Abstract
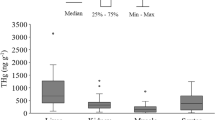
Due to their natural history and ecological attributes, turtles are excellent organisms for studies of heavy metal contamination. Turtles have a large geographical distribution, occupy different aquatic habitats, and occupy various trophic levels. The present study investigated mercury bioaccumulation in the carnivorous chelonian Chelus fimbriata (Matamata turtle) and Hg biomagnification in relation to its aquatic food chain in the middle Rio Negro, AM-Brazil. Tissue samples of muscle, carapace and claws were collected from 26 C. fimbriata individuals, as well as collections of autotrophic energy sources found in the turtle’s aquatic habitat area. The samples were collected in February-March/2014 and analyzed for THg concentrations and carbon (δ13C) and nitrogen (δ15N) stable isotopes. The highest THg levels were found in claws (3780 ng.g−1), carapace (3622 ng.g−1) and muscle (403 ng.g−1), which were found to be significantly different [F(2.73) = 49.02 p < 0.01]. However, THg concentrations in muscle tissue were below the consumption threshold indicated by the WHO and Brazilian Health Ministry. The average δ13C and δ15N values in Matamata samples were −31.7‰ and 11.9‰, respectively. The principal energy source sustaining the food chain of C. fimbriata was found to be terrestrial shrubs, with smaller contributions from emergent aquatic herbaceous plants and algae, while δ15N values showed its trophic position to be two levels above the autotrophic energy sources. There was a positive correlation between THg and turtle size, while a significant relationship was found between THg and δ15N, showing strong biomagnification in the food chain of C. fimbriata: y = 0.21x + 0.46; r2 = 0.45; p < 0.001, for which the slope presented a value of 0.21.









Similar content being viewed by others
References
Atwell L, Hobson KA, Welch HE (1998) Biomagnification and bioaccumulation of mercury in an arctic marine food web: insights from stable nitrogen isotope analysis. Can J Fish Aqua Sci 55:1114–1121. https://doi.org/10.1139/f98-001
Barbosa AC, Souza J, Dórea JG, Jardin WF, Fadini PS (2003) Mercury Biomagnification in a Tropical Black Water, Rio Negro, Brazil. Arch Environ Contam Toxicol 45:235–246. https://doi.org/10.1007/s00244-003-0207-1
Barletta M, Lucena LRR, Costa MF, Barbosa-Cintra SCT, Cysneiros FJA (2012) The interaction rainfall vs. Weight as determinant of total mercury concentration in fish from a tropical estuary. Environ Pollut 167:1–6. https://doi.org/10.1016/j.envpol.2012.03.033
Bastos WR, Dórea JG, Bernardi JVE, Lauthartte LC, Mussy MH, Lacerda LD, Malm O (2015) Mercury in fish of the Madeira River (temporal and spatial assessment), Brazilian Amazon. Environ Res 140:191–197. https://doi.org/10.1016/j.envres.2015.03.029
Bastos RF, Correa F, Winemiller KO, Garcia AM (2017) Are you what you eat? Effects of trophic discrimination factors on estimates of food assimilation and trophic position with a new estimation method. Ecol Indic 75:234–241. https://doi.org/10.1016/j.ecolind.2016.12.007
Beau F, Bustamante P, Michaud B, Brischoux F (2019) Environmental causes and reproductive correlates of mercury contamination in European pond turtles (Emys orbicularis). Environ Res 172:338–344. https://doi.org/10.1016/j.envres.2019.01.043
Belger L, Forsberg BR (2006) Factors controlling Hg levels in two predatory fish species in the Negro River basin, Brazilian Amazon. Sci Total Environ 367:451–459. https://doi.org/10.1016/j.scitotenv.2006.03.033
Beltran-Predreros S, Zuanon J, Leite RG, Peleja JRP, Mendonça AB, Forsberg BR (2011) Mercury bioaccumulation in fish of commercial importance from different trophic categories in an Amazon floodplain lake. Neotrop Ichthyol 9:901–908. https://doi.org/10.1590/S1679-62252011000400022
Bezerra MF, Lacerda LD, Costa BGB, Lima EHSM (2012) Mercury in the sea turtle Chelonia mydas (Linnaeus, 1958) from Ceará coast, NE Brazil. An Acad Bras Ciênc 84:123–128. https://doi.org/10.1590/S0001-37652012000100012
Borges AO, Erickson J, Silva LA, Fantin C, Domingo-Moreira FXV (2022) Mercury bioaccumulation, genotoxic and biochemical biomarkers reveal the health status of yellow-spotted Amazon River turtles (Podocnemis unifilis) in an environmental protection area in the Amazon. Acta Amaz 52:253–263. https://doi.org/10.1590/1809-4392202201230
Bourgoin LM, Quemerais B, Turcq PM, Seyler P (2002) Transport, distribution and speciation of mercury in the Amazon River at the confluence of black and white waters of the Negro and Solimões Rivers. Hydrol Process 17:1405–1417. https://doi.org/10.1002/hyp.1292
Brito CB, Forsberg BR, Kasper D, Amaral JHF, Vasconcelos MRR, Sousa OP, Cunha FAG, Bastos WR (2016) The influence of inundation and lake morphometry on the dynamics of mercury in the water and plankton in an Amazon floodplain lake. Hydrobiologia 790:35–48. https://doi.org/10.1007/s10750-016-3017-y
Bulté G, Gravel MA, Blouin-Demers G (2008) Intersexual niche divergence in northern map turtles (Graptemys geographica): the roles of diet and habitat. Can J Zool 86:1235–1243. https://doi.org/10.1139/Z08-107
Burger J, Jeitner C, Schneider L, Vogt R, Gochfeld M (2010) Arsenic, cadmium, chromium, lead, mercury, and selenium levels in blood of four species of turtles from the Amazon in Brazil. J Toxicol Environ Health A 73:33–40. https://doi.org/10.1080/15287390903248877
Campbell L, Verburg P, Dixon DG, Hecky RE (2008) Mercury biomagnification in the foodweb of Lake Tanganyika (Tanzania, East Africa). Sci Total Environ 402:184–191. https://doi.org/10.1016/j.scitotenv.2008.04.017
Carvalho F, Power M, Forsberg BR, Castello L, Martins EG, Freitas CEC (2017) Trophic Ecology of Arapaima sp. in a ria lake-river-floodplain transition zone of the Amazon. Ecol Freshw Fish 27:1–10. https://doi.org/10.1111/eff.12341
Châteauvert JL, Bulté G, Poulain AJ et al. (2015) Dietary Reliance on Benthic Primary Production as a Predictor of Mercury Accumulation in Freshwater Fish and Turtles. Water Air Soil Pollut 226:337. https://doi.org/10.1007/s11270-015-2610-4
Crewther WG, Fraser RD, Lennox FG, Lindley H (1965) The chemistry of keratins. Adv Prot Chem 20:191–346. https://doi.org/10.1016/s0065-3233(08)60390-3
Cunha FAG (2015) Biomagnificação do mercúrio na cadeia de um quelônio de água doce (Chelus fimbriata) e seu perfil genotóxico em ambientes fluviais do médio rio Negro, Amazonas, Brasil. Dissertation, Instituto Nacional de Pesquisa da Amazônia.
Cunha FAG, Fagundes CK, Brito ES, Vogt RC, Maffei F, Pezzuti J, Félix-Silva D, Rojas-Runjaic FJM, Lasso CA, Morales-Betancourt MA, de Carvalho VT, do Amaral JV, Balestra RAM, Acácio M, Malvasio A, Lustosa PG (2021) Distribution of Chelus fimbriata and Chelus orinocensis (Testudines: Chelidae). Chelon Conser Biol 20:109–115. https://doi.org/10.2744/CCB-1398.1
Cunha FAG, Sampaio I, Carneiro J, Vogt RC, Mittermeier RA, Rhodin AGJ, Andrade AC (2022) A New South American Freshwater Turtle of the Genus Mesoclemmys Gray 1873 from the Brazilian Amazon (Testudines: Pleurodira: Chelidae). Chelon Conser Biol 21:158–180. https://doi.org/10.2744/CCB-1448.1
Day RD, Christopher SJ, Becker PR, Whitaker DW (2005) Monitoring Mercury in the Loggerhead Sea Turtle, Caretta caretta. Environ Sci Technol 39:437–446. https://doi.org/10.1021/es049628q
DeNiro MJ, Epstein S (1978) Influence of diet on the distribution of carbon isotopes in animals. Geochim Cosmochim Acta 42:495–506. https://doi.org/10.1016/0016-7037(78)90199-0
Dorea JG, Barbosa AC, Ferrari I, De Souza JR (2003) Mercury in hair and in fish consumed by Riparian women of the Rio Negro, Amazon, Brazil. Int J Environ Health Res 13:239–248. https://doi.org/10.1080/0960312031000122398
Dorea JG, Barbosa AC, Silva GS (2006) Fish mercury bioaccumulation as a function of feeding behavior and hydrological cycles of the Rio Negro, Amazon. Comp Biochem Physiol C: Pharmacol Toxicol 141:275–283. https://doi.org/10.1016/j.cbpc.2005.10.014
Duméril AMC (1805) Zoologie analytique ou méthode naturelle de classification des animaux. Paris: Perronneau. France. pp. 344
Eggins S, Schneider L, Krikowa F, Vogt RC, Silveira R, Maher W (2015) Mercury concentrations in different tissues of turtle and caiman species from the Rio Purus, Amazonas, Brazil. Environ Toxicol Chem 34:2771–2791. https://doi.org/10.1002/etc.3151
Fachin-Teran A, Vogt RC, Gomez MFS (1995) Food habits of an assemblage of five species of turtles in the Rio Guapore, Rondonia, Brazil. J Herpeto 29:536–547. https://doi.org/10.2307/1564736
Fadini OS, Jardim WF (2001) Is the Negro River basin (Amazon) impacted by naturally occurring mercury? Sci Total Environ 275:71–82. https://doi.org/10.1016/s0048-9697(00)00855-x
Forsberg BR, Araújo-Lima CARM, Martinelli LA, Victoria RL, Bonassi JA (1993) Autotrophic carbon sources for fish of the central Amazon. Ecology 74:643–652. https://doi.org/10.2307/1940793
Forsberg BR, Castro JGD, Cargnin-Ferreira E, Rosenqvist A (2001) The structure and function of the Negro River Ecosystem: Insights from the Jau Project. Conservation and Management of Ornamental Fish Resources of the Rio Negro Basin, Amazonia, Brazil
German D, Miles R (2010) Stable carbon and nitrogen incorporation in blood and fin tissue of the catfish Pterygoplichthys disjunctivus (Siluriformes, Loricariidae). Environ Biol Fishes 89:117–133. https://doi.org/10.1007/s10641-010-9703-0
Golet WJ, Haines TA (2001) Snapping turtles (Chelydra serpentina) as monitors for mercury contamination of aquatic environments. Environ Monit Assess 71:211–220. https://doi.org/10.1023/A:1011802117198
Green AD, Buhlmann KA, Hagen C, Romanek C, Gibbons JW (2010) Mercury Contamination in Turtles and Implications for Human Health. J Environ Health 72:14–22. https://pubmed.ncbi.nlm.nih.gov/20556939/
Hoffmann M, Hilton-Taylor C, Angulo A, Bohm M, Brooks TM, Butchart SHM, Carpenter KE, Chanson J, Collen B, Cox NA (2010) The impact of conservation on the status of the world’s vertebrates. Science 330:1503–1509. https://doi.org/10.1126/science.1194442
Hopkins WA, Rowe CL (2010) Interdisciplinary and hierarchical approaches for studying the effects of metals and metalloids on amphibians. In: Sparling D, Linder G, Bishop CA (eds.) Ecotoxicology of amphibians and reptiles, 2nd ed. SETAC Press, Pensacola, p 325–336
Hopkins BC, Hepner MJ, Hopkins WA (2013a) Non-destructive techniques for biomonitoring of spatial, temporal, and demographic patterns of mercury bioaccumulation and maternal transfer in turtles. Environ Pollut 177:164–170. https://doi.org/10.1016/j.envpol.2013.02.018
Hopkins WA, Bodinof C, Budischak S, Perkins C (2013b) Non-destructive indices of mercury exposure in three species of turtles occupying different trophic niches downstream from a former chloralkali facility. Ecotoxicology 22:22–32. https://doi.org/10.1007/s10646-012-0999-8
Junk W, Bayley P, Sparks RE (1989) The Flood Pulse Concept in River-Floodplain Systems. Canadian J Fisher. Aquat Sci 106:110–127
Kar P, Mirsa M (2004) Use of keratin fiber for separation of heavy metals from water. J Chemi Techno Biotech 79:1313–1319. https://doi.org/10.1002/jctb.1132
Kasper D, Fernandes E, Palermo A, Branco CWC, Malm O (2012) Evidence of elevated mercury levels in carnivorous and omnivorous fishes downstream from an Amazon reservoir. Hydrobiologia 694:87–98. https://doi.org/10.1007/s10750-012-1133-x
Kasper D, Forsberg BR, Amaral JHF, Leitão RP, Py-Daniel SP, Bastos WR, Malm O (2014) Reservoir Stratification Affects Methylmercury Levels in River Water, Plankton, and Fish Downstream from Balbina Hydroelectric Dam, Amazonas, Brazil. Environ Sci Technol 48:1032–1040. https://doi.org/10.1021/es4042644
Kasper D, Forsberg BR, Amaral JHF, Py-Daniel SS, Bastos WR, Malm O (2017) Methylmercury Modulation in Amazon Rivers Linked to Basin Characteristics and Seasonal Flood-Pulse. Environ Sci Technol 51:14182–14191. https://doi.org/10.1021/acs.est.7b04374
Kehrig HA, Seixas TG, Palermo EA, Di Beneditto APM, Souza CMM, Malm O (2008) Different species of mercury in the livers of tropical dolphins. Anal Lett 41:1690–1698. https://doi.org/10.1080/00032710802122677
Kenyon LO, Landry AM, Gill GA (2001) Trace metal concentrations in blood of Kemp’s Ridley sea turtle (Lepidochelys kempii). Chelon Conser Biol 4:128–135
Kidd KA, Hesslein RH, Fudge RJP, Hallard KA (1995) The influence of trophic level as measured by δ15N on mercury concentrations in freshwater organisms. Water Air Soil Pollut 80:1011–1015. https://doi.org/10.1007/BF01189756
Komoroske LM, Lewison RL, Seminoff JA, Deheyn DD, Dutton PH (2011) Pollutants and the health of green sea turtles resident to an urbanized estuary in San Diego, CA. Chemosphere 84:544–552. https://doi.org/10.1016/j.chemosphere.2011.04.023
Lara NRF, Marques TS, Montelo KM, de Ataídes AG, Verdade LM, Malvásio A, de Camargo PB (2012) A trophic study of the sympatric Amazonian freshwater turtles Podocnemis unifilis and Podocnemis expansa (Testudines, Podocnemididae) using carbon and nitrogen stable isotope analyses. Can J Zool 90:1394–1401. https://doi.org/10.1139/cjz-2012-0143
Lavoie RA, Jardine TD, Chumchal MM, Kidd KA, Campbell LM(2013) Biomagnification of mercury in aquatic food webs: a worldwide meta-analysis Environ. Sci. Technol 47:13385–13394. https://doi.org/10.1021/es403103t
Lewis JWM, Hamilton SK, Rodríguez MA, Saunders JF, Lasi MA (2001) Foodweb analysis of the Orinoco floodplain based on production estimates and stable isotope data. J N Am Benthol Soc 20:241–254. https://doi.org/10.2307/1468319
Marshall BG, Forsberg BR, Thomé-Souza M, Peleja JRP, Moreira MZ, Freitas CEC (2016) Evidence of mercury biomagnification in the food chain of the cardinal treta Paracheirodon axelroldi (Osteichthyes: Characidae) in the Rio Negro, central Amazon, Brazil. J Fish Biol 89:220–40. https://doi.org/10.1111/jfb.12952
Matias RS, Guímaro HR, Bustamante P, Seco J, Chipev N, Fragão J, Tavares S, Ceia FR, Pereira ME, Barbosa A, Xavier JC (2022) Mercury biomagnification in an Antarctic food web of the Antarctic Peninsula. Environ Pollut 304:119–199. https://doi.org/10.1016/j.envpol.2022.119199
Melendez-Perez JJ, Fostier AH (2013) Assessment of Direct Mercury Analyzer to Quantify Mercury in Solis and Leaf Samples. J Braz Chem Soc 24:1880–1886. https://doi.org/10.5935/0103-5053.20130235
Negrazis L, Kikk KA, Erdozain M, Emilson EJS, Mitchell CPJ, Gray MA (2022) Effects of forest management on mercury bioaccumulation and biomagnification along the river continuum. Environ Pollut 310:119–810. https://doi.org/10.1016/j.envpol.2022.119810
Nyholt K, Jardine TD, Villamarín F, Jacobi CM, Hawes JE, Campos-Silva JV, Srayko S, Magnusson WE (2022) High rates of mercury biomagnification in fish from Amazonian floodplain-lake food webs. Sci Total Environ 833:155–161. https://doi.org/10.1016/j.scitotenv.2022.155161
Oliveira LC, Serudo RL, Botero WG, Mendonça AGR, Santos A, Rocha JC, Neto FSC (2007) Distribuição de Mercúrio em Diferentes Solos da Bacia do Médio rio Negro-Am: Influência da Matéria Orgânica no Ciclo Biogeoquímico do Mercúrio. Quím Nova 30:274–280. https://doi.org/10.1590/S0100-40422007000200006
Paiva TC, Dary EP, Pestana IA, Amadio SA, Malf O, Kasper D (2022) Flood-pulse and Trophic position modulate mercury concentrations in fishes from an Amazon floodplain lake. Environ Res 215:114–307. https://doi.org/10.1016/j.envres.2022.114307
Parnell AC et al. (2013) Bayesian stable isotope mixing models. Environmetrics 24:387–399. https://doi.org/10.1002/env.2221
Peleja JRP (2007) Balanço de massas de mercúrio (Hg) total em duas microbacias da Amazônia Central, Tese de doutorado. Instituto Nacional de Pesquisas da Amazônia, Manaus, Brasil
Pereira LA, Santos RV, Hauser M, Duponchelle F, Carvajal F, Pecheyran C, Bérail S, Pouilly M (2019) Commercial traceability of Arapaima spp. fisheries in the Amazon basin: can biogeochemical tags be useful? Biogeosciences 16:1781–1797. https://doi.org/10.5194/bg-16-1781-2019
Pezzuti JCB, Oliveira T, Pantoja-Lima J, Rebêlo GH, Félix-Silva D (2022) Temporal and Spatial Stability on the Population Structure of Consumed and Illegally Traded Big-Headed Amazon River Turtle in the Negro River Basin, Central Amazon, Brazil. Front Ecol Evol 10:640–961. https://doi.org/10.3389/fevo.2022.640961
Pignati MT, Pezzuti JCB, Souza LC, Lima MC, Pignati WA, Mendes RD (2018) Assessment of mercury concentration in turtles (Podocnemis unifilis) in the Xingu River Basin, Brazil 15:1–11. https://doi.org/10.3390/ijerph15061185
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods and assumptions. Ecology 83:703–718. https://doi.org/10.2307/3071875
Pritchard PCH (2008) Chelus fimbriata (Schneider 1783) — Matamata turtle. In: Rhodin, AGJ, Pritchard, PCH., van Dijk, PP, Saumure, RA, Buhlmann, KA, and Iverson, JB (Eds.) Conservation Biology of Freshwater Turtles and Tortoises: A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelon Res Monogra, pp 20-28.
R Core Team. 2021. Language and Environment for Statistical Computing version 4.1.2. https://www.R-project.org/.
Rebêlo GH, Pezzuti JCB (2000) Percepções sobre o consumo de quelônios na Amazônia: sustentabilidade e alternativas ao manejo atual. Ambient Soci 6:85–104. https://doi.org/10.1590/S1414-753X2000000100005
Sakai H, Saeki K, Ichihashi H, Kamezaki N, Tanabe S, Tatsukawa R (2000) Growth-related changes in heavy metal accumulation in green turtle (Chelonia mydas) from Yaeyama Islands, Okinawa, Japan. Arch Environ Contam Toxicol 39:378–385. https://doi.org/10.1007/s002440010118
Sampaio da Silva D, Lucotte M, Roulet M, Poirier H, Mergler D, Santos EO, Crossa M (2005) Trophic structure and bioaccumulation of mercury in fish of three natural lakes of the Brazilian Amazon. Water Air Soil Pollut 165:77–94. https://doi.org/10.1007/s11270-005-4811-8
Schneider JG (1783) Allgemeine Naturgeschichte der Schildkröten, nebst einem systematischen Verzeichnisse der einzelnen Arten und zwei Kupfern. J.G. Muller, Leipzig. pp. 364
Schneider L, Belger L, Burger J, Vogt RC, Ferrara CR (2010) Mercury levels in muscle of six species of turtles eaten by people along the Rio Negro of the Amazon Basin. Arch Envirom Contam Toxicol 10:444–450. https://doi.org/10.1007/s00244-009-9358-z
Schneider L, Belger L, Burger J, Vogt RC, Jeitner C, Peleja JRP (2011) Assessment of non-invasive techniques for monitoring mercury concentrations is species of Amazon turtles. Toxicol Environ Chemi 93:238–250. https://doi.org/10.1080/02772248.2010.517627
Schneider L, Eggins S, Maher W, Vogt RC, Krikowa F, Kinsley L, Eggins S, Silveira R (2015) An evaluation of the use of reptile dermal scutes as a non-invasive method to monitor mercury concentrations in the environment. Chemosphere 119:163–170. https://doi.org/10.1016/j.chemosphere.2014.05.065
Silva-Forsberg MC, Forsberg BR, Zeidemann VK (1999) Mercury contamination in humans linked to river chemistry in the Amazon basin. Ambio 28:519–521
Slimani T, Hassani EMS, Mouden EEH et al. (2018) Large-scale geographic patterns of mercury contamination in Morocco revealed by freshwater turtles. Environ Sci Pollut Res 25:2350–2360. https://doi.org/10.1007/s11356-017-0643-5
Soares JLF, Goch YGF, Peleja JRP, Forsberg BR, Lemos EJS, Sousa OP (2016) Bioacumulação de Mercúrio Total (Hgt) e hábitos alimentares de peixes da bacia do Rio Negro, Amazônia, Brasil. Bio Amazon 6:102–106. https://doi.org/10.18561/2179-5746/biotaamazonia.v6n1p102-106
Stock BC, Jackson AL, Ward EJ, Parnell AC, Phillips DL, Semmens BX (2018) Analyzing mixing systems using a new generation of Bayesian tracer mixing models. PeerJ 6:e5096. https://doi.org/10.7717/peerj.5096
Swartz CD, Donnelly KC, Islamzadeh A, Rowe GT, Rogers WJ, Palatnikov GM, Mekhtiev AA, Kasimov R, McDonald TJ, Wickliffe JK, Presley BJ, Bickham JW (2003) Chemical contaminants and their effects in fish and wildlife from the industrial zone of Sumgayit, Republic of Azerbaijan. Ecotoxicology 12:509–52. https://doi.org/10.1023/B:ECTX.0000003038.02643.08
TTWG [Turtle Taxonomy Working Group: Rhodin AGJ, Iverson JB, Bour R, Fritz U, Georges A, Shaffer HB, Van Dijk PP (2021) Turtles of the World: Annotated Checklist and Atlas of Taxonomy, Synonymy, Distribution, and Conservation Status (9th Ed.). In: Rhodin, AGJ, Iverson, JB, Van Dijk, PP, Stanford, CB, Goode, EV, Buhlmann, KA, Mittermeier RA (Eds.) Conservation Biology of Freshwater Turtles and Tortoises: A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelonian Research Monographs.
USEPA (1998) Method 7473 (SW-846): Mercury in solids and solutions by thermal decomposition, amalgamation, and atomic absorption spectrophotometry. Revision 0, Washington, DC
Vargas-Ramírez M, Caballero S, Morales-Betancourt MA, Lasso CA, Amaya L, Martínez JG, Viana MNS, Vogt RC, Farias IP, Hrbek T, Campbell PD, Fritz U (2020) Genomic analyses reveal two species of the matamata (Testudines: Chelidae: Chelus spp.) and clarity their phylogeography. Mol Phylogenet Evol 148:106–823. https://doi.org/10.1016/j.ympev.2020.106823
Vasconcellos ACS, Ferreira SRB, Sousa CR, Oliveira MW, Lima MO, Basta PC (2022) Health risk assessment attributed to consumption of fish contaminated with Mercury in Rio Branco Basin, Roraima, Amazon, Brazil. Toxics 10:516. https://doi.org/10.3390/toxics10090516
Vasconcelos MRR, Brito BC, Forsberg BR, Goch YGF, Malm O, Melo S, Kasper D (2022) Export and bioaccumulation of methylmercury in streams draining distinct soils in the Central Brazilian Amazon 2012-2013. J Trace Elements Minerals 2:100014. https://doi.org/10.1016/j.jtemin.2022.100014
Vogt RC (2008) Tartarugas da Amazônia. Gráfica Biblios, Lima, Perú
Yoshinaga J, Suzuki T, Hongo T, Minagawa M, Ohtsuka R, Kawabe T, Inaoka T, Akimichi T (1992) Mercury Concentration Correlates with the Nitrogen Stable Isotope Ratio in the Animal Food of Papuans. Ecotoxicol Environ Saf 24:37–45. https://doi.org/10.1016/0147-6513(92)90033-y
Yu S, Halbrook RS, Sparling DW, Colombo R (2011) Metal accumulation and evalution of effects in a freshwater turtle. Ecotoxicology 20:1801–1812. https://doi.org/10.1007/s10646-011-0716-z
Zanden HBV, Bjorndal KA, Mustin W, Ponciano JM, Bolten AB (2012) Inherent Variation in Stable Isotope Values and discrimination Factors in Two Life Stages of Green Turtles. Physiol Biochem Zool 85:431–441. https://doi.org/10.1086/666902
Zar JH (2010) Biostatistical analysis, 5th edition. Pearson Education, Harlow
Zhang L, Campbell LM, Johnson TB (2012) Seasonal variation in mercury and food web biomagnifications in Lake Ontario, Canada. Environ Pollut 161:178–184. https://doi.org/10.1016/j.envpol.2011.10.023
Acknowledgements
This study is part of the doctoral thesis of the first author (FAGC) at the Graduate Program on Aquatic Ecology and Fishing, Federal University of Pará, and was funded in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (Capes) – Finance Code 001, with additional support from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), of the Ministério de Ciência, Tecnologia e Inovação (MCTI). MÂ CNPq (#310522/2023-4). We would like to thank the Programa de Pós-Graduação em Biologia de Água Doce e Pesca Interior (BADPI) at Instituto Nacional de Pesquisas da Amazônia (INPA) and Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM) for all the logistical and intellectual support provided by PPG researchers and professors. By name, I want to thank Mr. Anízio from Barcelos and his sons Beto, Ruse and Sandro. The crew of the boat Mário, Luiz and Astro, especially Dr. Richard Vogt. To BADPI and INPA for their intellectual and logistical contribution to this research. To the Amazonian Ecosystems Laboratory (INPA, Manaus, AM) for all the logistical and intellectual support. To the Geological Survey of Brazil (CPRM Manaus, AM) for the partnership in the Hg analyses, on behalf of Raimundo Gato, Bruno Calvo and Ceel. To the Laboratory of Geochronology and Stable Isotopes of the University of Brasília (UnB, Brasília, DF) on behalf of Eduardo Carvalho and Luis Mancini. To the Fish Nutrition Laboratory (INPA, Manaus, AM) for lyophilizing the plankton samples. To the Laboratory of Ecophysiology and Molecular Evolution, on behalf of Dr. Rafael “Sussu” (INPA, Manaus, AM) for the COD analyses. To the Mauá Group, on behalf of Dr. Maria Tereza Piedade and Dr. Aline Lopes for identifying the plants.
Funding
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (Capes) – Finance Code 001. Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), of the Ministério de Ciência, Tecnologia e Inovação. Projeto CNPq Dr. Bruce R. Forsberg Processo n° 458036/2013-7. Projeto Tartarugas da Amazônia: Conservando para o futuro, coordenador Dr. Richard C. Vogt.
Author information
Authors and Affiliations
Contributions
FAG, Cunha: hypothesis, conceptualization, sampling, chemical analysis, data analysis, writing, discussion and submission; BC, Brito: conceptualization, sampling, data analysis, writing; B.R. Forsberg: conceptualization, data analysis, writing, funding acquisition and advisor; RC, Vogt: conceptualization and advisor; D, Kasper: conceptualization, writing; Bruce Marshall: conceptualization, writing, translation; O, Peleja: conceptualization; ALP, Santos: data analysis; FVX, Domingos-Moreira: conceptualization; MC, Andrade: conceptualization, data analysis, writing and map.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was performed in accordance with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of National Institute for Research of Amazonian (May 2014, N°003).
Informed consent
All authors consent their participation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cunha, F.A.G., Forsberg, B.R., Vogt, R.C. et al. Mercury biomagnification in the food chain of a piscivorous turtle species (Testudines: Chelidae: Chelus fimbriata) in the Central Amazon, Brazil. Ecotoxicology (2024). https://doi.org/10.1007/s10646-024-02756-w
Accepted:
Published:
DOI: https://doi.org/10.1007/s10646-024-02756-w