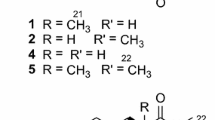
The ethyl acetate extract of the marine bacterial strain Streptomyces rochei sp. 81 was purified by silica gel column chromatography to obtain three diketopiperazines (1–3) and one straight chain fatty acid (4). The compounds were identified as cyclo(L-leu-N-ethenethiol-L-pro) (1), cyclo(L-Leu-L-Pro) (2), cyclo(L-Pro-D-Tyr) (3), and 2,4-dimethyldocasonoic acid (4) by HR-ESI-MS and NMR techniques. One new (1) and three known compounds (2–4) were screened for carbonic anhydrase-II (CA-II) inhibition and as a result 1 and 3 were found to be the most active compounds with IC50 of 9.8 ± 0.56 and 10.9 ± 0.88 μM, respectively. The results indicated that these two compounds (1 and 3) can be promising candidates for anticancer drugs.
Similar content being viewed by others
References
R. Solanki, M. Khanna, and R. Lal, Indian J. Microbiol., 48, 410 (2008).
A. Choudhary, L. M. Naughton, I. Montanchez, A. D. Dobson, and D. K. Rai, Mar. Drugs, 15, 272 (2017).
M. G. Watve, R. Tickoo, M. M. Jog, and B. D. Bhole, Arch. Microbiol., 176, 386 (2001).
A. C. Ward and N. Bora, Curr. Opin. Microbiol., 9, 279 (2006).
O. Zaborina, M. Latus, J. Eberspacher, and L. A. Golovleva, J. Bacteriol., 177, 229 (1995).
U. Anukool, W. H. Gaze, and E. M. Wellington, Appl. Environ. Microbiol., 70, 5222 (2004).
C. Kotake, T. Yamasaki, T. Moriyama, M. Shinoda, N. Komiyama, T. Furumai, M. Konishi, and T. Oki, J. Antibiot., 45, 1442 (1992).
G. S. Kanini, E. A. Katsifas, A. L. Savvides, and A. D. Karagouni, BioMed Res. Int., 2013, 387230 (2013).
L. A Golovleva, O. Zaborina, R. Pertsova, B. Baskunov, Y. Schurukhin, and S. Kuzmin, Biodegradation, 2, 201 (1991).
A. Yokota, O. Yamauchi, and F. Tomita, Lett. Appl. Microbiol., 21, 330 (1995).
A. Acharyabhatta, S. K. Kandula, and R. Terli, Int. J. Microbiol., 2013, 276921 (2013).
N. E. El-Naggar, Int. J. Pharmacol., 11, 644 (2015).
T. Wakabayashi, R. Kageyama, N. Naruse, N. Tsukahara, Y. Funahashi, K. Kitoh, and Y. Watanabet, J. Antibiot., 50, 671 (1997).
C. T. Supuran and A. Scozzafava, Expert Opin. Ther. Pat., 10, 575 (2000).
E. Masini, F. Carta, A. Scozzafava, and C. T. Supuran, Expert Opin. Ther. Pat., 23, 705 (2013).
A. H. Lee and I. F. Tannock, Cancer Res., 58, 1901 (1998).
M. L. Frazier, B. J. Lilly, E. F. Wu, T. Ota, and D. Hewett-Emmett, Pancreas, 5, 507 (1990).
S. Parkkila, H. Rajaniemi, A. K. Parkkila, J. Kivela, A. Waheed, S. Pastorekova, J. Pastorek, and W. S. Sly, Proc. Natl. Acad. Sci., 97, 2220 (2000).
V. Achal and X. Pan, Curr. Microbiol., 62, 894 (2011).
M. I. Gulcin, S. Beydemir, O. I. Kufrevioglu, and C. T. Supuran, Chem. Biol. Drug Des., 77, 494 (2011).
V. De Luca, S. Del Prete, C. T. Supuran, and C. Capasso, J. Enzyme Inhib. Med. Chem., 30, 277 (2015).
M. M. Alshaibani, N. Zin, J. Jalil, N. M. Sidik, S. J. Ahmad, N. Kamal, and R. Edrada-Ebel, J. Microbiol. Biotechnol., 27, 1249 (2017).
A. Noel, S. Ferron, I. Rouaud, N. Gouault, J. P. Hurvois, and S. Tomasi, Molecules, 22, 371 (2017).
R. M. Abed, B. Zein, A. Al-Thukair, and D. de Beer, Syst. Appl. Microbiol., 30, 319 (2007).
S. Kumar, G. Stecher, M. Li, C. Knyaz, and K. Tamura, Mol. Biol. Evol., 35, 1547 (2018).
A. L. Khan, M. Waqas, S. M. Kang, A. Al-Harrasi, J. Hussain, A. Al-Rawahi, S. Al-Khiziri, I. Ullah, L. Ali, H. Y. Jung, and I. J. Lee, J. Microbiol., 52, 689 (2014).
N. Kumar, C. Mohandas, B. Nambisan, D. S. Kumar, and R. S. Lankalapalli, World J. Microbiol. Biotechnol., 29, 355 (2013).
J. Cason, G. Lange, and H. R. Urscheler, Tetrahedron, 20, 1955 (1964).
R. P. Shank, D. R. Doose, A. J. Streeter, and M. Bialer, Epilepsy Res., 63, 103 (2005).
Acknowledgment
The project was supported by the funded project (BFP/RGP/CBS/18/011) awarded by The Oman Research Council (TRC). The study was also funded by the Gulf Cooperation Council (GCC) collaborative work between Kuwait University, Kuwait, Sultan Qaboos University, Oman and University of Nizwa, Oman.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 2, March–April, 2023, pp. 292–295.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Al-Rawahi, A.N., Abed, R.M.M., Rehman, N.U. et al. New Sulfur-Containing Diketopiperazine from Marine-Derived Bacteria Streptomyces rochei sp. 81 with Potent Carbonic Anhydrase II Inhibition. Chem Nat Compd 59, 346–350 (2023). https://doi.org/10.1007/s10600-023-03990-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-03990-0