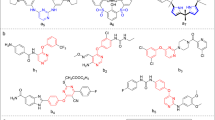
Herein, we present the design, synthesis, and biological activity of novel iminocoumarin imidazo[4,5-b]pyridine derivatives. The prepared compounds were designed to study the type of substituent in position 6 of the coumarin nucleus as well as the type of the substituent at the N atom of imidazo[4,5-b]pyridine core for their effect on the biological activity. Therefore, all compounds were tested for their antiproliferative action on several human cancer cell lines in vitro, for their antioxidative activity, antibacterial activity on several bacterial strains, and for their antiviral activity on several viruses. The results of the evaluation of biological activity revealed that the tested derivatives did not display significant biological activities. The majority of the tested compounds were not active at all, while some derivatives showed low activity in these assays. Therefore, we could conclude that the biological potential of 6-substituted iminocoumarin derivatives is very low – the substitution in position 6 of the coumarin nucleus, in comparison to 7-substituted iminocoumarins, strongly decreases biological activity.


Similar content being viewed by others
References
Sandhu, S.; Bansal, Y.; Silakari, O.; Bansal, G. Bioorg. Med. Chem. 2015, 22, 3806.
Song, X.-F.; Fan, J.; Liu, L.; Liu, X.-F.; Gao, F. Arch. Pharm. 2020, 353, 2000025..
Prusty, J. S.; Kumar, A. Mol. Diversity 2020, 24, 1367.
Li, H.; Cai, L.; Chen Z. In Advances in Chemical Sensors; Wang, W., Ed.; InTech: Rijeka, 2011, p.121.
Khemakhem, K.; Ammar, H.; Abid, S.; El Gharbi, R.; Fery-Forgues, S. Dyes Pigm. 2013, 99, 594.
Sinha, S.; Singh, K.; Ved, A.; Hasan, S. M.; Mujeeb, S. Mini- Rev. Med. Chem. 2022, 22, 1314.
Murray, R. D. H.; Mendez, J.; Brown, S. A. The Natural Coumarins: Ocurrence, Chemistry and Biochemistry; Wiley: Chichester, 1982.
Sharifi-Rad, J.; Cruz-Martins, N.; López-Jornet, P.; Pons-Fuster Lopez, E.; Harun, N.; Yeskaliyeva, B.; Beyatli, A.; Sytar, O.; Shaheen, S.; Sharopov, F.; Taheri, Y.; Docea, A. O.; Calina, D.; Cho, W. C. Oxid. Med. Cell. Longevity 2021, 6492346.
Roussaki, M.; Kontogiorgis, C. A.; Hadjipavlou-Litina, D.; Hamilakis, S.; Detsi, A. Bioorg. Med. Chem. Lett. 2020, 20, 3889.
Prusty, J. S.; Kumar, A. Mol. Diversity 2020, 24, 1367.
Carneiro, A.; Matos, M. J.; Uriarte, E.; Santana, L. Molecules 2021, 26, 501.
Tsay, S.-C.; Lin, S.-Y.; Huang, W.-C.; Hsu, M.-H.; Hwang, K. C.; Lin, C.-C.; Horng, J.-C.; Chen, I.-C.; Hwu, J. R.; Shieh, F.-K.; Leyssen, P.; Neyts, J. Molecules 2016, 21, 228.
Feng, D.; Zhang, A.; Yang, Y.; Yang, P. Arch. Pharm. 2020, 353, 1900380.
Emami, S.; Dadashpour, S. Eur. J. Med. Chem. 2015, 102, 611.
Thakur, A.; Singla, R.; Jaitak, V. Eur. J. Med. Chem. 2015, 101, 476.
Silverman, R. B. The Organic Chemistry of Drug Design and Drug Action; Elsevier Academic Press: Amsterdam, 2004, 2nd ed.
Dorababu, A. Eur. J. Med. Chem. Rep. 2021, 2, 100006.
Zhang, L.; Xu, Z. Eur. J. Med. Chem. 2019, 181, 111587.
Gabr, M. T.; Ibrahim, M. M. H.; Tripathi, A.; Prabhakar, C. Chemosensors 2019, 7, 34.
Dai, M.; Sarkar, S.; Song, C. W.; Reo, Y. J.; Yang, Y. J.; Ahn, K. H. ChemPhotoChem 2020, 4, 721.
Chemate, S. B.; Sekar, N. J. Fluoresc. 2015, 25, 1615.
Abid-Jarraya, N.; Khemakhem, K.; Turki-Guermazi, H.; Abid, S.; Saffon, N.; Fery-Forgues, S. Dyes Pigm. 2016, 132, 177.
Perin, N.; Cindrić, M.; Vervaeke, P.; Liekens, S.; Mašek, T.; Starčević, K.; Hranjec, M. Med. Chem. (Sharjah, United Arab Emirates) 2021, 17, 13.
Boček, I.; Hranjec, M.; Vianello, R. J. Mol. Liq. 2022, 355, 118982.
Boček, I.; Starčević, K.; Novak Jovanović, I.; Vianello, R.; Hranjec, M. J. Mol. Liq. 2021, 342, 117527.
Vasylevska, A. S.; Karasyov, A. A.; Borisov, S. M.; Krause, C. Anal. Bioanal. Chem. 2007, 387, 2131.
Boček Pavlinac, I.; Zlatić, K.; Persoons, L.; Daelemans, D.; Banjanac, M.; Radovanović, V.; Butković, K.; Kralj, M.; Hranjec, M. Molecules 2023, 28, 34.
(a) Grgičević, I.; Mikulandra, I.; Bukvić, M.; Banjanac, M.; Radovanović, V.; Habinovec, I.; Bertoša, B.; Novak, P. Int. J. Antimicrob. Agents 2020, 56, 106147. (b) https://clsi.org/
Perin, N.; Roškarić, P.; Sović, I.; Boček, I.; Starčević, K.; Hranjec, M.; Vianello, R. Chem. Res. Toxicol. 2018, 31, 974.
Cindrić, M.; Sović, I.; Mioč, M.; Hok, L.; Boček, I.; Roškarić, P.; Butković, K.; Martin-Kleiner, I.; Starčević, K.; Vianello, R.; Kralj, M.; Hranjec, M. Antioxidants 2019, 8, 477.
Gülçin, I.; Huyut, Z.; Elmastas, M.; Aboul-Enein, H. Y. Arab. J. Chem. 2010, 3, 43.
Beč, A.; Racané, L.; Žonja, L.; Persoons, L.; Daelemans, D.; Starčević, K.; Vianello, R.; Hranjec, M. RSC Med. Chem. 2023, 14, 957.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2024, 60(1/2), 75–82
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pavlinac, I.B., Starčević, K., Persoons, L. et al. Novel iminocoumarin imidazo[4,5-b]pyridine derivatives: design, synthesis, and biological evaluation. Chem Heterocycl Comp 60, 75–82 (2024). https://doi.org/10.1007/s10593-024-03296-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-024-03296-1