Abstract
Background and Aims
Absolute polymorphonuclear (PMN) counts in ascites define spontaneous bacterial peritonitis (SBP), a severe form of bacterial infection in liver cirrhosis. Bacterascites, another form of ascites infection, can progress to SBP or may resolve spontaneously but is not reflected by absolute PMN counts. We investigated whether the relative ascites PMN count (the absolute PMN count divided by the absolute leukocyte count) provides additional information to detect bacterascites or predict SBP.
Methods
Hospitalized patients with liver cirrhosis requiring paracentesis were stratified with respect to a diagnosis of bacterascites and SBP with a prospective follow-up for 1 year. Diagnostic power of relative PMN counts in ascites was evaluated by receiver operating characteristics curves.
Results
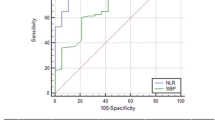
At inclusion, we observed 28/269 (10%) and 43/269 (16%) episodes of BA and SBP, respectively. Unlike absolute PMN counts, relative PMN counts in ascites were significantly elevated in bacterascites (p = 0.001). During follow-up, 16 and 30 further episodes of BA and SBP were detected, respectively. Relative PMN counts increased significantly once patients developed BA (p = 0.001). At a threshold of 0.20 for the relative PMN count, sensitivity, specificity, positive and negative predictive values for bacterascites which required antibiotic treatment were 83, 75, 26 and 98%, respectively (p < 0.001). Furthermore, a relative PMN count in ascites ≥0.13 and MELD score >17 was independent factors associated with occurrence of SBP during follow-up.
Conclusion
The relative PMN count is a cheap immunological marker linked to bacterascites and future SBP, which may help to stratify patients according to their risk of infection.





Similar content being viewed by others
Abbreviations
- SBP:
-
Spontaneous bacterial peritonitis
- BA:
-
Bacterascites
- PMN:
-
Polymorphonuclear
- HCC:
-
Hepatocellular carcinoma
- MELD:
-
Model for end-stage liver disease
- AUROC:
-
Area under the receiver operating characteristics curve
- ROC:
-
Receiver operating characteristics
- PPV:
-
Positive predictive value
- NPV:
-
Negative predictive value
References
Gustot T, Felleiter P, Pickkers P, et al. Impact of infection on the prognosis of critically ill cirrhotic patients: results from a large worldwide study. Liver Int Off J Int Assoc Study Liver. 2014;34:1496–1503. doi:10.1111/liv.12520.
Jalan R, Fernandez J, Wiest R, et al. Bacterial infections in cirrhosis: a position statement based on the EASL Special Conference 2013. J Hepatol. 2014;60:1310–1324. doi:10.1016/j.jhep.2014.01.024.
Arvaniti V, D’Amico G, Fede G, et al. Infections in patients with cirrhosis increase mortality four-fold and should be used in determining prognosis. Gastroenterology. 2010;139:1246–1256. doi:10.1053/j.gastro.2010.06.019.
Caly WR, Strauss E. A prospective study of bacterial infections in patients with cirrhosis. J Hepatol. 1993;18:353–358.
Fernández J, Navasa M, Gómez J, et al. Bacterial infections in cirrhosis: epidemiological changes with invasive procedures and norfloxacin prophylaxis. Hepatol Baltim Md. 2002;35:140–148. doi:10.1053/jhep.2002.30082.
Tandon P, Kumar D, Seo YS, et al. The 22/11 risk prediction model: a validated model for predicting 30-day mortality in patients with cirrhosis and spontaneous bacterial peritonitis. Am J Gastroenterol. 2013;108:1473–1479. doi:10.1038/ajg.2013.204.
Runyon BA, AASLD. Introduction to the revised American Association for the study of liver diseases practice guideline management of adult patients with ascites due to cirrhosis 2012. Hepatol Baltim Md. 2013;57:1651–1653. doi:10.1002/hep.26359.
European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53:397–417. doi:10.1016/j.jhep.2010.05.004.
Ortiz J, Soriano G, Coll P, et al. Early microbiologic diagnosis of spontaneous bacterial peritonitis with BacT/ALERT. J Hepatol. 1997;26:839–844.
Runyon BA. Monomicrobial nonneutrocytic bacterascites: a variant of spontaneous bacterial peritonitis. Hepatol Baltim Md. 1990;12:710–715.
Pelletier G, Lesur G, Ink O, et al. Asymptomatic bacterascites: is it spontaneous bacterial peritonitis? Hepatol Baltim Md. 1991;14:112–115.
Fernández J, Navasa M, Planas R, et al. Primary prophylaxis of spontaneous bacterial peritonitis delays hepatorenal syndrome and improves survival in cirrhosis. Gastroenterology. 2007;133:818–824. doi:10.1053/j.gastro.2007.06.065.
Bruns T, Lutz P, Stallmach A, Nischalke HD. Low ascitic fluid protein does not indicate an increased risk for spontaneous bacterial peritonitis in current cohorts. J Hepatol. 2015;63:527–528. doi:10.1016/j.jhep.2015.03.040.
Lutz P, Krämer B, Kaczmarek DJ, et al. A variant in the nuclear dot protein 52 kDa gene increases the risk for spontaneous bacterial peritonitis in patients with alcoholic liver cirrhosis. Dig Liver Dis Off J Ital Soc Gastroenterol Ital Assoc Study Liver. 2016;48:62–68. doi:10.1016/j.dld.2015.09.011.
Appenrodt B, Grünhage F, Gentemann MG, Thyssen L, Sauerbruch T, Lammert F. Nucleotide-binding oligomerization domain containing 2 (NOD2) variants are genetic risk factors for death and spontaneous bacterial peritonitis in liver cirrhosis. Hepatol Baltim Md. 2010;51:1327–1333. doi:10.1002/hep.23440.
Lutz P, Berger C, Langhans B, et al. A farnesoid X receptor polymorphism predisposes to spontaneous bacterial peritonitis. Dig Liver Dis Off J Ital Soc Gastroenterol Ital Assoc Study Liver. 2014;46:1047–1050. doi:10.1016/j.dld.2014.07.008.
Shapiro MF, Greenfield S. The complete blood count and leukocyte differential count: an approach to their rational application. Ann Intern Med. 1987;106:65–74.
Coburn B, Morris AM, Tomlinson G, Detsky AS. Does this adult patient with suspected bacteremia require blood cultures? JAMA. 2012;308:502–511. doi:10.1001/jama.2012.8262.
Kim JJ, Tsukamoto MM, Mathur AK, et al. Delayed paracentesis is associated with increased in-hospital mortality in patients with spontaneous bacterial peritonitis. Am J Gastroenterol. 2014;109:1436–1442. doi:10.1038/ajg.2014.212.
Gaieski DF, Mikkelsen ME, Band RA, et al. Impact of time to antibiotics on survival in patients with severe sepsis or septic shock in whom early goal-directed therapy was initiated in the emergency department. Crit Care Med. 2010;38:1045–1053. doi:10.1097/CCM.0b013e3181cc4824.
Soriano G, Esparcia O, Montemayor M, et al. Bacterial DNA in the diagnosis of spontaneous bacterial peritonitis. Aliment Pharmacol Ther. 2011;33:275–284. doi:10.1111/j.1365-2036.2010.04506.x.
Chu CM, Chang KY, Liaw YF. Prevalence and prognostic significance of bacterascites in cirrhosis with ascites. Dig Dis Sci. 1995;40:561–565.
Bruns T, Reuken PA, Stengel S, et al. The prognostic significance of bacterial DNA in patients with decompensated cirrhosis and suspected infection. Liver Int Off J Int Assoc Study Liver. 2016;36:1133–1142. doi:10.1111/liv.13095.
Zapater P, Francés R, González-Navajas JM, et al. Serum and ascitic fluid bacterial DNA: a new independent prognostic factor in noninfected patients with cirrhosis. Hepatol Baltim Md. 2008;48:1924–1931. doi:10.1002/hep.22564.
Fagan KJ, Rogers GB, Melino M, et al. Ascites bacterial burden and immune cell profile are associated with poor clinical outcomes in the absence of overt infection. PloS One. 2015;10:e0120642. doi:10.1371/journal.pone.0120642.
Schwabl P, Bucsics T, Soucek K, et al. Risk factors for development of spontaneous bacterial peritonitis and subsequent mortality in cirrhotic patients with ascites. Liver Int Off J Int Assoc Study Liver. 2015;35:2121–2128. doi:10.1111/liv.12795.
Runyon BA, Morrissey RL, Hoefs JC, Wyle FA. Opsonic activity of human ascitic fluid: a potentially important protective mechanism against spontaneous bacterial peritonitis. Hepatol Baltim Md. 1985;5:634–637.
Andreu M, Sola R, Sitges-Serra A, et al. Risk factors for spontaneous bacterial peritonitis in cirrhotic patients with ascites. Gastroenterology. 1993;104:1133–1138.
Francés R, Zapater P, González-Navajas JM, et al. Bacterial DNA in patients with cirrhosis and noninfected ascites mimics the soluble immune response established in patients with spontaneous bacterial peritonitis. Hepatol Baltim Md. 2008;47:978–985. doi:10.1002/hep.22083.
Colli A, Cocciolo M, Riva C, et al. Ascitic fluid analysis in hepatocellular carcinoma. Cancer. 1993;72:677–682.
Funding
This study was funded by a clinical leave stipend from the German Center for Infection Research to PL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Lutz, P., Goeser, F., Kaczmarek, D.J. et al. Relative Ascites Polymorphonuclear Cell Count Indicates Bacterascites and Risk of Spontaneous Bacterial Peritonitis. Dig Dis Sci 62, 2558–2568 (2017). https://doi.org/10.1007/s10620-017-4637-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4637-4