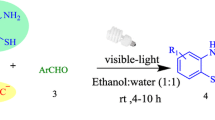
Functionalized benzofurans are attractive chemical motifs with various pharmacological and physiological applications, therefore chemists are constantly working to develop new and superior methods for their synthesis. During the past decade, significant upsurge of interest can be found in the application of visible-light-mediated chemical reactions for the synthesis of a variety of molecules, including benzofuran derivatives. This would be the first comprehensive review generalizing synthetic methodologies toward functionalized benzofurans under visible-light-assisted conditions. The review will describe different possible visible-light-mediated synthesis and briefly describe the mechanisms of the reactions.

















































Similar content being viewed by others
References
Chand, K.; Rajeshwari; Hiremathad, A.; Singh, M.; Santos, M. A.; Keri, R. S. Pharmacol. Rep. 2017, 69, 281.
Miao, Y.-h.; Hu, Y.-h.; Yang, J.; Liu, T.; Sun, J.; Wang, X.-j. RSC Adv. 2019, 9, 27510.
Abbas, A. A.; Dawood, K. M. RSC Adv. 2023, 13, 11096.
Asif, M. J. Anal. Pharm. Res. 2016, 3, 00048.
(a) Heravi, M. M.; Zadsirjan, V.; Hamidi, H.; Amiri, P. H. T. RSC Adv. 2017, 7, 24470. (b) Mahendra, C. K.; Tan, L. T. H.; Lee, W. L.; Yap, W. H.; Pusparajah, P.; Low, L. E.; Tang, S. Y.; Chan, K. G.; Lee, L. H.; Goh, B. H. Front. Pharmacol. 2020 11, 366. (c) De Amicis, F.; Aquila, S.; Morelli, C.; Guido, C.; Santoro, M.; Perrotta, I.; Mauro, L.; Giordano, F.; Nigro, A.; Andò, S.; Panno, M. L. Mol. Cancer 2015, 14, 130. (d) Zheng, G.; Luo, S.; Li, S.; Hua, J.; Li, W.; Li, S. Phytochemistry 2018, 148, 57. (e) Kapche, D. W. F. G.; Lekane, N. M.; Kulabas, S. S.; Ipek, H.;Tok, T. T.; Ngadjui, B. T.; Demirtas, I.; Tumer, T. B. Phytochemistry 2017, 141, 70. (f) Almutairi, B. O.; Alsayadi, A. I.; Abutaha, N.; Al-Mekhlafi, F. A.; Wadaan, M. A. BioMed. Res. Int. 2023, 9337763. (g) Ajitha, B.; Reddy, Y. A. K.; Reddy, P. S. Spectrochim. Acta, Part A 2014, 121, 164.
Yun, Y.; Miao, Y.; Sun, X.; Sun, J.; Wang, X. J. Enzyme Inhib. Med. Chem. 2021, 36, 1345.
Kushwaha, P.; Fatima, S.; Upadhyay, A.; Gupta, S.; Bhagwati, S.; Baghel, T.; Siddiqi, M. I.; Nazir, A.; Sashidhara, K. V. Bioorg. Med. Chem. Lett. 2019, 29, 66.
Chowdhury, S. R.; Gu, J.; Hu, Y.; Wang, J.; Lei, S.; Tavallaie, M. S.; Lam, C.; Lu, D.; Jiang, F.; Fu, L. Eur. J. Med. Chem. 2021, 222, 113541.
Hasanvand, Z.; Motahari, R.; Nadri, H.; Moghimi, S.; Foroumadi, R.; Ayati, A.; Akbarzadeh, T.; Bukhari, S. N. A.; Foroumadi, A. Front. Chem. 2022, 10, 882191.
Xue, S.-T.; Zhang, L.; Xie, Z.-S.; Jin, J.; Guo, H.-F.; Yi, H.; Liu, Z.-Y.; Li, Z.-R. Eur. J. Med. Chem. 2020, 200, 112465.
Zhou, Z.-y.; Sun, L.-q.; Han, X.-y.; Wang, Y.-j.; Xie, Z.-s.; Xue, S.-t.; Li, Z.-r. J. Med. Chem. 2023, 66, 1742.
Kushwaha, P.; Tripathi, A. K.; Gupta, S.; Kothari, P.; Upadhyay, A.; Ahmad, N.; Sharma, T.; Siddiqi, M. I.; Trivedi, R.; Sashidhara, K. V. Eur. J. Med. Chem. 2018, 156, 103.
Bernal, F. A.; Gerhards, M.; Kaiser, M.; Wünsch, B.; Schmidt, T. J. Eur. J. Med. Chem. 2020, 205, 112493.
Bernal, F. A.; Schmidt, T. J. Molecules 2023, 28, 3399.
Lei, S.; Hu, Y.; Yuan, C.; Sun, R.; Wang, J.; Zhang, Y.; Zhang, Y.; Lu, D.; Fu, L.; Jiang, F. Eur. J. Med. Chem. 2022, 229, 114032.
Elsebaie, M. M.; Nour El-Din, H. T.; Abutaleb, N. S.; Abuelkhir, A. A.; Liang, H.-W.; Attia, A. S.; Seleem, M. N.; Mayhoub, A. S. Eur. J. Med. Chem. 2022, 234, 114204.
Madieh, N. S.; Tanna, S.; Alqurayn, N. A.; Vaideanu, A.; Schatzlein, A.; Brucoli, F. RSC Adv. 2023, 13, 8420.
Jin, L.-P.; Xie, Q.; Huang, E.-F.; Wang, L.; Zhang, B.-Q.; Hu, J.-S.; Wan, D. C.-C.; Jin, Z.; Hu, C. Bioorg. Chem. 2020, 95, 103566.
Baldisserotto, A.; Demurtas, M.; Lampronti, I.; Moi, D.; Balboni, G.; Vertuani, S.; Manfredini, S.; Onnis, V. Eur. J. Med. Chem. 2018, 156, 118.
Trang, N. V.; Thuy, P. T.; Thanh, D. T. M.; Son, N. T. RSC Adv. 2021, 11, 12971.
Huong, L. T.; Thu, P. A.; Dao, P. T.; Huong, D. T. M.; Cuong, P. V.; Dang, N. H. Chem. Biodiversity 2021, 18, e2100518.
Kenchappa, R.; Bodke, Y. D. Chem. Data Collect. 2020, 28, 100453.
Rida, S. M.; El-Hawash, S. A.; Fahmy, H. T. Y.; Hazza, A. A.; El-Meligy, M. M. M. Arch. Pharm. Res. 2006, 1, 16.
Zhang, Y.; Wang, R.; Zhang, T.; Yan, W.; Chen, Y.; Zhang, Y.; Zhou, M. Chin. Chem. Lett. 2019, 30, 653.
Zhao, G.; Tian, X.; Wang, J.; Cheng, M.; Zhang, T.; Wang, Z. New J. Chem. 2021, 45, 1286.
Xu, H.; Hou, Z.; Liang, Z.; Guo, M.-B.; Su, X.; Guo, C. Chin. J. Chem. 2019, 37, 1245.
Dawood, K. M. Expert Opin. Ther. Pat. 2019, 29, 841.
Chiummiento, L.; D'Orsi, R.; Funicello, M.; Lupattelli, P. Molecules 2020, 25, 2327.
Abu-Hashem, A. A.; Hussein, H. A. R.; Aly, A. S.; Gouda, M. A. Synth. Comm. 2014, 44, 2285.
More, K. R.; J. Chem. Pharm. Res. 2017, 9, 210.
Shaw, M. H.; Twilton, J.; MacMillan, D. W. C. J. Org. Chem. 2016, 81, 6898.
Jana, S.; Verma, A.; Kadu, R.; Kumar, S. Chem. Sci. 2017, 8, 6633.
Wang, L.; Zhang, M.; Zhang, Y.; Liu, Q.; Zhao, X.; Li, J.-S.; Luo, Z.; Wei, W. Chin. Chem. Lett. 2020, 31, 67.
Li, X.; Xu, Z.; Wang, L.; Wang, F.; Yang, J.; Li, P. ChemPhotoChem 2021, 5, 142.
Chen, H.; Yan, Y.; Zhang, N.; Mo, Z.; Xu, Y.; Chen, Y. Org. Lett. 2021, 23, 376.
Chen, F.; Huang, X.; Yang, C.; Jiang, H.; Zeng, W. J. Org. Chem. 2021, 86, 14572.
Liu, J.; Tang, S.; Wang, S.; Cao, M.; Zhao, J.; Zhang, P.; Li, P. J. Org. Chem. 2022, 87, 9250.
Wang, H.; Huang, Y.; Wu, Q.; Lu, J.; Xu, Y.-Ll; Chen, Y.-y. J. Org. Chem. 2022, 87, 13288.
Mo, F.; Qui, D.; Zhang, L.; Wang, J. Chem. Rev. 2021, 121, 5741.
Xia, Z.; Khaled, O.; Mouriès-Mansuy, V.; Ollivier, C.; Fensterbank, L. J. Org. Chem. 2016, 81, 7182.
Alcaide, B.; Almendros, P.; Busto, E.; Lázaro-Milla, C. J. Org. Chem. 2017, 82, 2177.
Micic, N.; Polyzos, A. Org. Lett. 2018, 20, 4663.
Guo, W.; Lu, L.-Q.; Wang, Y.; Wang, Y.-N.; Chen, J.-R.; Xiao, W.-J. Angew. Chem., Int. Ed. 2015, 54, 2265.
Ghosh, S.; Das, J.; Saikh, F. Tetrahedron Lett. 2012, 53, 5883.
Sagadevan, A.; Hwang, K. C. Adv. Synth. Cat. 2012, 354, 3421.
Zhang, L.; Si, X.; Rominger, F.; Hashmi, A. S. K. J. Am. Chem. Soc. 2020, 142, 10485.
Koy, M.; Bellotti, P.; Katzenburg, F.; Daniliuc, C. G.; Glorius, F. Angew. Chem., Int. Ed. 2020, 59, 2375.
Li, Y.; Zhang, S.-Y.; Yan, X.-L.; Zhu, J.; Luo, K.; Wu, L. Org. Lett. 2023, 25, 4720.
Blum, T. R.; Zhu, Y.; Nordeen, S. A.; Yoon, T. P. Angew. Chem., Int. Ed. 2014, 53, 11056.
Borra, S.; Chandrashekhar, D.; Khound, S.; Maurya, R. A. Org. Lett. 2017, 19, 5364.
Liu, W.; Chen, N.; Yang, X.; Li, L.; Li, C.-J. Chem. Commun. 2016, 52, 13120.
Song, T.; Zhou, B.; Peng, G.-W.; Zhang, Q.-B.; Wu, L.-Z.; Liu, Q.; Wang, Y. Chem.–Eur. J. 2014, 20, 678.
Münster, N.; Parker, N. A.; van Dijk, L.; Paton, R. S.; Smith, M. D. Angew. Chem., Int. Ed. 2017, 56, 9468.
Ghosh, S.; Das, J. Tetrahedron Lett. 2011, 52, 1112.
Xie, S.; Li, Y.; Liu, P.; Sun, P. Org. Lett. 2020, 22, 8774.
He, X.; Ling, J.; Fang, Z.; Zhou, L. J. Org. Chem. 2023, 88, 7586.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2024, 60(1/2), 1–22
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kushwaha, P. Visible-light-mediated synthesis of functionalized benzofurans: an update. Chem Heterocycl Comp 60, 1–22 (2024). https://doi.org/10.1007/s10593-024-03284-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-024-03284-5