Abstract
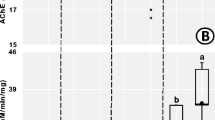
Assessment of organophosphorus (OP) pesticide exposure in non-target organisms rarely involves non-neural molecular targets. Here we performed a 30-d microcosm experiment with Lumbricus terrestris to determine whether the activity of digestive enzymes (phosphatase, β-glucosidase, carboxylesterase and lipase) was sensitive to chlorpyrifos (5 mg kg−1 wet soil). Likewise, acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activities were measured in the wall muscle and gastrointestinal tissues as indicators of OP exposure. Chlorpyrifos inhibited the acid phosphatase (34% of controls), carboxylesterase (25.6%) and lipase activities (31%) in the gastrointestinal content. However, in the gastrointestinal tissue, only the carboxylesterase and lipase activities were significantly depressed (42–67% carboxylesterase inhibition in the foregut and crop/gizzard, and 15% lipase inhibition in the foregut). Chlorpyrifos inhibited the activity of both cholinesterases in the gastrointestinal tissues, whereas the AChE activity was affected in the wall muscle. These results suggested chlorpyrifos was widely distributed throughout the earthworm body after 30 d of incubation. Interestingly, we found muscle carboxylesterase activity strongly inhibited (92% of control) compared with that detected in the gastrointestinal tissues of the same OP-exposed individuals. This finding was explained by the occurrence of pesticide-resistant esterases in the gastrointestinal tissues, which were evidenced by zymography. Our results suggest that digestive processes of L. terrestris may be altered by chlorpyrifos, as a consequence of the inhibitory action of the insecticide on some digestive enzymes.




Similar content being viewed by others
References
Asensio-Ramos M, Hernández-Borges J, Ravelo-Pérez LM, Rodríguez-Delgado MA (2010) Evaluation of a modified QuEChERS method for the extraction of pesticides from agricultural, ornamental and forestal soils. Anal Bioanal Chem 396:2307–2319
Barron MG, Woodburn KB (1995) Ecotoxicology of chlorpyrifos. Rev Environ Contam Toxicol 144:1–93
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Brown GG, Barois I, Lavelle P (2000) Regulation of soil organic matter dynamics and microbial activity in the drilosphere and the role of interactions with other edaphic functional domains. Eur J Soil Biol 36:177–198
Chambers J, Meek E, Chambers H (2010) The metabolism of organophosphorus insecticides. In: Krieger R (ed) Hayes’ handbook of pesticide toxicology. Elsevier, New York (USA), pp 1399–1407
Drake HL, Horn MA (2007) As the worm turns: the earthworm gut as a transient habitat for soil microbial biomes. Annu Rev Microbiol 61:169–189
Ellman GL, Courtney KD, Valentino AJ, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Engelmann P, Cooper EL, Opper B, Németh P (2011) Earthworm innate immune system. In: Karaca A (Ed.) Biology of earthworms, soil biology 24. Springer, Berlin Heidelberg, pp 229–245
Estevez J, Vilanova E (2009) Model equations for the kinetics of covalent irreversible enzyme inhibition and spontaneous reactivation: esterases and organophosphorus compounds. Crit Rev Toxicol 39:427–448
Eyer P, Worek F, Kiderlen D, Sinko G, Stuglin A, Simeon-Rudolf V, Reiner E (2003) Molar absorption coefficients for the reduced Ellman reagent: reassessment. Anal Biochem 312(2):224–227
Fischer E (1993) The myelo-erythroid nature of the chloragogenous-like tissues of the annelids. Comp Biochem Physiol A 106:449–453
Fukuto TR (1990) Mechanism of action of organophosphorus and carbamate insecticides. Environ Health Perspect 87:245–254
Garvı́n MH, Lattaud C, Trigo D, Lavelle P (2000) Activity of glycolytic enzymes in the gut of Hormogaster elisae (Oligochaeta, Hormogastridae). Soil Biol Biochem 32:929–934
Giesy JP, Solomon KR (2014) Ecological risk assessment for chlorpyrifos in terrestrial and aquatic systems in the United States, 3rd edn. Springer, Cham
Griffith B, Türke M, Weisser WW, Eisenhauer N (2013) Herbivore behavior in the anecic earthworm species Lumbricus terrestris L.? Eur J Soil Biol 55:62–65
Gupta N, Rathi P, Gupta R (2002) Simplified para-nitrophenyl palmitate assay for lipases and esterases. Anal Biochem 311:98–99
IUSS Working Group WRB. (2015) World Reference Base for Soil Resources 2014, update 2015 International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome
John EM, Shaike JM (2015) Chlorpyrifos: pollution and remediation. Environ Chem Lett 13:269–291
Kadian N, Malik A, Satya S, Dureja P (2012) Effect of organic amendments on microbial activity in chlorpyrifos contaminated soil. J Environ Manage 95:S199–S202
Kalita MK, Haloi K, Devi D (2016) Larval exposure to chlorpyrifos affects nutritional physiology and induces genotoxicity in silkworm Philosamia ricini (Lepidoptera: Saturniidae). Front Physiol 7:50–14
Lattaud C, Locati S, Mora P et al. (1998) The diversity of digestive systems in tropical geophagous earthworms. Appl Soil Ecol 9:189–195
Mackay D, Shiu W-Y, Ma K-C, Lee SC (2006) Handbook of physical-chemical properties and environmental fate for organic chemicals. Vol. IV. CRC Press, Boca Raton, Florida, USA
Manchenko GP (2002) Handbook of detection of enzymes on electrophoretic gels. CRC Press, Boca Raton, Florida, USA
Masson P, Lockridge O (2010) Butyrylcholinesterase for protection from organophosphorus poisons: catalytic complexities and hysteretic behavior. Arch Biochem Biophys 494:107–120
Mayr S, Erdfelder E, Buchner A, Faul F (2007) A short tutorial of GPower. Tutor Quant Methods Psychol 3:51–59
Muangphra P, Tharapoom K, Euawong N, Namchote S, Gooneratne R (2015) Chronic toxicity of commercial chlorpyrifos to earthworm Pheretima peguana. Environ Toxicol 31:1450–1459
Nozaki M, Ito K, Miura C, Miura T (2013) Examination of digestive enzyme distribution in gut tract and functions of intestinal caecum, in Megascolecid earthworms (Oligochaeta: Megascolecidae) in Japan. Zool Sci 30:710–715
Nunes B (2010) The use of cholinesterases in ecotoxicology. Rev Environ Contam Toxicol 212:29–59
Ponsankar A, Vasantha-Srinivasan P, Thanigaivel A et al. (2018) Response of Spodoptera litura Fab. (Lepidoptera: Noctuidae) larvae to Citrullus colocynthis L. (Cucurbitales: Cucurbitaceae) chemical constituents: Larval tolerance, food utilization and detoxifying enzyme activities. Physiol Mol Plant Pathol 101:16–28
Pozo C, Martinez‐Toledo MV, Salmeron V, Rodelas B, Gonzalez‐Lopez J (1995) Effect of chlorpyrifos on soil microbial activity. Environ Toxicol Chem 14:187–192
Quistad GB, Casida JE (2000) Sensitivity of blood-clotting factors and digestive enzymes to inhibition by organophosphorus pesticides. J Biochem Mol Toxicol 14:51–56
Quistad GB, Liang SN, Fisher KJ, Nomura DK, Casida JE (2006) Each lipase has a unique sensitivity profile for organophosphorus inhibitors. Toxicol Sci 91:166–172
Racke KD (1993) Environmental fate of chlorpyrifos. Rev Environ Contam Toxicol 131:1–154
Rault M, Mazzia C, Capowiez Y (2007) Tissue distribution and characterization of cholinesterase activity in six earthworm species. Comp Biochem Physiol 147B:340–346
Rault M, Collange B, Mazzia C, Capowiez Y (2008) Dynamics of acetylcholinesterase activity recovery in two earthworm species following exposure to ethyl-parathion. Soil Biol Biochem 40:3086–3091
Ríos JM, Barceló G, Narváez C, Maldonado K, Sabat P (2014) Feeding and digestive responses to fatty acid intake in two South American passerines with different food habits. J Comp Physiol B 184:729–739
Rodrigues Macedo ML, Machado Freire MG (2011) Insect digestive enzymes as a target for pest control. Invertebr Surv J 8:190–198
Römbke J, Jänsch S, Didden W (2005) The use of earthworms in ecological soil classification and assessment concepts. Ecotoxicol Environ Saf 62:249–265
Ross MK, Edelmann MJ (2012) Carboxylesterases: a multifunctional enzyme involved in pesticide and lipid metabolism. In: Knaak JB, Timchalk C, Tornero-Velez R (eds) Parameters for pesticide QSAR and PBPK/PD models for human risk assessment. American Chemical Society, Washington, DC, pp 149–164
Sanchez-Hernandez JC, Aira M, Domínguez J (2014) Extracellular pesticide detoxification in the gastrointestinal tract of the earthworm Aporrectodea caliginosa. Soil Biol Biochem 79:1–4
Sanchez-Hernandez JC, Sandoval M, Pierart A (2017) Short-term response of soil enzyme activities in a chlorpyrifos-treated mesocosm: use of enzyme-based indexes. Ecol Indic 73:525–535
Singh B (2008) Organophosphorus-degrading bacteria: ecology and industrial applications. Nat Rev Microbiol 7:156–164
Skjemstad J, Baldock J (2008) Total and organic carbon. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis. CRC Press, Boca Raton, FL, pp 225–237
Sogorb MA, Vilanova E (2010) Detoxification of anticholinesterase pesticides. In: Satoh T, Gupta RC (Eds.) Anticholinesterase pesticides: metabolism, neurotoxicity, and epidemiology. Wiley, New Jersey, pp 121–132
Stygar D, Michalczyk K, Dolezych B et al. (2013) Digestive enzymes activity in subsequent generations of Cameraria ohridella larvae harvested from horse chestnut trees after treatment with imidacloprid. Pest Biochem Physiol 105:5–12
Thompson HM (1999) Esterases as markers of exposure to organophosphates and carbamates. Ecotoxicology 8:369–384
Velki M, Hackenberger BK (2013) Inhibition and recovery of molecular biomarkers of earthworm Eisenia andrei after exposure to organophosphate dimethoate. Soil Biol Biochem 57:100–108
Wheelock CE, Eder KJ, Werner I et al. (2005) Individual variability in esterase activity and CYP1A levels in Chinook salmon (Oncorhynchus tshawytscha) exposed to esfenvalerate and chlorpyrifos. Aquat Toxicol 74:172–192
Wheelock CE, Phillips BM, Anderson BS et al. (2008) Applications of carboxylesterase activity in environmental monitoring and toxicity identification evaluations (TIEs). Rev Environ Contam Toxicol 195:117–178
Yu YL, Wu XM, Li SN et al. (2006) An exploration of the relationship between adsorption and bioavailability of pesticides in soil to earthworm. Environ Pollut 141:428–433
Zhang BG, Rouland C, Lattaud C, Lavelle P (1993) Activity and origin of digestive enzymes in gut of the tropical earthworm Pontoscolex corethrurus. Eur J Soil Biol 29:7–11
Acknowledgements
We thank the regional Goverment Junta de Comunidades de Castilla-La Mancha (grant no. PEII-2014-001-P) and the Ministerio de Economía y Competitividad (grant no. CTM2014-53915-R) and for their financial support of this research. JM Ríos and AM Attademo thank to Vicerrectorado de Investigación of University Castilla-La Mancha for funding grants as visiting researchers. The comments of two anonymous reviewers were greatly appreciated and contributed to improve this manuscript.
Funding
This study was funded by the regional Government Junta de Comunidades de Castilla-La Mancha (grant no. PEII-2014-001-P) and the Spanish Ministerio de Economía y Competitividad (grant no. CTM2014-53915-R).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors, according to the Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes (Official Journal of the European Union L 276/33, 20-10-2010).
Rights and permissions
About this article
Cite this article
Sanchez-Hernandez, J.C., Ríos, J.M. & Attademo, A.M. Response of digestive enzymes and esterases of ecotoxicological concern in earthworms exposed to chlorpyrifos-treated soils. Ecotoxicology 27, 890–899 (2018). https://doi.org/10.1007/s10646-018-1914-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-018-1914-8