Abstract
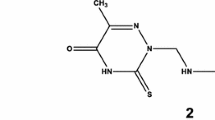
New series of pyrimidinone-5-carbonitriles 3a–i, 4a–e, 5a–c, 6 and 7 have been synthesized and explored for their activities as anticancer, antibacterial and antifungal agents. Investigation of the anticancer activity revealed that several newly synthesized derivatives displayed potent cytotoxic activity against different cancer cells. Among them, compound 3g was the most potent on the MCF-7, A549 and Caco-2 cell lines (IC50 = 1.42, 1.98 and 9.50 μM, respectively), as compared with 5-fluorouracil (IC50 = 1.71, 10.32 and 20.22 μM, respectively), while compound 3f was found especially effective against MCF-7 and Caco-2 cell lines (IC50 = 1.48 and 16.15 μM, respectively). Furthermore, the antimicrobial evaluation showed that compounds 3f and 3g have potent antibacterial activity against Gram-positive bacteria Staphylococcus aureus (MIC = 4 and 8 μg/mL, respectively) and promising activity against Escherichia coli (IZ. = 19 and 17 mm, respectively). Meanwhile, compound 4b displayed the highest activity toward Bacillus subtilis (MIC = 8 μg/mL). In particular, the results suggested that hydrazone derivatives bearing heterocyclic rings 3f and 3g are good lead compounds for the future design of more potent anticancer or antimicrobial agents.






Similar content being viewed by others
References
A.M. Fargualy, N.S. Habib, K.A. Ismail, A.M.M. Hassan, M.T.M. Sarg, Eur. J. Med. Chem. 66, 276 (2013)
J. Esteban-Gamboa, R. Balzarini, E.De Esnouf, M.J. Clercq, M.J.Pérez-Pérez Camarasa, J. Med. Chem. 43, 971 (2000)
R.C. Reynolds, A. Tiwari, J.E. Harwell, D.G. Gordon, B.D. Garrett, K.S. Gilbert, S.M. Schmid, W.R. Waud, R.F. Struck, J. Med. Chem. 43, 1484 (2000)
V.V. Iyer, G.W. Griesgraber, M.R. Radmer, E.J. McIntee, C.R. Wagner, J. Med. Chem. 43, 2266 (2000)
M.T. Cocco, C. Congiu, V. Lilliu, V. Onnis, Bioorg. Med. Chem. 14, 366 (2006)
N.J. Curtin, H.C. Barlow, K.J. Bowman, A.H. Calvert, R. Davison, B.T. Golding, B. Huang, P.J. Loughlin, D.R. Newell, P.G. Smith, R.J. Griffin, J. Med. Chem. 47, 4905 (2004)
A. Conejo-Garcia, M.E. Garcia-Rubino, J.A. Marchal, M.C. Nunez, A. Ramirez, S. Cimino, M.A. Garcia, A. Aranega, M.A. Gallo, J.M. Campos, Eur. J. Med. Chem. 46, 3795 (2011)
L.C. Lopez-Cara, A. Conejo-Garcia, J.A. Marchal, G. Macchione, O. Cruz-Lopez, H. Boulaiz, M.A. Garcia, F. Rodriguez-Serrano, A. Ramirez, C. Cativiela, A.I. Jimenez, J.M. Garcia-Ruiz, D. Choquesillo-Lazarte, A. Aranega, J.M. Campos, Eur. J. Med. Chem. 46, 249 (2011)
F. Morales, A. Ramirez, A. Conejo-Garcia, C. Morata, J.A. Marchal, J.M. Campos, Eur. J. Med. Chem. 76, 118 (2014)
N.S. Habib, R. Soliman, K. Ismail, A.M. Hassan, M. Sarg, Boll. Chim. Farm. 142, 396 (2003)
D.W. Boykin, A. Kuma, M. Baji, G. Xiao, W.D. Wilson, B.C. Bender, D.R. McCurdy, J.E. Hall, R.R. Tidwell, Eur. J. Med. Chem. 32, 965 (2004)
S.F. Chowdhury, V.B. Villamor, R.H. Guerrero, I. Leal, R. Brun, S.L. Croft, J.M. Goodman, L. Maes, L.M. Ruiz-Perez, D.G. Pacanowska, I.H. Gilbert, J. Med. Chem. 42, 4300 (1999)
V.J. Ram, D.A.V. Berghe, A.J. Vlietinck, J. Heterocyclic Chem. 21, 1307 (1984)
M.T. Cocco, C. Congiu, V. Onnis, M.L. Schivo, A. De Logu, Farmaco 50, 73 (1995)
K.S. Jain, T.S. Chitre, P.B. Miniyar, M.K. Kathiravan, V.S. Bendre, V.S. Veer, S.R. Shahane, C.J. Shishoo, Curr. Sci. 90, 793 (2006)
D. Bixby, M. Talpaz, Leukemia 25, 7 (2010)
H.T. Abdel-Mohsen, F.A.F. Ragab, M.M. Ramala, H.I. El-Diwani, Eur. J. Med. Chem. 45, 2336 (2010)
E.S. Al-Abdullah, A.R.M. Al-Obaid, O.A. Al-Deeb, E.E. Habib, A.A. El-Emam, Eur. J. Med. Chem. 46, 4642 (2011)
A.T. Taher, A.A. Helwa, Chem. Pharm. Bull. 60(4), 521 (2012)
M.M. Mohamed, A.K. Khalil, E.M. Abbass, A.M. El-Naggar, Synth. Commun. 47(16), 1441 (2017)
A.T. Taher, S.M. Abou-Seri, Molecules 17, 9868 (2012)
S. Rollas, S.G. Küçükgüzel, Molecules 12, 1910 (2007)
S. Kambe, K. Saito, H. Kishi, Synth. 287 (1979)
N.W. Roehm, G.H. Rodgers, S.M. Hatfield, A.L. Glasebrook, J. Immunol. Methods 142, 257 (1991)
H. Jahangirian, M.J. Haron, M.H. Shah, Y. Abdollahi, M. Rezayi, N. Vafaei, Digest. J. Nanomater. Biostruct. 8, 1263 (2013)
J.M. Andrews, J. Antimicrob. Chemother. 48, 5 (2001)
Z. Zhang, X. Yan, G. Zhang, Q. Liu, N. Ma, T. Liu, L. Shi, Tetrahedron 72, 3077 (2016)
X. Yan, Z. Zhang, G. Zhang, N. Ma, Q. Liu, T. Liu, L. Shi, Tetrahedron 72, 4245 (2016)
F.A. Attaby, S.M. Eldin, E.A.Z. Hanafi, Arch. Pharm. Res. 20(6), 620 (1997)
F.A. Attaby, S.M. Eldin, Z. Naturforsch. 54, 788 (1999)
B.F. Abdel-Wahab, S.F. Mohamed, A.E. Amr, M.M. Abdalla, Monatsh. Chem. 139, 1083 (2008)
M.M. Abdalla, B.F. Abdel-Wahab, A.G.E. Amr, Monatshefte für Chemie Chem. Monthly 140, 129 (2008)
Acknowledgements
The authors would like to express their gratitude to Dr. Mohammed Fathy, Department of Pharmacology, Faculty of Pharmacy, Al-Azhar University for performing the cytotoxic activity of the synthesized compounds. The authors would like to express their sincere thanks to the Department of Microbiology, Misr University for Science and Technology, for carrying out the antimicrobial screening.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Helwa, A.A., Gedawy, E.M., Abou-Seri, S.M. et al. Synthesis and bioactivity evaluation of new pyrimidinone-5-carbonitriles as potential anticancer and antimicrobial agents. Res Chem Intermed 44, 2685–2702 (2018). https://doi.org/10.1007/s11164-018-3254-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3254-y