicology and Environmental Safety, 179, 310–317. https://doi.org/10.1016/j.ecoenv.2019.04.065
Jeong, C.-B., Kang, H.-M., Lee, M.-C., Kim, D.-H., Han, J., Hwang, D.-S., Souissi, S., Lee, S.-J., Shin, K.-H., & Lee, J.-S. (2017). Adverse effects of microplastics and oxidative stress-induced MAPK/Nrf2 pathway-mediated defense mechanisms in the marine copepod Paracyclopina nana. Scientific Reports, 7(1), 41323. https://doi.org/10.1038/srep41323
Jeong, C.-B., Won, E.-J., Kang, H.-M., Lee, M.-C., Hwang, D.-S., Hwang, U.-K., Zhou, B., Souissi, S., Lee, S.-J., & Lee, J.-S. (2016). Microplastic size-dependent toxicity, oxidative stress induction, and p-JNK and p-p38 activation in the monogonont rotifer (Brachionus koreanus). Environmental Science & Technology, 50(16), 8849–8857. https://doi.org/10.1021/acs.est.6b01441
Jeong, T.-Y., & Simpson, M. J. (2020). Reproduction stage specific dysregulation of Daphnia magna metabolites as an early indicator of reproductive endocrine disruption. Water Research, 184, 116107. https://doi.org/10.1016/j.watres.2020.116107
Johnston, P. A. (1987). Acute toxicity of inorganic selenium to Daphnia magna (Straus) and the effect of sub-acute exposure upon growth and reproduction. Aquatic Toxicology, 10(5–6), 335–352. https://doi.org/10.1016/0166-445X(87)90007-5
Kalhan, S. C., Mahajan, S., Burkett, E., Reshef, L., & Hanson, R. W. (2001). Glyceroneogenesis and the source of glycerol for hepatic triacylglycerol synthesis in humans. Journal of Biological Chemistry, 276(16), 12928–12931. https://doi.org/10.1074/jbc.m006186200
Kim, B.-M., Kang, S., Kim, R.-O., Jung, J.-H., Lee, K.-W., Rhee, J.-S., & Lee, Y.-M. (2018). De novo transcriptome assembly of brackish water flea Diaphanosoma celebensis based on short-term cadmium and benzo[a]pyrene exposure experiments. Hereditas, 155(1), 36. https://doi.org/10.1186/s41065-018-0075-3
Klumpen, E., Hoffschröer, N., Schwalb, A., Gigengack, U., Koch, M., Paul, R. J., & Zeis, B. (2021). Metabolic adjustments during starvation in Daphnia pulex. Comparative Biochemistry and Physiology Part b: Biochemistry and Molecular Biology, 255, 110591. https://doi.org/10.1016/j.cbpb.2021.110591
Kovacevic, V., Simpson, A. J., & Simpson, M. J. (2019). The concentration of dissolved organic matter impacts the metabolic response in Daphnia magna exposed to 17α-ethynylestradiol and perfluorooctane sulfonate. Ecotoxicology and Environmental Safety, 170, 468–478. https://doi.org/10.1016/j.ecoenv.2018.12.008
Landrigan, P. J., Stegeman, J. J., Fleming, L. E., Allemand, D., Anderson, D. M., Backer, L. C., Brucker-Davis, F., Chevalier, N., Corra, L., Czerucka, D., Bottein, M.-Y.D., Demeneix, B., Depledge, M., Deheyn, D. D., Dorman, C. J., Fénichel, P., Fisher, S., Gaill, F., Galgani, F., … Rampal, P. (2020). Human health and ocean pollution. Annals of Global Health, 86(1), 151. https://doi.org/10.5334/aogh.2831
Lankadurai, B. P., Nagato, E. G., & Simpson, M. J. (2013). Environmental metabolomics: An emerging approach to study organism responses to environmental stressors. Environmental Reviews, 21(3), 180–205. https://doi.org/10.1139/er-2013-0011
Lee, Y. H., Kim, D.-H., Kang, H.-M., Wang, M., Jeong, C.-B., & Lee, J.-S. (2017). Adverse effects of methylmercury (MeHg) on life parameters, antioxidant systems, and MAPK signaling pathways in the rotifer Brachionus koreanus and the copepod Paracyclopina nana. Aquatic Toxicology, 190, 181–189. https://doi.org/10.1016/j.aquatox.2017.07.006
Lu, K., Qiao, R., An, H., & Zhang, Y. (2018). Influence of microplastics on the accumulation and chronic toxic effects of cadmium in zebrafish (Danio rerio). Chemosphere, 202, 514–520. https://doi.org/10.1016/j.chemosphere.2018.03.145
Luo, J., Pei, S., Jing, W., Zou, E., & Wang, L. (2015). Cadmium inhibits molting of the freshwater crab Sinopotamon henanense by reducing the hemolymph ecdysteroid content and the activities of chitinase and N-acetyl-β-glucosaminidase in the epidermis. Comparative Biochemistry and Physiology Part C, 169, 1–6. https://doi.org/10.1016/j.cbpc.2014.10.003
Marcial, H. S., & Hagiwara, A. (2007). Multigenerational effects of 17?-estradiol and nonylphenol on euryhaline cladoceran Diaphanosoma celebensis. Fisheries Science, 73(2), 324–330. https://doi.org/10.1111/j.1444-2906.2007.01338.x
Morcillo, P., Esteban, M. A., & Cuesta, A. (2017). Mercury and its toxic effects on fish. AIMS Environmental Science, 4(3), 386–402. https://doi.org/10.3934/environsci.2017.3.386
Moreira, R. A., de Araujo, G. S., Silva, A. R. R. G., Daam, M. A., Rocha, O., Soares, A. M. V. M., & Loureiro, S. (2020). Effects of abamectin-based and difenoconazole-based formulations and their mixtures in Daphnia magna: A multiple endpoint approach. Ecotoxicology, 29(9), 1486–1499. https://doi.org/10.1007/s10646-020-02218-z
Naidoo, T., & Glassom, D. (2019). Decreased growth and survival in small juvenile fish, after chronic exposure to environmentally relevant concentrations of microplastic. Marine Pollution Bulletin, 145, 254–259. https://doi.org/10.1016/j.marpolbul.2019.02.037
Nelms, S. E., Galloway, T. S., Godley, B. J., Jarvis, D. S., & Lindeque, P. K. (2018). Investigating microplastic trophic transfer in marine top predators. Environmental Pollution, 238, 999–1007. https://doi.org/10.1016/j.envpol.2018.02.016
Oliveira, P., Barboza, L. G. A., Branco, V., Figueiredo, N., Carvalho, C., & Guilhermino, L. (2018). Effects of microplastics and mercury in the freshwater bivalve Corbicula fluminea (Müller, 1774): Filtration rate, biochemical biomarkers and mercury bioconcentration. Ecotoxicology and Environmental Safety, 164, 155–163. https://doi.org/10.1016/j.ecoenv.2018.07.062
Pacheco-Alvarez, D., Solórzano-Vargas, R. S., & Del Rı́o, A. L. (2002). Biotin in metabolism and its relationship to human disease. Archives of Medical Research, 33(5), 439–447. https://doi.org/10.1016/s0188-4409(02)00399-5
Parrish, C. C. (2009). Essential fatty acids in aquatic food webs. Lipids in Aquatic Ecosystems. https://doi.org/10.1007/978-0-387-89366-2_13
PlasticsEurope. (2021). Plastics – The facts 2021: An analysis of European plastics production, demand and waste data. https://plasticseurope.org/knowledge-hub/plastics-the-facts-2021/.
Rehse, S., Kloas, W., & Zarfl, C. (2018). Microplastics Reduce short-term effects of environmental contaminants. Part I: Effects of bisphenol a on freshwater zooplankton are lower in presence of polyamide particles. International Journal of Environmental Research and Public Health, 15(2), 280. https://doi.org/10.3390/ijerph15020280
Rohlfs, E. M., Garner, S. C., Mar, M.-H., & Zeisel, S. H. (1993). Glycerophosphocholine and phosphocholine are the major choline metabolites in rat milk. The Journal of Nutrition, 123(10), 1762–1768. https://doi.org/10.1093/jn/123.10.1762
Schettino, T., Caricato, R., Calisi, A., Giordano, M. E., & Lionetto, M. G. (2012). Biomarker approach in marine monitoring and assessment: New insights and perspectives. Open Environmental Sciences, 6, 20–27. https://doi.org/10.2174/1876325101206010020
Sıkdokur, E., Belivermiş, M., Sezer, N., Pekmez, M., Bulan, Ö. K., & Kılıç, Ö. (2020). Effects of microplastics and mercury on manila clam Ruditapes philippinarum: Feeding rate, immunomodulation, histopathology and oxidative stress. Environmental Pollution, 262, 114247. https://doi.org/10.1016/j.envpol.2020.114247
Sokolova, I. M. (2013). Energy-limited tolerance to stress as a conceptual framework to integrate the effects of multiple stressors. Integrative and Comparative Biology, 53(4), 597–608. https://doi.org/10.1093/icb/ict028
Surjana, D., Halliday, G. M., & Damian, D. L. (2010). Role of nicotinamide in DNA damage, mutagenesis, and DNA repair. Journal of Nucleic Acids, 2010, 1–13. https://doi.org/10.4061/2010/157591
Sussarellu, R., Suquet, M., Thomas, Y., Lambert, C., Fabioux, C., Pernet, M. E. J., Le Goïc, N., Quillien, V., Mingant, C., Epelboin, Y., Corporeau, C., Guyomarch, J., Robbens, J., Paul-Pont, I., Soudant, P., & Huvet, A. (2016). Oyster reproduction is affected by exposure to polystyrene microplastics. Proceedings of the National Academy of Sciences, 113(9), 2430–2435. https://doi.org/10.1073/pnas.1519019113
Tang, Y., Rong, J., Guan, X., Zha, S., Shi, W., Han, Y., Du, X., Wu, F., Huang, W., & Liu, G. (2020). Immunotoxicity of microplastics and two persistent organic pollutants alone or in combination to a bivalve species. Environmental Pollution, 258, 113845. https://doi.org/10.1016/j.envpol.2019.113845
Thompson, R. C. (2015). Microplastics in the marine environment: sources consequences and solutions. Marine Anthropogenic Litter. https://doi.org/10.1007/978-3-319-16510-3_7
Wen, B., Jin, S.-R., Chen, Z.-Z., Gao, J.-Z., Liu, Y.-N., Liu, J.-H., & Feng, X.-S. (2018). Single and combined effects of microplastics and cadmium on the cadmium accumulation, antioxidant defence and innate immunity of the discus fish (Symphysodon aequifasciatus). Environmental Pollution, 243, 462–471. https://doi.org/10.1016/j.envpol.2018.09.029
Xie, D., Wei, H., Lee, J.-S., & Wang, M. (2022). Mercury can be transported into marine copepod by polystyrene nanoplastics but is not bioaccumulated: An increased risk? Environmental Pollution, 303, 119170. https://doi.org/10.1016/j.envpol.2022.119170
Yoo, J.-W., Cho, H., Jeon, M., Jeong, C.-B., Jung, J.-H., & Lee, Y.-M. (2021). Effects of polystyrene in the brackish water flea Diaphanosoma celebensis: Size-dependent acute toxicity, ingestion, egestion, and antioxidant response. Aquatic Toxicology, 235, 105821. https://doi.org/10.1016/j.aquatox.2021.105821
Yoo, J.-W., Jeon, M., Lee, K. W., Jung, J. H., Jeong, C. B., & Lee, Y. M. (2022). The single and combined effects of mercury and polystyrene plastic beads on antioxidant-related systems in the brackish water flea: Toxicological interaction depending on mercury species and plastic bead size. Aquatic Toxicology, 252, 106325. https://doi.org/10.1016/j.aquatox.2022.106325
Yu, L., Li, K., & Zhang, X. (2017). Next-generation metabolomics in lung cancer diagnosis, treatment and precision medicine: Mini review. Oncotarget, 8(70), 115774–115786. https://doi.org/10.18632/oncotarget.22404
Zhang, Q.-L., Dong, Z.-X., Luo, Z.-W., Zhang, M., Deng, X.-Y., Guo, J., Wang, F., & Lin, L.-B. (2020). The impact of mercury on the genome-wide transcription profile of zebrafish intestine. Journal of Hazardous Materials, 389, 121842. https://doi.org/10.1016/j.jhazmat.2019.121842
Zhu, J., Wang, C., Gao, X., Zhu, J., Wang, L., Cao, S., Wu, Q., Qiao, S., Zhang, Z., & Li, L. (2019). Comparative effects of mercury chloride and methylmercury exposure on early neurodevelopment in zebrafish larvae. RSC Advances, 9(19), 10766–10775. https://doi.org/10.1039/c9ra00770a
Zhu, J., Zhang, Y., Xu, Y., Wang, L., Wu, Q., Zhang, Z., & Li, E. (2022). Effects of microplastics on the accumulation and neurotoxicity of methylmercury in zebrafish larvae. Marine Environmental Research, 176, 105615. https://doi.org/10.1016/j.marenvres.2022.105615
Acknowledgements
This research was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No.2020R1F1A1069736) to Young-Mi Lee, and the Hankuk University of Foreign Studies Research Fund (20211441001) and NRF (MSIT project: 2021R1C1C1006155) to Tae-Yong Jeong.
Funding
This research was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No.2020R1F1A1069736) to Young-Mi Lee, and the Hankuk University of Foreign Studies Research Fund (20211441001) and NRF (MSIT project: 2021R1C1C1006155) to Tae-Yong Jeong.
Author information
Authors and Affiliations
Contributions
YML contributed to conceptualization, writing, review and editing, and funding; T-YJ contributed to writing, review and editing, and funding; J-WY contributed to chemicals exposure, sample preparation, and original draft writing. H-JB contributed to metabolome data analysis, MJJ contributed to culture maintenance and chemical exposure. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or nonfinancial interests to disclose.
Ethics approval
No approval of research ethics committees was required to accomplish the goals of this study because experimental work was conducted with an unregulated invertebrate species.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoo, JW., Bae, HJ., Jeon, M.J. et al. Metabolomic analysis of combined exposure to microplastics and methylmercury in the brackish water flea Diaphanosoma celebensis. Environ Geochem Health 45, 6807–6822 (2023). https://doi.org/10.1007/s10653-022-01435-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-022-01435-1